-
Name
2-Aminobenzonitrile
- EINECS 217-549-9
- CAS No. 1885-29-6
- Article Data135
- CAS DataBase
- Density 1.14g/cm3
- Solubility insoluble in water
- Melting Point 45-48 °C(lit.)
- Formula C7H6N2
- Boiling Point 267.5 °C at 760 mmHg
- Molecular Weight 118.138
- Flash Point 122.2 °C
- Transport Information
- Appearance yellow solid
- Safety 26-36/37
- Risk Codes 20/21/22-36/37/38-43
-
Molecular Structure
-
Hazard Symbols
 Xi,
Xi, T,
T, Xn
Xn
- Synonyms Anthranilonitrile (6CI,7CI,8CI);1-Amino-2-cyanobenzene;2-Aminobenzonitrile;2-Cyano-1-aminobenzene;2-Cyanoaniline;2-Cyanophenylamine;o-Aminobenzonitrile;o-Cyanoaniline;Benzonitrile, 2-amino-;
- PSA 49.81000
- LogP 1.72168
Synthetic route

| Conditions | Yield |
|---|---|
| With sodium tetrahydroborate In methanol; water at 20℃; for 0.0666667h; Sealed tube; Green chemistry; chemoselective reaction; | 99% |
| With cyclohexa-1,4-diene; 5%-palladium/activated carbon In methanol at 120℃; for 0.0833333h; Microwave irradiation; | 98% |
| With borane-ammonia complex In methanol; water at 20℃; for 0.166667h; | 98% |
-
-
28144-70-9
anthranilic acid amide

-
-
1885-29-6
anthranilic acid nitrile

| Conditions | Yield |
|---|---|
| With 3 Angstroem melecular sieves at 180 - 500℃; under 0.005 Torr; for 0.666667h; Pyrolysis; | 99% |
| With thionyl chloride In toluene at 90 - 100℃; | 91.6% |
| With oxalyl dichloride; 2,3-bis(4-methoxyphenyl)cyclopropenone; 1,8-diazabicyclo[5.4.0]undec-7-ene In dichloromethane for 5h; Reflux; | 89% |
| With vanadium oxide on hydrotalcite (V/HT) In 1,3,5-trimethyl-benzene for 24h; Reflux; | 79% |
| With sieve-supported lead catalyst at 200℃; under 2250.23 Torr; for 0.3h; |

| Conditions | Yield |
|---|---|
| With tri-tert-butyl phosphine; [Pd2(dba)5]; zinc In tetrahydrofuran; acetonitrile at 70℃; for 2h; | 99% |

| Conditions | Yield |
|---|---|
| With C68H64Cl2N6P2Ru2(4+)*2F6P(1-)*2Cl(1-); caesium carbonate In N,N-dimethyl-formamide at 100℃; for 24h; Inert atmosphere; Green chemistry; | 96.2% |

| Conditions | Yield |
|---|---|
| Stage #1: potassium cyanide With acetic acid In ethylene glycol at 20℃; Sealed tube; Stage #2: 2-bromoaniline With P(t-Bu)3 Palladacycle Gen. 3; potassium acetate In 1,4-dioxane; water at 60℃; for 16h; Sealed tube; | 95% |

| Conditions | Yield |
|---|---|
| With eosin; carbon tetrabromide; N,N-dimethyl-formamide In acetonitrile at 25℃; for 14h; Irradiation; Inert atmosphere; | 93% |
| Stage #1: 2-aminobenzaldehyde oxime With triethylamine In dichloromethane at 0 - 23℃; Inert atmosphere; Stage #2: With methanesulfonyl chloride In dichloromethane at 0 - 23℃; for 5h; Inert atmosphere; | 69% |
| With formamide at 140 - 150℃; |
-
-
31656-77-6
2-azidobenzonitrile

-
-
1885-29-6
anthranilic acid nitrile

| Conditions | Yield |
|---|---|
| With Decaborane; palladium on activated charcoal In methanol at 20℃; for 2.5h; | 91% |
| With hexamethyldisilathiane In methanol for 1.5h; Ambient temperature; | 90% |
| With dicobalt octacarbonyl In water at 20℃; for 5h; | 87% |
| In decalin at 144℃; for 5h; Product distribution; | |
| With 2,6-di-tert-butyl-4-methyl-phenol In decalin at 161.3℃; Rate constant; Kinetics; Thermodynamic data; ΔEact, ΔSact, other temperatures, other solvent; |
-
-
603-35-0
triphenylphosphine

-
-
174969-51-8
N-(2-cyanophenyl)cyanothioformamide

-
A
-
1192690-93-9
3-amino-1H-indole-2-carbonitrile

-
B
-
1885-29-6
anthranilic acid nitrile

| Conditions | Yield |
|---|---|
| Stage #1: N-(2-cyanophenyl)cyanothioformamide With toluene-4-sulfonic acid In methanol at 20℃; for 0.0833333h; Stage #2: triphenylphosphine In methanol at 20℃; for 0.833333h; | A 90% B 8% |


| Conditions | Yield |
|---|---|
| With hydrogenchloride multistep reaction; other arylmagnesium bromides; product yields; | A 89% B n/a |


| Conditions | Yield |
|---|---|
| With tetrabutylammomium bromide; sodium fluoride; palladium diacetate In water at 150℃; for 0.333333h; | 88% |

| Conditions | Yield |
|---|---|
| With potassium phosphate In N,N-dimethyl-formamide at 120℃; for 2h; | 88% |

| Conditions | Yield |
|---|---|
| With palladium diacetate; sodium carbonate; 1,3-bis[(2,6-diisopropyl)phenyl]imidazolinium chloride In N,N-dimethyl acetamide at 120℃; for 8h; | 87% |

| Conditions | Yield |
|---|---|
| With tris-(dibenzylideneacetone)dipalladium(0); tris(2-morpholinophenyl)phosphine; potassium carbonate In water; tert-butyl alcohol at 85℃; for 12h; Schlenk technique; Inert atmosphere; | 84% |
| With sodium carbonate In water; N,N-dimethyl-formamide at 120℃; for 15h; | 84% |

| Conditions | Yield |
|---|---|
| With ammonium hydroxide; copper(I) iodide; 1-ethylacetoacetate-3-methyl imidazolium hydroxide In acetonitrile at 120℃; for 12h; Inert atmosphere; | 81% |
| With ((+/-)-binap)Ni[P(OPh)3]2; ammonia; 2,2'-bis-(diphenylphosphino)-1,1'-binaphthyl; sodium t-butanolate In 1,4-dioxane at 120℃; for 18h; Sealed tube; | < 10 %Chromat. |

| Conditions | Yield |
|---|---|
| With sodium azide; copper; L-proline In water; dimethyl sulfoxide at 150℃; under 37503.8 Torr; for 0.583333h; Flow reactor; chemoselective reaction; | 79% |

-
A
-
14663-46-8
3-amino-3,4-dihydroquinazolin-4-one

-
B
-
1885-29-6
anthranilic acid nitrile

| Conditions | Yield |
|---|---|
| With hydrazine dihydrochloride; triethylamine In methanol; N,N-dimethyl-formamide at 80℃; for 36h; | A 77% B n/a |
-
-
615-43-0
2-iodophenylamine

-
-
68-12-2, 33513-42-7
N,N-dimethyl-formamide

-
-
1885-29-6
anthranilic acid nitrile

| Conditions | Yield |
|---|---|
| With ammonium cerium (IV) nitrate; copper(II) bis(trifluoromethanesulfonate); potassium carbonate at 130℃; for 24h; | 75% |

| Conditions | Yield |
|---|---|
| With 1,10-Phenanthroline; copper(II) nitrate trihydrate; potassium carbonate In dimethyl sulfoxide at 100℃; for 16h; | 73% |

| Conditions | Yield |
|---|---|
| With potassium fluoride; copper(l) iodide; 1,10-Phenanthroline; bis(di-tert-butyl(4-dimethylaminophenyl)phosphine)dichloropalladium(II); sodium t-butanolate In N,N-dimethyl-formamide at 130℃; for 24h; | 72% |
-
-
2400-12-6
benzo[c]isothiazol-3-ylamine

-
A
-
27259-73-0
2-(2'-aminophenyl)-4(3H)-quinazolinone

-
B
-
1885-29-6
anthranilic acid nitrile

| Conditions | Yield |
|---|---|
| With sodium hydrogensulfite In water for 60h; Heating; | A 66% B 15% |

| Conditions | Yield |
|---|---|
| With ammonium hydroxide; potassium phosphate; copper(II) ferrite at 100℃; for 12h; Sealed tube; chemoselective reaction; | 64% |
-
-
77287-34-4, 77287-35-5, 60100-09-6
formamide

-
-
615-36-1
2-bromoaniline

-
-
1885-29-6
anthranilic acid nitrile

| Conditions | Yield |
|---|---|
| With 1,3,5-trichloro-2,4,6-triazine; palladium on activated charcoal; potassium carbonate; triphenylphosphine at 25 - 130℃; for 12.1667h; | 64% |

| Conditions | Yield |
|---|---|
| In N,N-dimethyl-formamide at 60℃; for 48h; | A 63% B n/a |


| Conditions | Yield |
|---|---|
| With potassium fluoride; 1,10-Phenanthroline; copper diacetate; sodium t-butanolate In N,N-dimethyl-formamide at 120℃; for 18h; | 60% |

-
-
1885-29-6
anthranilic acid nitrile

| Conditions | Yield |
|---|---|
| With thionyl chloride In dichloromethane | 60% |
-
-
612-24-8
o-nitrobenzonitrile

-
A
-
28144-70-9
anthranilic acid amide

-
B
-
1885-29-6
anthranilic acid nitrile

| Conditions | Yield |
|---|---|
| With baker's yeast In ethanol; water at 32℃; | A 56% B 10% |

| Conditions | Yield |
|---|---|
| In methanol; N,N-dimethyl-formamide at 20℃; for 10h; | A 56% B n/a |


| Conditions | Yield |
|---|---|
| With 2-(dicyclohexylphosphino)-1-(2,6-diisopropylphenyl)-1H-imidazole; sodium carbonate; palladium dichloride In N,N-dimethyl acetamide at 140℃; for 16h; Inert atmosphere; | 55% |

-
-
1885-29-6
anthranilic acid nitrile

| Conditions | Yield |
|---|---|
| With sodium azide In acetonitrile for 20h; Heating; | 53% |
-
-
1476-23-9
allylisocyanate

-
-
1885-29-6
anthranilic acid nitrile

-
-
122372-27-4
2-(3-allylureido)benzonitrile

| Conditions | Yield |
|---|---|
| for 48h; Ambient temperature; | 100% |
| for 48h; Ambient temperature; | 100% |
| In dichloromethane for 3h; Ambient temperature; | 88% |
-
-
1885-29-6
anthranilic acid nitrile

-
-
2949-22-6
Glycine ethyl ester isocyanate

-
-
78754-91-3
2-(3-ethoxycarbonylmethylureido)benzonitrile

| Conditions | Yield |
|---|---|
| for 48h; Ambient temperature; | 100% |

| Conditions | Yield |
|---|---|
| With aluminium trichloride In 1,2-dichloro-ethane for 12h; Condensation; Heating; | 100% |
-
-
15568-85-1
5-methoxymethylene-2,2-dimethyl-1,3-dioxane-4,6-dione

-
-
1885-29-6
anthranilic acid nitrile

-
-
1020252-22-5
2-{[(2,2-dimethyl-4,6-dioxo-1,3-dioxan-5-ylidene)methyl]amino}benzonitrile

| Conditions | Yield |
|---|---|
| In acetonitrile at 20℃; for 0.25h; | 100% |
| In N,N-dimethyl-formamide for 2h; Heating; | |
| In acetonitrile for 0.0333333h; |
-
-
122-51-0
orthoformic acid triethyl ester

-
-
1885-29-6
anthranilic acid nitrile

-
-
104326-76-3
ethyl (2-cyanophenyl)imidoformate

| Conditions | Yield |
|---|---|
| for 0.75h; Reflux; | 100% |

| Conditions | Yield |
|---|---|
| In pyridine at 0℃; for 4h; | 100% |
-
-
1071146-39-8
1,1,1-trifluoro-4,4-dimethoxy-but-3-en-2-one

-
-
1885-29-6
anthranilic acid nitrile

| Conditions | Yield |
|---|---|
| In acetonitrile for 18h; Reflux; | 100% |
| In acetonitrile for 18h; Reflux; | 100% |
-
-
68-12-2, 33513-42-7
N,N-dimethyl-formamide

-
-
1885-29-6
anthranilic acid nitrile

-
-
86-96-4
1,2,3,4-tetrahydro-quinazoline-2,4-dione

| Conditions | Yield |
|---|---|
| With carbon dioxide In water at 150℃; under 37503.8 Torr; for 5h; | 99.2% |
| With zinc(II) chloride at 200℃; for 5h; sealed tube; | 90% |
-
-
100-52-7
benzaldehyde

-
-
1885-29-6
anthranilic acid nitrile

-
-
125743-39-7
(E)-2-<(Phenylmethylene)amino>benzonitrile

| Conditions | Yield |
|---|---|
| With 4 A molecular sieve In toluene at 23℃; for 10h; | 99% |
| In toluene Schlenk technique; Inert atmosphere; Molecular sieve; Reflux; | 89% |
| With toluene-4-sulfonic acid In toluene Reflux; Molecular sieve; Inert atmosphere; Schlenk technique; | 89% |
-
-
122-51-0
orthoformic acid triethyl ester

-
-
1885-29-6
anthranilic acid nitrile

-
-
58314-88-8
N-ethoxymethylene-2-aminobenzonitrile

| Conditions | Yield |
|---|---|
| In acetic acid; toluene at 100 - 110℃; for 3h; | 99% |
| Heating; |
-
-
13010-19-0
3-Chloropropyl isocyanate

-
-
1885-29-6
anthranilic acid nitrile

-
-
94507-24-1
2-<3-(3-Chloropropyl)ureido>benzonitrile

| Conditions | Yield |
|---|---|
| Heating; | 99% |
-
-
1885-29-6
anthranilic acid nitrile

-
-
69084-37-3
2,2'-(iminodimethylene)bisaniline

| Conditions | Yield |
|---|---|
| With hydrogen In toluene at 90℃; under 750.075 Torr; for 20h; chemoselective reaction; | 99% |
| With hydrogen; Rh/Al2O3 |
-
-
1885-29-6
anthranilic acid nitrile

-
-
28144-70-9
anthranilic acid amide

| Conditions | Yield |
|---|---|
| With water; potassium carbonate at 150℃; for 0.5h; Microwave irradiation; | 99% |
| With phosphate buffer at 30℃; for 1h; rhodococcus rhodocrous AJ270, pH 7.0; | 98% |
| With potassium phosphate buffer at 30℃; for 1h; Rhodococcus sp. AJ270 cells; | 97.7% |
-
-
112245-13-3
(S)-tert-leucinol

-
-
1885-29-6
anthranilic acid nitrile

-
-
194350-71-5
2-[(4S)-4-(tert-butyl)-4,5-dihydro-1,3-oxazol-2-yl]aniline

| Conditions | Yield |
|---|---|
| With zinc(II) chloride In chlorobenzene for 24h; Inert atmosphere; Reflux; | 99% |
| With zinc(II) chloride In dichloromethane at 140℃; for 24h; | 85% |
| With zinc(II) chloride In chlorobenzene at 130℃; for 36h; | 82% |
-
-
115-80-0
Triethyl orthopropionate

-
-
1885-29-6
anthranilic acid nitrile

-
-
132121-43-8
ethyl N-(2-cyanophenyl)propanimidate

| Conditions | Yield |
|---|---|
| With trifluoroacetic acid | 99% |
-
-
156-57-0
2-mercaptoethylamine hydrochloride

-
-
1885-29-6
anthranilic acid nitrile

-
-
1370477-92-1
2-(4,5-dihydrothiazol-2-yl)aniline

| Conditions | Yield |
|---|---|
| With sodium hydroxide In neat (no solvent) at 80℃; for 2h; | 99% |
| With sodium hydroxide at 80℃; | 96% |
| With tetrakis(μ2-α-methacrylate-O,O')-bis(H2O)-dicopper(II); sodium acetate at 80℃; for 3.5h; Neat (no solvent); | 92% |

-
-
1885-29-6
anthranilic acid nitrile

| Conditions | Yield |
|---|---|
| In dichloromethane at 20℃; Inert atmosphere; | 99% |
-
-
1885-29-6
anthranilic acid nitrile

-
-
148-87-8
N-ethyl-N-(2-cyanoethyl)aniline

-
-
52547-43-0
2-{4-[(2-Cyano-ethyl)-ethyl-amino]-phenylazo}-benzonitrile

| Conditions | Yield |
|---|---|
| Stage #1: anthranilic acid nitrile With sulfuric acid; nitrosyl sulphuric acid In water at 0 - 20℃; for 2h; Stage #2: N-ethyl-N-(2-cyanoethyl)aniline With sulfuric acid In water at 0℃; for 1h; | 99% |
-
-
56914-82-0
3,4-dichloro-isothiazole-5-carbonyl chloride

-
-
1885-29-6
anthranilic acid nitrile

-
-
224049-04-1
isotianil

| Conditions | Yield |
|---|---|
| In toluene at 80℃; for 2h; Solvent; Temperature; | 98.8% |
| With pyridine In tetrahydrofuran at 5 - 20℃; for 1.25h; Product distribution / selectivity; | 89% |
| With pyridine In tetrahydrofuran at 5 - 20℃; for 1.25h; Product distribution / selectivity; | 89% |
| With pyridine In tetrahydrofuran; water; ethyl acetate; Petroleum ether |
-
-
124-38-9
carbon dioxide

-
-
1885-29-6
anthranilic acid nitrile

-
-
17796-47-3
4-thioxo-1H,3H-quinazolin-2-one

| Conditions | Yield |
|---|---|
| Stage #1: anthranilic acid nitrile With sodium hydrogen sulfide In N,N-dimethyl-formamide for 0.5h; Autoclave; Heating; Stage #2: carbon dioxide In N,N-dimethyl-formamide at 100℃; under 30003 Torr; for 24h; Reagent/catalyst; Temperature; Pressure; Solvent; Autoclave; | 98.4% |
-
-
1885-29-6
anthranilic acid nitrile

-
-
68385-95-5
3,5-dibromoanthranilonitrile

| Conditions | Yield |
|---|---|
| With bromine In acetic acid at 20℃; | 98% |
| With bromine In acetic acid at 15 - 20℃; for 5h; | 98% |
| With bromine; acetic acid at 15 - 20℃; for 5h; Cooling with ice; | 98% |

| Conditions | Yield |
|---|---|
| With lithium aluminium tetrahydride In tetrahydrofuran | 98% |
| With [Ru(H)(BH4)(CO)(PPh3)(3-(di-tert-butylphosphino)-N-((1-methyl-1H-imidazol-2 yl)methyl)propylamine)]; hydrogen In isopropyl alcohol at 50℃; for 3h; Inert atmosphere; Autoclave; | 98% |
| With C28H29Cl2CoNP2; hydrogen; sodium triethylborohydride In 1,4-dioxane at 80℃; under 37503.8 Torr; for 6h; | 78% |

| Conditions | Yield |
|---|---|
| With zinc(II) chloride at 120℃; for 16h; | 98% |
| With silica gel; toluene-4-sulfonic acid for 0.133333h; microwave irradiation; | 95% |
| With sulfuric acid-modified polyethylene glycol 6000 at 130℃; for 0.133333h; Friedlaender synthesis; Microwave irradiation; Neat (no solvent); | 93% |
-
-
87-13-8
diethyl 2-ethoxymethylenemalonate

-
-
1885-29-6
anthranilic acid nitrile

-
-
127285-75-0
8-cyano-3-ethoxycarbonyl-1,4-dihydro-4-oxoquinolone

| Conditions | Yield |
|---|---|
| 1.) 130 degC, 1-2 h, 2.) C6H5OC6H5, 250 degC, 1h; | 98% |
| In diphenylether at 250℃; Gould-Jacobs condensation; | 60% |
-
-
1885-29-6
anthranilic acid nitrile

-
-
18216-38-1
5-(2-aminophenyl)tetrazole

| Conditions | Yield |
|---|---|
| With sodium azide In N,N-dimethyl-formamide at 120℃; for 4h; | 98% |
| Stage #1: anthranilic acid nitrile With sodium azide In N,N-dimethyl-formamide at 120℃; for 36h; Stage #2: With hydrogenchloride In water; ethyl acetate for 0.0833333h; | 95% |
| With iron(III) oxide; sodium azide In N,N-dimethyl-formamide at 120℃; for 36h; | 92% |
-
-
79-04-9
chloroacetyl chloride

-
-
1885-29-6
anthranilic acid nitrile

-
-
71993-21-0
[(2-cyanophenyl)aminocarbonylmethyl]chloride

| Conditions | Yield |
|---|---|
| With pyridine In N,N-dimethyl-formamide at 0 - 40℃; Microwave irradiation; | 98% |
| With triethylamine In dichloromethane at 0 - 20℃; for 48h; | 55% |
| With pyridine In N,N-dimethyl-formamide at 20℃; Acylation; | 54% |
Anthranilonitrile Chemical Properties
IUPAC Name: 2-Aminobenzonitrile
Molecular Structure: 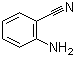
Molecular Formula: C7H6N2
Molecular Weight: 118.14
CAS NO: 1885-29-6
EINECS: 217-549-9
H bond acceptors: 2
H bond donors: 2
Freely Rotating Bonds: 1
Polar Surface Area: 27.03 Å2
Index of Refraction: 1.59
Molar Refractivity: 34.93 cm3
Molar Volume: 103.4 cm3
Surface Tension: 54.3 dyne/cm
Density: 1.14 g/cm3
Flash Point: 122.2 °C
Enthalpy of Vaporization: 50.55 kJ/mol
Boiling Point: 267.5 °C at 760 mmHg
Vapour Pressure: 0.00812 mmHg at 25°C
Melting Point: 45-48 °C(lit.)
Water Solubility: INSOLUBLE
Appearance: Yellow Solid
SMILES: N#Cc1ccccc1N
InChI: InChI=1/C7H6N2/c8-5-6-3-1-2-4-7(6)9/h1-4H,9H2
InChIKey: HLCPWBZNUKCSBN-UHFFFAOYAV
Std. InChI: InChI=1S/C7H6N2/c8-5-6-3-1-2-4-7(6)9/h1-4H,9H2
Std. InChIKey: HLCPWBZNUKCSBN-UHFFFAOYSA-N
Synonyms of Anthranilonitrile (CAS NO.1885-29-6): 2-Cyanoaniline ; 2-Aminobenzonitrile
Product Categories: FINE Chemical & INTERMEDIATES;Aromatic Nitriles;Nitriles;Chemical Amines;Amines;Aromatics
Anthranilonitrile Uses
Anthranilonitrile (CAS NO.1885-29-6) is used for induction of nitrilase activity in Arthrobacter. And, it is also used as radioprotective agent.
Anthranilonitrile Toxicity Data With Reference
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| mouse | LD50 | intraperitoneal | 1gm/kg (1000mg/kg) | Farmaco, Edizione Scientifica. Vol. 41, Pg. 41, 1986. | |
| mouse | LD50 | intravenous | 180mg/kg (180mg/kg) | U.S. Army Armament Research & Development Command, Chemical Systems Laboratory, NIOSH Exchange Chemicals. Vol. NX00381, |
Anthranilonitrile Consensus Reports
Reported in EPA TSCA Inventory. Cyanide and its compounds are on the Community Right-To-Know List.
Anthranilonitrile Safety Profile
Safety Information about Anthranilonitrile (CAS NO.1885-29-6):
Hazard Codes:  XnXi ,
XnXi , T
T
Risk Statements: 20/21/22-36/37/38-43
R20/21/22: Harmful by inhalation, in contact with skin and if swallowed.
R36/37/38: Irritating to eyes, respiratory system and skin.
R43: May cause sensitization by skin contact.
Safety Statements: 26-36/37
S26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice.
S36/37: Wear suitable protective clothing and gloves.
RIDADR: 3276
WGK Germany: 2
RTECS: CB4575000
TSCA: T
HazardClass: 6.1
PackingGroup: III
HS Code: 29269095
Poison by intravenous route. See also NITRILES. When heated to decomposition it emits toxic fumes of NOx and CN−.
Anthranilonitrile Specification
Chemical Stability: Not available
Conditions to Avoid: Incompatible materials.
Incompatibilities with Other Materials Strong oxidizing agents, strong reducing agents, strong acids, strong bases.
Hazardous Decomposition Products Nitrogen oxides, carbon monoxide, carbon dioxide.
Hazardous Polymerization Has not been reported.
Related Products
- Anthranilonitrile
- 188530-20-3
- 18853-32-2
- 188534-09-0
- 1885-35-4
- 1885-38-7
- 18854-48-3
- 188548-57-4
- 18855-04-4
- 188554-13-4
- 18856-63-8
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View