-
Name
Dimethyl methylphosphonate
- EINECS 212-052-3
- CAS No. 756-79-6
- Article Data127
- CAS DataBase
- Density 1.079 g/cm3
- Solubility >=10 g/100 mL at 21 ºC
- Melting Point <50°
- Formula C3H9O3P
- Boiling Point 181 °C at 760 mmHg
- Molecular Weight 124.076
- Flash Point 68.9 °C
- Transport Information UN No 199
- Appearance colourless liquid
- Safety 53-26-45
- Risk Codes 36-46
-
Molecular Structure
-
Hazard Symbols
 T
T
- Synonyms Phosphonicacid, methyl-, dimethyl ester (6CI,8CI,9CI);DMMP;Dimethoxymethyl phosphineoxide;Dimethyl methanephosphonate;Dimethyl methylphosphonate;Fran TF 2000;Fyrol DMMP;Metaran;Methanephosphonic acid dimethyl ester;Methylphosphonicacid dimethyl ester;NSC 62240;O,O-Dimethyl methylphosphonate;Reoflam DMMP;
- PSA 45.34000
- LogP 1.10210
Synthetic route
-
-
121-45-9
phosphorous acid trimethyl ester

-
-
74-88-4
methyl iodide

-
-
756-79-6
dimethyl methane phosphonate

| Conditions | Yield |
|---|---|
| In neat (no solvent) at 100℃; for 0.138833h; Solvent; Arbuzov Reaction; Flow reactor; | 99% |
| for 1h; Irradiation; | 95% |
| for 0.0833333h; Michaelis-Arbuzov reaction; microwave irradiation; | 95% |

| Conditions | Yield |
|---|---|
| With iodine for 24h; Heating; | 99% |
| With trimethylsilyl trifluoromethanesulfonate In chloroform at 60℃; for 18h; Michaelis-Arbuzov rearrangement; | 98% |
| With trimethylsilyl iodide at 50℃; for 6h; Michaelis-Arbuzov rearrangement; | 95% |
-
-
77-99-6
1,1,1-tri(hydroxymethyl)propane

-
-
121-45-9
phosphorous acid trimethyl ester

-
-
756-79-6
dimethyl methane phosphonate

| Conditions | Yield |
|---|---|
| Stage #1: 1,1,1-tri(hydroxymethyl)propane; phosphorous acid trimethyl ester at 80 - 120℃; for 4h; Stage #2: With methyl p-toluene sulfonate at 170 - 180℃; for 2h; Reagent/catalyst; Temperature; | 96% |
-
-
676-97-1
methylphosphonic acid dichloroanhydride

-
-
67-56-1
methanol

-
-
756-79-6
dimethyl methane phosphonate

| Conditions | Yield |
|---|---|
| With aluminum oxide at 20℃; | 95% |
| With triethylamine | 80% |
-
-
100-39-0
benzyl bromide

-
-
121-45-9
phosphorous acid trimethyl ester

-
A
-
773-47-7
dimethyl benzylphosphonate

-
B
-
756-79-6
dimethyl methane phosphonate

| Conditions | Yield |
|---|---|
| at 120℃; for 9h; | A 95% B 144 g |

-
-
121-45-9
phosphorous acid trimethyl ester

-
-
3433-80-5
1-Bromo-2-bromomethyl-benzene

-
A
-
103474-07-3
dimethyl (2-bromobenzyl)phosphonate

-
B
-
756-79-6
dimethyl methane phosphonate

| Conditions | Yield |
|---|---|
| A 93% B n/a |


| Conditions | Yield |
|---|---|
| at 100 - 140℃; for 0.2h; Arbuzov reaction; | A n/a B 90% |


| Conditions | Yield |
|---|---|
| With tetrachlorosilane at 0℃; | 89% |
| With p-TsOH-Celite at 20℃; | 87% |
| Stage #1: methylphosphonic acid With 1H-imidazole; iodine In dichloromethane at 45 - 50℃; for 0.5h; Gareg-Samuelsson reaction; Stage #2: methanol In dichloromethane at 45 - 50℃; for 0.666667h; Gareg-Samuelsson reaction; | 85% |
-
-
15145-29-6
1,2,2,2-tetrachloro-1-isocyanato-ethane

-
-
121-45-9
phosphorous acid trimethyl ester

-
A
-
85978-66-1
2,2-dichloro-1-(dimethoxyphosphinyl)vinyl isocyanate

-
B
-
85978-65-0
2,2,2-trichloro-1-(dimethoxyphosphinyl)ethyl isocyanate

-
C
-
756-79-6
dimethyl methane phosphonate

| Conditions | Yield |
|---|---|
| at 100℃; for 1h; | A 7% B n/a C 81% |

-
-
121-45-9
phosphorous acid trimethyl ester

-
-
5437-45-6
Benzyl bromoacetate

-
A
-
57443-18-2
benzyl 2-(dimethoxyphosphoryl)acetate

-
B
-
756-79-6
dimethyl methane phosphonate

| Conditions | Yield |
|---|---|
| at 85℃; for 60h; | A 81% B n/a |

-
-
591-50-4
iodobenzene

-
-
868-85-9
Dimethyl phosphite

-
A
-
2240-41-7
dimethyl phenylphosphonate

-
B
-
756-79-6
dimethyl methane phosphonate

| Conditions | Yield |
|---|---|
| With N-benzyl-N,N,N-triethylammonium chloride; potassium carbonate; palladium diacetate; triphenylphosphine In acetonitrile at 70℃; for 4h; Title compound not separated from byproducts; | A 80% B n/a |


| Conditions | Yield |
|---|---|
| Stage #1: Dimethyl phosphite With sodium hydroxide for 0.25h; Michaelis-Becker reaction; Ionic liquid; Stage #2: methyl iodide for 12h; Michaelis-Becker reaction; Ionic liquid; | 80% |
| Stage #1: Dimethyl phosphite With sodium In dichloromethane for 40h; Heating; Stage #2: methyl iodide In dichloromethane for 10h; Michaelis-Becker reaction; Heating; |
-
-
31142-23-1
trimethyl phosphonoformate

-
A
-
512-56-1
trimethyl phosphite

-
B
-
756-79-6
dimethyl methane phosphonate

| Conditions | Yield |
|---|---|
| at 295℃; for 6h; | A 18% B 75% |

| Conditions | Yield |
|---|---|
| With tetraethylammonium bromide; cyclohexene at 20℃; electrolysis, Pt electrodes; | 75% |

| Conditions | Yield |
|---|---|
| With polymer supported sulfonated magnetic resin In toluene at 20 - 70℃; for 0.75h; | 74% |
-
-
121-45-9
phosphorous acid trimethyl ester

-
A
-
512-56-1
trimethyl phosphite

-
B
-
152-18-1
O,O,O-trimethylthiophosphate

-
C
-
756-79-6
dimethyl methane phosphonate

| Conditions | Yield |
|---|---|
| With sulfur trioxide In dichloromethane at -95℃; | A 25% B 4% C 71% |


-
-
121-45-9
phosphorous acid trimethyl ester

-
A
-
749887-15-8
1-ethoxyethyl O,O-dimethylphosphorylethanoate

-
B
-
756-79-6
dimethyl methane phosphonate

| Conditions | Yield |
|---|---|
| at 50 - 60℃; for 2.5h; Arbuzov reaction; | A 71% B 25.9% |

-
-
67-56-1
methanol

-
A
-
512-56-1
trimethyl phosphite

-
B
-
868-85-9
Dimethyl phosphite

-
C
-
756-79-6
dimethyl methane phosphonate

| Conditions | Yield |
|---|---|
| With phosphorous; tetraethylammonium iodide In acetonitrile at 18℃; electrolysis; | A 70% B 11% C 3% |
| With phosphorous; tetraethylammonium iodide In acetonitrile at 18℃; Mechanism; Product distribution; electrolysis; var. nucleophiles; var. temperatures; | A 70% B 11% C 3% |
| With phosphorous In acetonitrile at 18℃; Electrolysis; | A 70% B 11% C 3% |


| Conditions | Yield |
|---|---|
| With potassium carbonate at 25℃; for 4h; | 70% |
| With potassium carbonate at 25℃; for 4h; other phosphonates; | 70% |

-
-
121-45-9
phosphorous acid trimethyl ester

-
A
-
749887-15-8
1-ethoxyethyl O,O-dimethylphosphorylethanoate

-
B
-
756-79-6
dimethyl methane phosphonate

| Conditions | Yield |
|---|---|
| at 40 - 50℃; for 2h; Arbuzov reaction; | A 67.2% B 27.4% |

-
-
52956-34-0
dimethyl (1-methylethyl) phosphite

-
-
74-88-4
methyl iodide

-
A
-
690-64-2
isopropyl methyl methylphosphonate

-
C
-
756-79-6
dimethyl methane phosphonate

| Conditions | Yield |
|---|---|
| for 2h; Arbusov rearrangement; Heating; | A 66% B 7 % Chromat. C 3 % Chromat. |


| Conditions | Yield |
|---|---|
| With tetraethylammonium bromide In acetonitrile at 20℃; on a platinum electrode; | 65% |
-
-
121-45-9
phosphorous acid trimethyl ester

-
A
-
512-56-1
trimethyl phosphite

-
B
-
152-18-1
O,O,O-trimethylthiophosphate

-
C
-
77-78-1
dimethyl sulfate

-
D
-
756-79-6
dimethyl methane phosphonate

| Conditions | Yield |
|---|---|
| With sulfur trioxide In dichloromethane at -78℃; Product distribution; other trialkyl phosphite,trialkyl phosphine, trialkyl arsines, trialkoxyarsines, var. molar ratio and temperatures; | A 22% B 4% C 55% D 19% |
| With sulfur trioxide In dichloromethane at -78℃; | A 48% B 16% C 3% D 9% |


| Conditions | Yield |
|---|---|
| With phosphorous; tetraethylammonium iodide In acetonitrile at 50℃; electrolysis; | A 51% B 28% |
| With phosphorous In acetonitrile at 50℃; Electrolysis; | A 51% B 28% |
-
-
119-61-9
benzophenone

-
-
13433-42-6
dimethyl α-trimethylsilylmethylphosphonate

-
A
-
530-48-3
1,1-Diphenylethylene

-
B
-
71265-07-1
dimethyl (2,2-diphenylvinyl)phosphonate

-
C
-
756-79-6
dimethyl methane phosphonate

| Conditions | Yield |
|---|---|
| With cesium fluoride | A 20% B 30% C 50% |
| With cesium fluoride In tetrahydrofuran for 24h; Heating; | A 20% B 30% C 50% |

-
-
106-96-7
propargyl bromide

-
-
121-45-9
phosphorous acid trimethyl ester

-
A
-
4885-44-3
Dimethyl propargylphosphonate

-
B
-
756-79-6
dimethyl methane phosphonate

| Conditions | Yield |
|---|---|
| for 5h; Heating; | A 45% B n/a |

-
-
121-45-9
phosphorous acid trimethyl ester

-
A
-
4885-44-3
Dimethyl propargylphosphonate

-
B
-
756-79-6
dimethyl methane phosphonate

| Conditions | Yield |
|---|---|
| With propargyl bromide for 5h; Heating; | A 45% B n/a |
-
-
333-27-7
methyl trifluoromethanesulfonate

-
-
121-45-9
phosphorous acid trimethyl ester

-
A
-
64294-66-2
methyltrimethoxyphosphonium triflate

-
B
-
756-79-6
dimethyl methane phosphonate

| Conditions | Yield |
|---|---|
| In chloroform-d1 at 0 - 20℃; for 1h; Time; Inert atmosphere; Schlenk technique; | A 45% B 27 %Spectr. |


| Conditions | Yield |
|---|---|
| Stage #1: dimethyl methane phosphonate With n-butyllithium In tetrahydrofuran; hexane at -78℃; for 0.0833333h; Metallation; Stage #2: benzaldehyde In tetrahydrofuran; hexane at -78℃; for 0.0833333h; Addition; | 100% |
| With n-butyllithium 1.) THF, hexane, -78 deg C, 15 min, 2.) THF, hexane, -78 -> -20 deg C; Yield given. Multistep reaction; | |
| Stage #1: dimethyl methane phosphonate With n-butyllithium In tetrahydrofuran at -70℃; Inert atmosphere; Stage #2: benzaldehyde In tetrahydrofuran Inert atmosphere; |
-
-
231291-83-1
(S)-3-(4-Methoxy-benzyl)-2,2-dimethyl-oxazolidine-4-carboxylic acid methyl ester

-
-
756-79-6
dimethyl methane phosphonate

-
-
231291-85-3
{2-[(S)-3-(4-Methoxy-benzyl)-2,2-dimethyl-oxazolidin-4-yl]-2-oxo-ethyl}-phosphonic acid dimethyl ester

| Conditions | Yield |
|---|---|
| With n-butyllithium In tetrahydrofuran at -78℃; | 100% |
-
-
35909-92-3
N-carbobenzoxy-L-phenylalanine methyl ester

-
-
756-79-6
dimethyl methane phosphonate

-
-
347851-53-0
dimethyl [(3S)-4-phenyl-3-[(benzyloxycarbonyl)amino]-2-oxobutyl]phosphonate

| Conditions | Yield |
|---|---|
| Stage #1: dimethyl methane phosphonate With n-butyllithium In tetrahydrofuran; hexane at -78℃; for 1h; Stage #2: N-carbobenzoxy-L-phenylalanine methyl ester In tetrahydrofuran; hexane Further stages.; | 100% |
| Stage #1: dimethyl methane phosphonate With n-butyllithium In tetrahydrofuran; hexanes at -78℃; under 760.051 Torr; for 0.333333h; Stage #2: N-carbobenzoxy-L-phenylalanine methyl ester In tetrahydrofuran; hexanes at -78 - 20℃; under 760.051 Torr; for 1.25h; Stage #3: With water; acetic acid In tetrahydrofuran; hexanes at 0℃; under 760.051 Torr; | 100% |
-
-
150765-78-9
2-methyl-3-oxocyclohex-1-en-1-yl trifluoromethanesulfonate

-
-
756-79-6
dimethyl methane phosphonate

-
-
475467-02-8
1-(dimethylphosphonato)-2-oxo-6-octyne

| Conditions | Yield |
|---|---|
| Stage #1: dimethyl methane phosphonate With n-butyllithium In tetrahydrofuran; hexane at -78℃; for 0.333333h; Stage #2: 2-methyl-3-oxocyclohex-1-en-1-yl trifluoromethanesulfonate In tetrahydrofuran; hexane at -78 - 60℃; for 1.33333h; Inert atmosphere; | 100% |
| Stage #1: dimethyl methane phosphonate With n-butyllithium In tetrahydrofuran; hexane at -78℃; for 0.333333h; Inert atmosphere; Stage #2: 2-methyl-3-oxocyclohex-1-en-1-yl trifluoromethanesulfonate In tetrahydrofuran; hexane at -78 - 60℃; Inert atmosphere; Stage #3: With water; ammonium chloride In tetrahydrofuran; diethyl ether; hexane Inert atmosphere; | 97% |
| Stage #1: dimethyl methane phosphonate With lithium hexamethyldisilazane In tetrahydrofuran at -78℃; for 0.5h; Stage #2: 2-methyl-3-oxocyclohex-1-en-1-yl trifluoromethanesulfonate In tetrahydrofuran at -78 - 60℃; for 1.33333h; Claisen condensation; | 21% |
| Stage #1: dimethyl methane phosphonate With lithium hexamethyldisilazane In tetrahydrofuran at -78℃; for 0.5h; Stage #2: 2-methyl-3-oxocyclohex-1-en-1-yl trifluoromethanesulfonate In tetrahydrofuran at -78 - 60℃; Claisen condensation; | 21% |
-
-
2930-05-4
Benzyloxymethyl-oxiran

-
-
756-79-6
dimethyl methane phosphonate

-
-
476310-16-4
dimethyl 4-(benzyloxy)-3-hydroxybutanephosphonate

| Conditions | Yield |
|---|---|
| Stage #1: dimethyl methane phosphonate With n-butyllithium In tetrahydrofuran; hexane at -78℃; for 0.25h; Stage #2: Benzyloxymethyl-oxiran With boron trifluoride diethyl etherate In tetrahydrofuran; hexane at -70℃; for 2h; | 100% |
| Stage #1: dimethyl methane phosphonate With n-butyllithium In tetrahydrofuran at -78℃; for 0.25h; Stage #2: Benzyloxymethyl-oxiran With boron trifluoride diethyl etherate In tetrahydrofuran at -70℃; for 2h; | 100% |
-
-
29542-59-4
methyl 3-(adamantan-1-yl)propanoate

-
-
756-79-6
dimethyl methane phosphonate

-
-
53273-30-6
dimethyl 2-oxo-4-(1-adamantyl)butylphosphonate

| Conditions | Yield |
|---|---|
| With n-butyllithium In tetrahydrofuran; hexane; water | 100% |
-
-
154698-92-7
methyl 2-((tert-butyl(diphenyl)silyl)oxy)acetate

-
-
756-79-6
dimethyl methane phosphonate

-
-
1075244-38-0
dimethyl 3-(tert-butyldiphenylsilyl)oxy-2-oxopropylphosphonate

| Conditions | Yield |
|---|---|
| Stage #1: dimethyl methane phosphonate With n-butyllithium In tetrahydrofuran; hexane at -78℃; for 0.5h; Inert atmosphere; Stage #2: methyl 2-((tert-butyl(diphenyl)silyl)oxy)acetate In tetrahydrofuran; hexane for 0.5h; Inert atmosphere; Stage #3: With ammonium chloride In tetrahydrofuran; hexane; water | 100% |
-
-
73323-65-6
1-(tert-butyl) 2-methyl (R)-pyrrolidine-1,2-dicarboxylate

-
-
756-79-6
dimethyl methane phosphonate

-
-
1140496-59-8
(R)-1-tert-butyl 2-(2-(dimethoxyphosphoryl)acetyl)pyrrolidine-1-carboxylate

| Conditions | Yield |
|---|---|
| Stage #1: dimethyl methane phosphonate With n-butyllithium In tetrahydrofuran; hexanes at -78℃; for 0.5h; Stage #2: 1-(tert-butyl) 2-methyl (R)-pyrrolidine-1,2-dicarboxylate In tetrahydrofuran; hexanes at -78℃; for 3h; Stage #3: With water; ammonium chloride In tetrahydrofuran; hexanes | 100% |
-
-
116393-72-7
dimethyl (2S)-N-(triphenylmethyl)aspartate

-
-
756-79-6
dimethyl methane phosphonate

-
-
1197335-24-2
methyl (2S)-2-(tritylamino)-4-oxo-5-(dimethoxyphosphoryl)pentanoate

| Conditions | Yield |
|---|---|
| With n-butyllithium In tetrahydrofuran at -78℃; Inert atmosphere; regioselective reaction; | 100% |
| With n-butyllithium In tetrahydrofuran at -78℃; for 2h; Inert atmosphere; | 92% |
| Stage #1: dimethyl methane phosphonate With n-butyllithium In tetrahydrofuran; hexane at -78℃; for 1h; Inert atmosphere; Stage #2: dimethyl (2S)-N-(triphenylmethyl)aspartate In tetrahydrofuran at -78℃; for 2h; Inert atmosphere; | 92% |
-
-
1309567-74-5
ethyl 4-methoxy-5-(4-((4-methoxybenzyl)oxy)phenyl)-2-oxo-1,2-dihydropyridine-3-carboxylate

-
-
756-79-6
dimethyl methane phosphonate

-
-
1309567-75-6
dimethyl (2-(4-methoxy-5-(4-((4-methoxybenzyl)oxy)phenyl)-2-oxo-1,2-dihydropyridin-3-yl)-2-oxoethyl)phosphonate

| Conditions | Yield |
|---|---|
| Stage #1: dimethyl methane phosphonate With n-butyllithium In tetrahydrofuran; hexane at -78 - -20℃; for 0.25h; Inert atmosphere; Stage #2: ethyl 4-methoxy-5-(4-((4-methoxybenzyl)oxy)phenyl)-2-oxo-1,2-dihydropyridine-3-carboxylate In tetrahydrofuran; hexane at -78 - 0℃; for 0.166667h; Inert atmosphere; | 100% |

| Conditions | Yield |
|---|---|
| Stage #1: dimethyl methane phosphonate With n-butyllithium In tetrahydrofuran; hexane at -78℃; for 0.5h; Stage #2: C17H28O4 In tetrahydrofuran; hexane at -78℃; for 2h; Stage #3: With ammonium chloride In tetrahydrofuran; hexane; water at -78℃; | 100% |
-
-
29681-38-7
methyl 5-methylpyridine-2-carboxylate

-
-
756-79-6
dimethyl methane phosphonate

-
-
1369491-73-5
C10H14NO4P

| Conditions | Yield |
|---|---|
| Stage #1: dimethyl methane phosphonate With n-butyllithium In tetrahydrofuran; hexane at -78℃; for 0.333333h; Inert atmosphere; Stage #2: methyl 5-methylpyridine-2-carboxylate In tetrahydrofuran; hexane at -78℃; for 0.333333h; Inert atmosphere; Stage #3: With ammonium chloride In tetrahydrofuran; hexane; water | 100% |
-
-
139978-82-8
methyl 2,3-O-isopropylidene-α-D-mannopyranoside-4,6-cyclic sulfate

-
-
756-79-6
dimethyl methane phosphonate

-
-
454183-61-0
[2-((2R,3S,4S,5S,6S)-3,4,5-Trihydroxy-6-methoxy-tetrahydro-pyran-2-yl)-ethyl]-phosphonic acid dimethyl ester

| Conditions | Yield |
|---|---|
| Stage #1: methyl-2,3-O-isopropylidene-4,6-cyclic sulfate-α-D-mannopyranoside; dimethyl methane phosphonate With 1,1-Diphenylethylene; N,N,N,N,N,N-hexamethylphosphoric triamide; n-butyllithium In tetrahydrofuran at -78℃; Stage #2: In tetrahydrofuran; methanol regioselective reaction; | 100% |
-
-
756-79-6
dimethyl methane phosphonate

-
-
1569075-94-0
dimethyl (5-iodo-2-oxohex-5-en-1-yl)phosphonate

| Conditions | Yield |
|---|---|
| Stage #1: dimethyl methane phosphonate With n-butyllithium In tetrahydrofuran; hexane at -78℃; for 0.333333h; Inert atmosphere; Stage #2: ethyl 4-iodopent-4-enoate In tetrahydrofuran; hexane at -78℃; for 1h; Inert atmosphere; | 100% |

-
-
756-79-6
dimethyl methane phosphonate

| Conditions | Yield |
|---|---|
| Stage #1: dimethyl methane phosphonate With n-butyllithium In tetrahydrofuran; hexane at -78℃; for 0.5h; Stage #2: methyl 2-(((tert-butoxycarbonyl)oxy)methyl)benzofuran-5-carboxylate In tetrahydrofuran; hexane at -78℃; for 2h; | 100% |
-
-
756-79-6
dimethyl methane phosphonate

-
A
-
676-97-1
methylphosphonic acid dichloroanhydride

-
B
-
74-87-3
methylene chloride

| Conditions | Yield |
|---|---|
| With thionyl chloride; N-Formylpiperidine for 19h; Heating; various conditions and cactalasts investigated; | A 99.2% B n/a |
-
-
117625-50-0
3-[2-(tert-Butyl-dimethyl-silanyloxy)-4-(trityl-amino)-phenyl]-propionic acid methyl ester

-
-
756-79-6
dimethyl methane phosphonate

-
-
110828-45-0
{4-[2-(tert-Butyl-dimethyl-silanyloxy)-4-(trityl-amino)-phenyl]-2-oxo-butyl}-phosphonic acid dimethyl ester

| Conditions | Yield |
|---|---|
| With n-butyllithium | 99% |
| With n-butyllithium Yield given; |
-
-
260970-23-8
methyl (4Z)-6,6,9-trimethyl-10-(tetrahydropyranyloxy)dec-6-en-2-ynoate

-
-
756-79-6
dimethyl methane phosphonate

-
-
260970-36-3
(7Z)-1-dimethoxyphosphiryl-7,10,10-trimethyl-11-(tetrahydropyranyloxy)undec-7-en-3-yn-2-one

| Conditions | Yield |
|---|---|
| With n-butyllithium In tetrahydrofuran; hexane at -78 - 20℃; Substitution; | 99% |
-
-
112-54-9
Dodecanal

-
-
756-79-6
dimethyl methane phosphonate

-
-
88708-59-2
dimethyl 2-hydroxytridecylphosphonate

| Conditions | Yield |
|---|---|
| Stage #1: dimethyl methane phosphonate With n-butyllithium In tetrahydrofuran; hexane at -78℃; for 0.0833333h; Metallation; Stage #2: Dodecanal In tetrahydrofuran; hexane at -78℃; for 0.0833333h; Addition; | 99% |
-
-
935548-14-4
(S)-methyl 2-palmitamido-3-(4-((4-(2,2,2-trifluoroethoxy)pyridin-2-yl)methoxy)phenyl)propanoate

-
-
756-79-6
dimethyl methane phosphonate

-
-
935548-32-6
(S)-dimethyl 2-oxo-3-palmitamido-4-(4-((4-(2,2,2-trifluoroethoxy)pyridin-2-yl)methoxy)phenyl)butylphosphonate

| Conditions | Yield |
|---|---|
| Stage #1: dimethyl methane phosphonate With n-butyllithium In tetrahydrofuran at -78℃; for 0.25h; Inert atmosphere; Stage #2: (S)-methyl 2-palmitamido-3-(4-((4-(2,2,2-trifluoroethoxy)pyridin-2-yl)methoxy)phenyl)propanoate In tetrahydrofuran at -78℃; for 0.333333h; Inert atmosphere; | 99% |
| Stage #1: dimethyl methane phosphonate With n-butyllithium Stage #2: (S)-methyl 2-palmitamido-3-(4-((4-(2,2,2-trifluoroethoxy)pyridin-2-yl)methoxy)phenyl)propanoate at -78℃; for 3h; |

| Conditions | Yield |
|---|---|
| With n-butyllithium In tetrahydrofuran at -78℃; | 99% |

-
-
756-79-6
dimethyl methane phosphonate

| Conditions | Yield |
|---|---|
| With n-butyllithium In tetrahydrofuran; hexane at -60 - 20℃; for 2h; | 99% |
-
-
141-75-3
butyryl chloride

-
-
756-79-6
dimethyl methane phosphonate

-
-
65921-74-6
dimethyl (2-oxopentyl)phosphonate

| Conditions | Yield |
|---|---|
| 99% |
-
-
13520-92-8
ziconium(IV) oxychloride octahydrate

-
-
756-79-6
dimethyl methane phosphonate

| Conditions | Yield |
|---|---|
| With aq. HCl In water reflux (24 h or 112 h); aq. HBr or H2O alone may also be used; filtration, washing (H2O; acetone), drying (40°C, 18 h, then 200°C, 24 h); elem. anal.; | 99% |

| Conditions | Yield |
|---|---|
| With lithium hexamethyldisilazane In tetrahydrofuran Barbier reaction; Cooling with ice; Inert atmosphere; | 99% |

| Conditions | Yield |
|---|---|
| With lithium hexamethyldisilazane In tetrahydrofuran Barbier reaction; Cooling with ice; Inert atmosphere; | 99% |
-
-
7098-07-9
N-Ethylimidazole

-
-
756-79-6
dimethyl methane phosphonate

-
-
1059603-87-0
[EMIM] methyl methylphosphonate

| Conditions | Yield |
|---|---|
| at 100℃; for 24h; Neat (no solvent); | 99% |
-
-
1439462-50-6
methyl-2-[N-(2,4,6-trimethylbenzenesulfonyl)amino]-3-phenyl-propionate

-
-
756-79-6
dimethyl methane phosphonate

-
-
1439462-62-0
dimethyl-{3-[N-(2,4,6-trimethylbenzenesulfonyl)amino]-2-oxo-4-phenyl-butyl}phosphonate

| Conditions | Yield |
|---|---|
| Stage #1: dimethyl methane phosphonate With n-butyllithium In tetrahydrofuran; hexane at -78℃; for 0.5h; Inert atmosphere; Stage #2: methyl-2-[N-(2,4,6-trimethylbenzenesulfonyl)amino]-3-phenyl-propionate In tetrahydrofuran; hexane at -78 - 0℃; for 5h; Inert atmosphere; | 99% |
DIMETHYL METHYLPHOSPHONATE Chemical Properties
Dimethyl methylphosphonate(756-79-6)'s molecular formula is C3H9O3P and its formula weight is 124.08.
The density of dimethyl methylphosphonate(756-79-6) is 1.145 g/mL at 25 °C(lit.) and it has a boiling point of 181 °C(lit.). The refractive index is about 1.413(lit.). Its flash point is 156 °F. The water solubility should be equal to or greater than 10 g/100 mL at 21 oC.
Dimethyl methylphosphonate(756-79-6) has the property of being dissolved in water.
The molecular structure of dimethyl methylphosphonate(756-79-6):
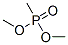
DIMETHYL METHYLPHOSPHONATE Uses
The primary commercial use of dimethyl methylphosphonate(756-79-6) is as a flame retardant. Other commercial uses are a preignition additive for gasoline, anti-foaming agent, plasticizer, stabilizer, textile conditioner, antistatic agent, and an additive for solvents and low-temperature hydraulic fluids. It can be used as a catalyst and a reagent in organic synthesis, as it can generate a highly reactive ylide. The yearly production in the United States varies between 91,000 and 910,000 kilograms.
DIMETHYL METHYLPHOSPHONATE Toxicity Data With Reference
| 1. | dlt-mus-orl 65 g/kg/13W-C | MUREAV Mutation Research. 138 (1984),213. | ||
| 2. | cyt-ham:ovr 250 mg/L | NTIS** National Technical Information Service. (Springfield, VA 22161) (Formerly U.S. Clearinghouse for Scientific and Technical Information) AD-A124-785 . | ||
| 3. | orl-mus TDLo:33 g/kg (female 7-14D post):REP | NTIS** National Technical Information Service. (Springfield, VA 22161) (Formerly U.S. Clearinghouse for Scientific and Technical Information) PB85-220143 . | ||
| 4. | orl-rat LD50:8210 mg/kg | TSCAT* Office of Toxic Substances Report. (U.S. Environmental Protection Agency, Office of Toxics Substances, 401M Street SW, Washington, DC 20460) FYI-OTS-0483-0242 . | ||
| 5. | ivn-rat LD50:1050 mg/kg | TSCAT* Office of Toxic Substances Report. (U.S. Environmental Protection Agency, Office of Toxics Substances, 401M Street SW, Washington, DC 20460) FYI-OTS-0483-0242 . | ||
| 6. | orl-mus LD50:>6810 mg/kg | NTPTR* National Toxicology Program Technical Report Series. (Research Triangle Park, NC 27709) No. NTP-TR-323 ,1987. | ||
| 7. | ivn-mus LD50:912 mg/kg | TSCAT* Office of Toxic Substances Report. (U.S. Environmental Protection Agency, Office of Toxics Substances, 401M Street SW, Washington, DC 20460) FYI-OTS-0483-0242 . |
DIMETHYL METHYLPHOSPHONATE Consensus Reports
DIMETHYL METHYLPHOSPHONATE Safety Profile
DIMETHYL METHYLPHOSPHONATE Specification
Incompatible with strong oxidizing agents and strong bases Reacts with organic halides at 302-392 F Reacts with enol lactones.
Storage
Keep in a cool, dry, dark location in a tightly sealed container or cylinder. Keep away from incompatible materials, ignition sources and untrained individuals. Secure and label area. Protect containers/cylinders from physical damage.
Handling
All chemicals should be considered hazardous. Avoid direct physical contact. Use appropriate, approved safety equipment. Untrained individuals should not handle this chemical or its container. Handling should occur in a chemical fume hood.
Related Products
- Dimethyl ( )-2,3-O-Isopropylidene-D-tartrate
- Dimethyl ((1-methyl-5-nitro-1H-imidazol-2-yl)methylene)propanedioate
- Dimethyl (2-oxo-3,3-difluoroheptyl)phosphonate
- Dimethyl (2-oxo-4-phenylbutyl)phosphonate
- Dimethyl (2-oxoheptyl)phosphonate
- Dimethyl (2S, 2'S)-1, 1'-((2S, 2'S)-2, 2'-(4, 4'-(biphenyl-4, 4'-diyl)bis(1H-imidazole-4, 2-diyl))bis(pyrrolidine-2, 1-diyl))bis(3-methyl-1-oxobutane-2, 1-diyl)dicarbamate
- Dimethyl (3-phenoxy-2-oxopropyl)phosphonate
- Dimethyl (R)-(+)-methylsuccinate
- Dimethyl (S)-(-)-methylsuccinate
- Dimethyl (S)-3-hydroxy-L-aspartate
- 756-80-9
- 75680-92-1
- 75681-13-9
- 75-68-3
- 756859-05-9
- 7568-92-5
- 7568-93-6
- 75690-75-4
- 75-69-4
- 75694-39-2
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View