-
Name
Diethyl ketomalonate
- EINECS 210-176-2
- CAS No. 609-09-6
- Article Data50
- CAS DataBase
- Density 1.175 g/cm3
- Solubility Fully miscible with water.
- Melting Point -70 °C
- Formula C7H10O5
- Boiling Point 209 °C at 760 mmHg
- Molecular Weight 174.153
- Flash Point 90.2 °C
- Transport Information
- Appearance yellow to yellow-green liquid.
- Safety 24/25
- Risk Codes
-
Molecular Structure
-
Hazard Symbols
 Xi
Xi
- Synonyms Mesoxalicacid, diethyl ester (6CI,8CI);Propanedioic acid, oxo-, diethyl ester (9CI);2-Oxomalonic acid diethyl ester;Diethyl 2-oxomalonate;Diethyl2-oxopropane-1,3-dioate;Diethyl ketomalonate;Diethyl mesoxalate;Diethyloxomalonate;Diethyl oxopropanedioate;Ethyl ketomalonate;Ethyl mesoxalate;Ethyl oxomalonate;NSC 53478;Oxopropanedioic acid diethyl ester;
- PSA 69.67000
- LogP -0.31830
Synthetic route

| Conditions | Yield |
|---|---|
| With 3,3-dimethyldioxirane In acetone for 29h; Ambient temperature; | 100% |
| With tert-butylhypochlorite In formic acid | |
| With chloro(1,5-cyclooctadiene)rhodium(I) dimer; 1,1'-sulfinylbisbenzene; lanthanum(lll) triflate In 1,4-dioxane at 120℃; for 16h; Glovebox; Sealed tube; | 50 %Spectr. |
-
-
18856-68-3
diethyl N,N-dimethylaminomethylenemalonate

-
-
609-09-6
Diethyl ketomalonate

| Conditions | Yield |
|---|---|
| With ozone | 94% |

| Conditions | Yield |
|---|---|
| With hydrogenchloride; sodium chlorate; dihydrogen peroxide; sodium hydroxide In water at 10 - 35℃; pH=5 - 10; | 89% |
| With chlorine dioxide; sodium hydroxide In water at 10 - 18℃; for 5h; pH=10; | 89% |
| Stage #1: diethyl malonate With acetic acid In water at 70 - 80℃; for 0.5h; Stage #2: With sodium chlorite In water at 0 - 80℃; | 56% |

| Conditions | Yield |
|---|---|
| With aluminum oxide; tetrabutylammomium bromide; oxygen; cyclohexene In acetonitrile Hg cathode, Pt anode, -1.0 V vs SCE; | 83% |

| Conditions | Yield |
|---|---|
| With formaldehyd; ozone; Pd-on-C In methanol | 82% |
| Yield given. Multistep reaction; |

| Conditions | Yield |
|---|---|
| With silver nitrate In ethanol; acetonitrile for 22h; Heating; | 77% |
| With Amberlyst A-26; nitrate form In benzene for 7h; Heating; | 70% |
| With pyridine N-oxide In acetone; acetonitrile; Petroleum ether | |
| Multi-step reaction with 2 steps 1: alcohol / 40 - 50 °C 2: bromine / 110 °C / Destillation des Reaktionsprodukts unter verminderten Druck View Scheme |

| Conditions | Yield |
|---|---|
| With hydrogenchloride; sodium chlorate; dihydrogen peroxide; sodium hydroxide In water at 10 - 35℃; pH=5 - 10; | A n/a B 76% |
| With chlorine dioxide; sodium hydroxide In water at 10 - 18℃; for 5h; | A n/a B 76% |
-
-
91473-89-1
5--4,5-dihydro-2,5-dimethyl-4-phenyl-3-furancarbonsaeure-ethylester

-
A
-
16206-31-8
2,5-Dimethyl-4-phenyl-3-pyrrolcarbonsaeure-ethylester

-
B
-
609-09-6
Diethyl ketomalonate

| Conditions | Yield |
|---|---|
| With hydrogenchloride In methanol; water Heating; | A 51% B n/a |
-
-
82255-47-8
5-Methoxy-[1,2,4]trioxolane-3,3-dicarboxylic acid diethyl ester

-
-
609-09-6
Diethyl ketomalonate

| Conditions | Yield |
|---|---|
| With phosphorus trichloride for 8h; Ambient temperature; | 44% |
| With phosphorus trichloride for 8h; Ambient temperature; | 44% |
-
-
119174-42-4
2-ethoxy-2-oxoacetic anhydride

-
-
609-09-6
Diethyl ketomalonate

| Conditions | Yield |
|---|---|
| at 240℃; |
-
-
64-17-5
ethanol

-
-
631-22-1
diethyl dibromomalonate

-
-
127-08-2
potassium acetate

-
-
609-09-6
Diethyl ketomalonate


| Conditions | Yield |
|---|---|
| With phosphorus pentoxide Destillieren; |
-
-
59743-08-7
diethyl 2,3-dioxosuccinate

-
-
609-09-6
Diethyl ketomalonate

| Conditions | Yield |
|---|---|
| With bromine at 110℃; Destillation des Reaktionsprodukts unter verminderten Druck; |
-
-
42065-95-2
bromo-nitro-malonic acid diethyl ester

-
-
609-09-6
Diethyl ketomalonate
-
-
105-53-3
diethyl malonate

-
A
-
631-23-2
dihydroxymalonic acid diethyl ester

-
B
-
609-09-6
Diethyl ketomalonate

| Conditions | Yield |
|---|---|
| With nitrogen oxides | |
| With ammonium cerium (IV) nitrate In acetonitrile at 0 - 20℃; for 4h; Overall yield = 69 %; |
-
-
13937-08-1
2-hydroxy-malonic acid diethyl ester

-
-
609-09-6
Diethyl ketomalonate

| Conditions | Yield |
|---|---|
| With bromine |

| Conditions | Yield |
|---|---|
| With 2-morpholin-4-yldisulfanyl-benzothiazole | |
| Multi-step reaction with 2 steps 1: ethanol / 1.) 50 deg C, 2 h, 2.) reflux, overnight 2: tetrabutylammonium bromide View Scheme |

| Conditions | Yield |
|---|---|
| With ozone; triphenylphosphine 1.) CH2Cl2, -78 deg C, 2 h; Yield given. Multistep reaction; |
-
-
541-41-3
chloroformic acid ethyl ester

-
-
105-56-6
ethyl 2-cyanoacetate

-
-
1227476-15-4
Azobenzene

-
A
-
122-66-7
diphenyl hydrazine

-
B
-
609-09-6
Diethyl ketomalonate

| Conditions | Yield |
|---|---|
| With tetrabutylammomium bromide; oxygen; cyclohexene 1.) acetonitrile, -1.3 V vs. SCE 2.) acetonitrile, -1.0 V vs. SCE; Yield given. Multistep reaction; |

-
-
60308-73-8
diethyl α-acetoxy-α-bromomalonate

-
-
609-09-6
Diethyl ketomalonate

| Conditions | Yield |
|---|---|
| With tetrabutylammomium bromide Yield given; |

| Conditions | Yield |
|---|---|
| at 120 - 130℃; |
-
-
7446-08-4
selenium(IV) oxide

-
-
105-53-3
diethyl malonate

-
A
-
124-38-9
carbon dioxide

-
B
-
141-78-6
ethyl acetate

-
C
-
609-09-6
Diethyl ketomalonate

| Conditions | Yield |
|---|---|
| bei Siedetemperatur; |

-
-
56-23-5
tetrachloromethane

-
-
7446-08-4
selenium(IV) oxide

-
-
105-53-3
diethyl malonate

-
A
-
27728-17-2
oxomalonic acid monoethyl ester

-
B
-
95-92-1
oxalic acid diethyl ester

-
C
-
609-09-6
Diethyl ketomalonate

| Conditions | Yield |
|---|---|
| Produkt 5: CO2; |


-
-
609-09-6
Diethyl ketomalonate

| Conditions | Yield |
|---|---|
| With dinitrogen pentoxide at 0℃; |

| Conditions | Yield |
|---|---|
| With hydrogenchloride |
-
-
930-87-0
1,2,5-trimethyl-1H-pyrrole

-
-
609-09-6
Diethyl ketomalonate

-
-
95679-85-9
2-Hydroxy-2-(1,2,5-trimethyl-1H-pyrrol-3-yl)-malonic acid diethyl ester

| Conditions | Yield |
|---|---|
| In dichloromethane at 20℃; under 750.06 Torr; for 24h; | 100% |
-
-
150-78-7
1,4-dimethoxybezene

-
-
609-09-6
Diethyl ketomalonate

-
-
83026-20-4
diethyl (2,5-dimethoxyphenyl)tartronate

| Conditions | Yield |
|---|---|
| With tin(IV) chloride In dichloromethane 1.) 0 deg C, 10 min, 2.) 20 deg C, 19 h; | 100% |
-
-
940926-96-5
1-methoxy-3-trimethylsilyl-1,3-butadiene

-
-
609-09-6
Diethyl ketomalonate

-
-
103560-11-8
6-Methoxy-4-trimethylsilanyl-3,6-dihydro-pyran-2,2-dicarboxylic acid diethyl ester

| Conditions | Yield |
|---|---|
| In benzene at 50℃; for 10h; | 100% |
-
-
92406-83-2
(3R,5R,7R)-1-(buta-1,3-dien-2-yl)adamantane

-
-
609-09-6
Diethyl ketomalonate

-
-
92406-95-6
4-Adamantan-1-yl-3,6-dihydro-pyran-2,2-dicarboxylic acid diethyl ester

| Conditions | Yield |
|---|---|
| In toluene for 39h; Heating; | 100% |
-
-
137073-22-4
2-(2,2,2-trifluoroethoxy)-4-oxo-5,6-benzo-1,3,2-dioxaphosphorinane

-
-
609-09-6
Diethyl ketomalonate

| Conditions | Yield |
|---|---|
| In dichloromethane | 100% |
-
-
137073-23-5
2-(2,2,3,3-tetrafluoropropoxy)benzo[e][1,3,2]dioxaphosphinin-4-one

-
-
609-09-6
Diethyl ketomalonate

| Conditions | Yield |
|---|---|
| In dichloromethane | 100% |
-
-
139101-75-0
Methyl-5-hydroxy-2,2-diphenyl-4H-1,3-dithiin-6-carboxylat

-
-
609-09-6
Diethyl ketomalonate

-
-
139101-79-4
Diethyl-2-hydroxy-2-(6-methoxycarbonyl-5-oxo-2,2-diphenyl-1,3-dithian-4-yl)malonat

| Conditions | Yield |
|---|---|
| With 4-methyl-morpholine In benzene for 12h; | 100% |
-
-
68014-21-1
triphenyl(t-butoxycarbonylimino)phosphorane

-
-
609-09-6
Diethyl ketomalonate

-
-
408346-61-2
2-(N-tert-butoxycarbonylimino)malonic acid diethyl ester

| Conditions | Yield |
|---|---|
| In tetrahydrofuran for 24h; aza-Wittig reaction; Heating; | 100% |
| In tetrahydrofuran at 80℃; for 12h; | 89% |
| In tetrahydrofuran for 18h; Heating; | |
| In tetrahydrofuran for 18h; Reflux; |
-
-
1449105-33-2
4,4-difluoro-3,5,8-trimethyl-4-bora-3a,4a-diaza-s-indacene

-
-
609-09-6
Diethyl ketomalonate

| Conditions | Yield |
|---|---|
| With piperidine; acetic acid In toluene at 60℃; | 100% |
-
-
121207-31-6
4,4-difluoro-1,3,5,7,8-pentamethyl-4-bora-3a,4a-diaza-s-indecene

-
-
609-09-6
Diethyl ketomalonate

| Conditions | Yield |
|---|---|
| With piperidine; acetic acid In toluene at 60℃; | 100% |
-
-
108-88-3
toluene

-
-
609-09-6
Diethyl ketomalonate

-
-
412014-15-4
diethyl 2-hydroxy-2-(4-methylphenyl)propane-1,3-dioate

| Conditions | Yield |
|---|---|
| With tin(IV) chloride at 20℃; for 2h; | 99% |
| With tin(IV) chloride |
-
-
53339-59-6
3,7-dimethyl-6-octenylamine

-
-
609-09-6
Diethyl ketomalonate

-
-
119392-65-3
Diethyl (3',7'-Dimethyl-6-octenylimino)malonate

| Conditions | Yield |
|---|---|
| In benzene Heating; | 99% |
-
-
100507-81-1
2-(n-pentyl)-1-ethoxy-1,3-butadiene

-
-
609-09-6
Diethyl ketomalonate

| Conditions | Yield |
|---|---|
| at 50℃; for 6h; | 99% |
-
-
10034-09-0
1-methoxy-1,3-butadiene

-
-
609-09-6
Diethyl ketomalonate

-
-
23012-61-5
6-Methoxy-3,6-dihydro-pyran-2,2-dicarboxylic acid diethyl ester

| Conditions | Yield |
|---|---|
| 99% |
-
-
95832-86-3
(S)-3-acetoxy-1-triphenylphosphoranylidene-2-butanone

-
-
609-09-6
Diethyl ketomalonate

| Conditions | Yield |
|---|---|
| In chloroform for 48h; Ambient temperature; | 99% |

| Conditions | Yield |
|---|---|
| With trimethylsilyl trifluoromethanesulfonate In 1,2-dichloro-ethane at 60℃; for 12h; stereoselective reaction; | 99% |

| Conditions | Yield |
|---|---|
| With trimethylsilyl trifluoromethanesulfonate In 1,2-dichloro-ethane at 60℃; for 12h; | 99% |
-
-
56594-98-0
2-(3-methylphenyl)-1-cyclopropylethan-1-one

-
-
609-09-6
Diethyl ketomalonate

-
-
1198272-10-4
C17H18O5

| Conditions | Yield |
|---|---|
| With trimethylsilyl trifluoromethanesulfonate In 1,2-dichloro-ethane at 60℃; for 12h; | 99% |

-
-
609-09-6
Diethyl ketomalonate

| Conditions | Yield |
|---|---|
| Stage #1: 1-benzyl-3-(1-cyanovinyl)-2-oxoindolin-3-yl tert-butyl carbonate; Diethyl ketomalonate In ethyl acetate at -40℃; for 0.0833333h; Stage #2: With C29H28N2O In ethyl acetate at -40℃; enantioselective reaction; | 99% |

| Conditions | Yield |
|---|---|
| In acetonitrile at 60℃; for 18h; | 99% |
-
-
603-76-9
1-methylindole

-
-
609-09-6
Diethyl ketomalonate

-
-
127744-44-9
diethyl α-hydroxy-α-(1-methylindol-3-yl)-malonate

| Conditions | Yield |
|---|---|
| In toluene for 3h; Heating; | 98% |
| In toluene at 100℃; for 3h; Heating; | 95% |
-
-
17015-31-5
1-ethoxy-2-methyl-1,3-butadien

-
-
609-09-6
Diethyl ketomalonate

| Conditions | Yield |
|---|---|
| at 50℃; for 6h; | 98% |
-
-
81420-09-9
Acetic acid (3E,5Z)-6-methoxy-hexa-3,5-dienyl ester

-
-
609-09-6
Diethyl ketomalonate

-
-
81420-13-5
(3S,6R)-3-(2-Acetoxy-ethyl)-6-methoxy-3,6-dihydro-pyran-2,2-dicarboxylic acid diethyl ester

| Conditions | Yield |
|---|---|
| In benzene at 60℃; under 6375510 Torr; for 48h; | 98% |
-
-
20205-48-5, 77003-43-1, 77131-40-9, 98856-65-6
8-oxo-7-azabicyclo<4.2.0>oct-2-ene

-
-
609-09-6
Diethyl ketomalonate

-
-
77131-41-0
2-Hydroxy-2-((1S,6R)-8-oxo-7-aza-bicyclo[4.2.0]oct-3-en-7-yl)-malonic acid diethyl ester

| Conditions | Yield |
|---|---|
| for 24h; Ambient temperature; | 98% |
-
-
84028-77-3
(3-methoxycarbonylpropionyl)methylenetriphenylphosphorane

-
-
609-09-6
Diethyl ketomalonate

-
-
206999-00-0
2-Ethoxycarbonyl-4-oxo-hept-2-enedioic acid 1-ethyl ester 7-methyl ester

| Conditions | Yield |
|---|---|
| In chloroform for 48h; Ambient temperature; | 98% |

| Conditions | Yield |
|---|---|
| With trifluoroacetic acid In toluene at 85℃; for 2h; Pictet-Spegler condensation; | 98% |

| Conditions | Yield |
|---|---|
| With trifluoroacetic acid In toluene at 85℃; for 2h; Pictet-Spegler condensation; | 98% |

-
-
609-09-6
Diethyl ketomalonate

| Conditions | Yield |
|---|---|
| With trifluoroacetic acid In toluene at 85℃; for 4h; Pictet-Spegler condensation; | 98% |
-
-
1086018-36-1, 1086018-32-7, 1086018-34-9
α-tert-butoxycarbonyl-α-phenyl-γ-methylidene-δ-valerolactone

-
-
609-09-6
Diethyl ketomalonate

-
-
1181692-87-4
3-tert-butyl 2,2-diethyl 5-methylene-3-phenyl-3,4-dihydro-2H-pyran-2,2,3(3H,4H)-tricarboxylatecarboxylate

| Conditions | Yield |
|---|---|
| With allyl(cyclopentadiene)palladium(II); C38H30NO2P In tetrahydrofuran at 40℃; for 3h; diastereoselective reaction; | 98% |
Diethyl ketomalonate Specification
The CAS registry number of Propanedioic acid,2-oxo-,1,3-diethyl ester is 609-09-6. Its EINECS registry number is 210-176-2. The IUPAC name is diethyl 2-oxopropanedioate. In addition, the molecular formula is C7H10O5 and the molecular weight is 174.15. It is also called Oxopropanedioic acid diethyl ester. What's more, it is a kind of clear yellow liquid and belongs to the class of Pharmaceutical Intermediates. It should be stored in sealed container, and placed in a cool and dry place.
Physical properties about this chemical are: (1)ACD/LogP: 0.65; (2)ACD/LogD (pH 5.5): 0.65; (3)ACD/LogD (pH 7.4): 0.65; (4)ACD/BCF (pH 5.5): 1.83; (5)ACD/BCF (pH 7.4): 1.83; (6)ACD/KOC (pH 5.5): 53.58; (7)ACD/KOC (pH 7.4): 53.58; (8)#H bond acceptors: 5; (9)#Freely Rotating Bonds: 6; (10)Polar Surface Area: 69.67 Å2; (11)Index of Refraction: 1.427; (12)Molar Refractivity: 38.05 cm3; (13)Molar Volume: 148.1 cm3; (14)Polarizability: 15.08 ×10-24cm3; (15)Surface Tension: 36.8 dyne/cm; (16)Density: 1.175 g/cm3; (17)Flash Point: 90.2 °C; (18)Enthalpy of Vaporization: 44.53 kJ/mol; (19)Boiling Point: 209 °C at 760 mmHg; (20)Vapour Pressure: 0.208 mmHg at 25°C.
Preparation of Propanedioic acid,2-oxo-,1,3-diethyl ester: it can be prepared by diethyl propanedioate. Pass dinitrogen trioxide gas into diethyl propanedioate. The reactant will become green. The reaction should react for 2 days at room temperature. At last, you should collect 103-108 °C(2.0kPa) fractions through vacuum distillation. The yield is about 74-76%.
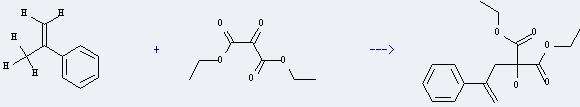
Uses of Propanedioic acid,2-oxo-,1,3-diethyl ester: it is used in the organic synthesis. And it can react with isopropenylbenzene to get Diethyl 2-phenylallyl hydroxymalonat. This reaction will need solvent xylene. The reaction time is 144 hours by heating.
When you are using this chemical, please be cautious about it as the following:
During using it, you should avoid contact with skin and eyes.
You can still convert the following datas into molecular structure:
(1)SMILES: O=C(OCC)C(=O)C(=O)OCC
(2)InChI: InChI=1/C7H10O5/c1-3-11-6(9)5(8)7(10)12-4-2/h3-4H2,1-2H3
(3)InChIKey: DBKKFIIYQGGHJO-UHFFFAOYAK
Related Products
- Diethyl (1-phenylpropyl)malonate
- Diethyl (2-(cyclohexylamino)vinyl)phosphonate
- Diethyl (2-(triethoxysilyl)ethyl)phosphonate
- Diethyl (2,4,6-trifluorophenyl)malonate
- Diethyl (2-oxopropyl)phosphonate
- Diethyl (2-thienylmethyl)phosphonate
- Diethyl (4-cyanobenzyl)phosphonate
- Diethyl (4-nitrobenzyl)phosphonate
- Diethyl (E)-2,3-diethylbut-2-enedioate
- Diethyl (hydroxymethyl)phosphonate
- 609-12-1
- 60912-51-8
- 60912-52-9
- 60912-78-9
- 609-14-3
- 6091-44-7
- 609-15-4
- 6091-64-1
- 60919-46-2
- 609-19-8
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View