-
Name
Dimethyl sulfate
- EINECS 201-058-1
- CAS No. 77-78-1
- Article Data54
- CAS DataBase
- Density 1.323 g/cm3
- Solubility 2.8 g/100 mL (18 ºC)
- Melting Point -32 °C
- Formula C2H6O4S
- Boiling Point 187.999 °C at 760 mmHg
- Molecular Weight 126.133
- Flash Point 83.333 °C
- Transport Information UN 1595 6.1/PG 1
- Appearance colourless liquid
- Safety 53-45
- Risk Codes 45-25-26-34-43-68
-
Molecular Structure
-
Hazard Symbols
 T+
T+
- Synonyms Dimethyl sulphate;Methyl sulfate;NSC 56194;
- PSA 60.98000
- LogP 0.60480
Synthetic route

| Conditions | Yield |
|---|---|
| With sulfur trioxide In dichloromethane at -50℃; | A 7% B 93% |

| Conditions | Yield |
|---|---|
| With sulfuric acid for 18h; Reflux; Inert atmosphere; | A 88% B n/a |

-
-
121-45-9
phosphorous acid trimethyl ester

-
A
-
512-56-1
trimethyl phosphite

-
B
-
152-18-1
O,O,O-trimethylthiophosphate

-
C
-
77-78-1
dimethyl sulfate

-
D
-
756-79-6
dimethyl methane phosphonate

| Conditions | Yield |
|---|---|
| With sulfur trioxide In dichloromethane at -78℃; Product distribution; other trialkyl phosphite,trialkyl phosphine, trialkyl arsines, trialkoxyarsines, var. molar ratio and temperatures; | A 22% B 4% C 55% D 19% |
| With sulfur trioxide In dichloromethane at -78℃; | A 48% B 16% C 3% D 9% |


| Conditions | Yield |
|---|---|
| With sulfur trioxide In dichloromethane at -50℃; | A 39% B 27% |
-
-
6596-95-8
trimethylarsenite

-
A
-
80398-43-2
methoxyarsenic bis(methyl sulfate)

-
B
-
77-78-1
dimethyl sulfate

| Conditions | Yield |
|---|---|
| With sulfur trioxide In dichloromethane at -50℃; | A 28% B 38% |

| Conditions | Yield |
|---|---|
| With sulfur trioxide In dichloromethane at -50℃; | A 19% B 21% |

| Conditions | Yield |
|---|---|
| With sulfuric acid Etherification; Heating; | A n/a B 10% |
-
-
186581-53-3, 908094-01-9
diazomethane

-
-
77-78-1
dimethyl sulfate

| Conditions | Yield |
|---|---|
| With diethyl ether; sulfuric acid |
-
-
67-56-1
methanol

-
-
812-01-1
methyl chlorosulfate

-
A
-
74-87-3
methylene chloride

-
B
-
75-93-4
methyl bisulfate

-
C
-
77-78-1
dimethyl sulfate

| Conditions | Yield |
|---|---|
| at -15 - -10℃; und folgenden Destillieren unter vermindertem Druck; |


| Conditions | Yield |
|---|---|
| With chlorosulfonic acid at -15 - -10℃; | |
| With sulfuric acid bei der Destillation; | |
| With sulfuric acid anschliessend man destilliert das Reaktionsgemisch im Vakuum; |
-
-
624-91-9
methyl nitrite

-
-
625-01-4
ethyl chlorosulfate

-
A
-
64-67-5
diethyl sulfate

-
B
-
814-40-4
ethyl methane sulfate

-
C
-
77-78-1
dimethyl sulfate


| Conditions | Yield |
|---|---|
| at 120 - 190℃; |
-
-
616-42-2
dimethylsulfite

-
-
812-01-1
methyl chlorosulfate

-
A
-
74-87-3
methylene chloride

-
B
-
77-78-1
dimethyl sulfate

| Conditions | Yield |
|---|---|
| at 120 - 190℃; |


| Conditions | Yield |
|---|---|
| With chlorine | |
| With chlorine at 120 - 140℃; |

| Conditions | Yield |
|---|---|
| bei der Destillation im Vakuum; |

| Conditions | Yield |
|---|---|
| With chlorosulfonic acid at 90 - 100℃; |

| Conditions | Yield |
|---|---|
| With sulfur trioxide | |
| With sulfuric acid at 160℃; man leitet, erhitzt ist, und destilliert dann das Gemisch; | |
| With sulfur trioxide Darstellung; | |
| With sulfuric acid byproducts: H2O; | |
| With H2SO4 byproducts: H2O; |

| Conditions | Yield |
|---|---|
| at 60 - 100℃; reagiert analog mit Monoaethylsulfat; | |
| at 60 - 100℃; | |
| at 60 - 100℃; |
-
-
78456-50-5
2-(4-methylphenyl)-2,3-dimethyl-1,3-oxazolidine

-
A
-
109-83-1
(2-hydroxyethyl)(methyl)amine

-
B
-
77-78-1
dimethyl sulfate

| Conditions | Yield |
|---|---|
| With sodium hydroxide; acetic acid buffer In acetonitrile at 25℃; Rate constant; Mechanism; var. time, var. buffer, var. pH; |
-
-
80-48-8
methyl p-toluene sulfonate

-
A
-
10506-59-9
dimethyl disulfate

-
B
-
4124-41-8
p-toluenesulfonylanhydride

-
C
-
49829-22-3
methyl 4-toluenepyrosulfonate

-
D
-
76443-13-5
C8H10O9S3

-
E
-
76443-14-6
mixed anhydride of 4-toluenesulfonic acid and 4-toluenepyrosulfonic acid

-
F
-
77-78-1
dimethyl sulfate

| Conditions | Yield |
|---|---|
| With sulfur trioxide at 100℃; Product distribution; other temperature, other molar ratio of reagents; | A n/a B n/a C 26.7 % Spectr. D 4.5 % Spectr. E n/a F n/a |

-
-
186581-53-3, 908094-01-9
diazomethane

-
-
60-29-7
diethyl ether

-
-
7664-93-9
sulfuric acid

-
-
77-78-1
dimethyl sulfate

| Conditions | Yield |
|---|---|
| reagiert analog mit Selensaeure; |


-
-
77-78-1
dimethyl sulfate

-
-
77-78-1
dimethyl sulfate

| Conditions | Yield |
|---|---|
| Darstellung; |

| Conditions | Yield |
|---|---|
| Alkaline conditions; | 100% |
| With sodium hydroxide In water at 70 - 80℃; for 1h; | 90% |
| With sodium hydroxide for 0.5h; | 90% |

| Conditions | Yield |
|---|---|
| With tetraethylammonium hydroxide at 0 - 20℃; for 1h; | 100% |
| With sodium hydroxide In water for 1h; Heating; | 99% |
| With potassium hydroxide | |
| With potassium carbonate In acetone Heating; Yield given; | |
| Alkaline conditions; |

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetone | 100% |
| Stage #1: salicylaldehyde With sodium hydroxide In water Stage #2: dimethyl sulfate In 1,4-dioxane; water at 70℃; for 0.0333333h; Temperature; Solvent; Concentration; | 95.6% |
| With sodium hydroxide | 86% |
-
-
89-63-4
4-Chloro-2-nitroaniline

-
-
77-78-1
dimethyl sulfate

-
-
15950-17-1
4-chloro-N-methyl-2-nitroaniline

| Conditions | Yield |
|---|---|
| With sodium hydroxide; tetra(n-butyl)ammonium hydrogensulfate In water; toluene at 20℃; Methylation; | 100% |
| With tetra(n-butyl)ammonium hydrogensulfate; sodium hydroxide In water; toluene at 20℃; | 94% |
-
-
5792-36-9
2',4'-dihydroxypropiophenone

-
-
77-78-1
dimethyl sulfate

-
-
6270-44-6
2-hydroxy-4-methoxypropiophenone

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetone for 7h; Heating; | 100% |
| With potassium carbonate; benzene | |
| With potassium carbonate In acetone Heating; |

| Conditions | Yield |
|---|---|
| In acetonitrile Cooling with ice; | 100% |
| In acetone for 5h; | 75% |

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetone at 20℃; | 100% |
| With potassium carbonate In acetone at 20℃; for 4h; | 93% |
| With potassium hydroxide | |
| With potassium carbonate at 25℃; for 1h; |

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetone for 22h; Reflux; | 100% |
| With potassium carbonate; acetone |

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetone for 3h; | 100% |
| With potassium carbonate In acetone for 3h; Ambient temperature; | 100% |
| With sodium hydroxide |
-
-
480-66-0
2,4,6-trihydroxyacetophenone

-
-
77-78-1
dimethyl sulfate

-
-
90-24-4
2-hydroxy-4,6-dimethoxyacetophenone

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetone Inert atmosphere; | 100% |
| With potassium carbonate In acetone for 3h; Reflux; | 100% |
| With potassium carbonate In acetone at 66℃; for 2h; Inert atmosphere; | 100% |
-
-
70547-87-4
2,6-dimethyl-4-hydroxybenzaldehyde

-
-
77-78-1
dimethyl sulfate

-
-
19447-00-8
4-methoxy-2,6-dimethylbenzylaldehyde

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetonitrile at 25℃; for 16h; | 100% |
| With sodium hydroxide In water at 10℃; for 2h; | 85% |
| With potassium hydroxide at 35℃; |

| Conditions | Yield |
|---|---|
| With potassium carbonate | 100% |
| With potassium hydroxide at 50℃; | |
| With potassium hydroxide | |
| With potassium carbonate In DMF (N,N-dimethyl-formamide) at 70 - 115℃; for 10.5h; | |
| With potassium carbonate In acetone |

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetone at 20℃; for 24h; | 100% |
| With potassium carbonate In butanone for 24h; | 94% |
| With potassium hydroxide In methanol for 8h; steam distillation; | 82% |

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetone | 100% |
| With potassium hydroxide | |
| With potassium carbonate In acetone Heating; | |
| With potassium carbonate In acetone |

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetonitrile Reflux; Inert atmosphere; | 100% |
| With potassium hydroxide |
-
-
23121-32-6
1-(2-hydroxy-4,6-dimethoxy-3-methyl-phenyl)-ethanone

-
-
77-78-1
dimethyl sulfate

-
-
39701-13-8
2',4',6'-trimethoxy-3'-methylacetophenone

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetone | 100% |
| With potassium hydroxide | |
| With potassium carbonate; acetone |
-
-
52427-05-1
2-bromo-5-nitrophenol

-
-
77-78-1
dimethyl sulfate

-
-
77337-82-7
1-bromo-2-methoxy-4-nitrobenzene

| Conditions | Yield |
|---|---|
| Stage #1: 2-bromo-5-nitrophenol With lithium hydroxide In tetrahydrofuran at 0℃; for 1h; Stage #2: dimethyl sulfate at 20℃; | 100% |
| With sodium hydroxide |
-
-
197243-48-4
2-iodo-3-nitrophenol

-
-
77-78-1
dimethyl sulfate

-
-
98991-08-3
2-iodo-1-methoxy-3-nitrobenzene

| Conditions | Yield |
|---|---|
| With tetraethylammonium bromide; sodium hydroxide In dichloromethane; water at 20℃; for 6h; | 100% |
| With sodium hydroxide | |
| With potassium hydroxide Yield given; |

| Conditions | Yield |
|---|---|
| With sodium hydroxide; triisooctyl amine In dichloromethane; water at 0℃; for 10h; | 100% |
| With sodium hydroxide In dichloromethane; water at 0℃; for 10h; | 100% |
| With potassium carbonate In acetone for 15h; Heating; | 94% |
-
-
77-78-1
dimethyl sulfate

-
-
86-48-6
1-hydroxy-2-naphthoic acid

-
-
948-03-8
1-hydroxy-naphthalene-2-carboxylic acid methyl ester

| Conditions | Yield |
|---|---|
| With lithium hydroxide In tetrahydrofuran for 1h; Methylation; esterification; Heating; | 100% |
| Stage #1: 1-hydroxy-2-naphthoic acid With lithium hydroxide In tetrahydrofuran for 0.5h; Stage #2: dimethyl sulfate In tetrahydrofuran for 3h; Heating; | 95.9% |
| Stage #1: 1-hydroxy-2-naphthoic acid With triethylamine In acetone for 0.25h; Inert atmosphere; Stage #2: dimethyl sulfate In acetone at 55℃; for 12h; Inert atmosphere; | 95% |

| Conditions | Yield |
|---|---|
| With potassium fluoride In water at 100℃; for 5h; Solvent; Reagent/catalyst; Temperature; | 100% |
| With potassium fluoride In water at 100℃; under 1125.11 Torr; for 5h; Reagent/catalyst; Solvent; Temperature; | 100% |
| With potassium fluoride In sulfolane at 100℃; under 750.075 Torr; for 0.166667h; Solvent; Temperature; Autoclave; | 92% |
| With potassium fluoride | |
| With sodium fluoride In sulfolane |
-
-
68-12-2, 33513-42-7
N,N-dimethyl-formamide

-
-
77-78-1
dimethyl sulfate

-
-
21511-55-7, 34643-89-5
(methoxymethylidene)dimethylammonium methyl sulfate

| Conditions | Yield |
|---|---|
| at 80℃; for 2h; Schlenk technique; Inert atmosphere; | 100% |
| at 20 - 80℃; Inert atmosphere; | 100% |
| In neat (no solvent) at 20 - 80℃; Inert atmosphere; | 100% |
-
-
87-91-2
diethyl (2R,3R)-tartrate

-
-
77-78-1
dimethyl sulfate

-
-
27957-93-3
(+)-(2R,3R)-diethyl di-O-methyltartrate

| Conditions | Yield |
|---|---|
| With sodium hydride In diethyl ether at 0 - 20℃; Inert atmosphere; | 100% |
| With sodium hydride In diethyl ether at 0 - 20℃; Inert atmosphere; | 100% |
| With sodium hydride In diethyl ether at 0 - 20℃; | 100% |
| With sodium hydride In diethyl ether |

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetone for 32h; Inert atmosphere; Reflux; | 100% |
| With potassium carbonate In acetone | |
| With potassium carbonate In acetone for 32h; Inert atmosphere; Reflux; |
-
-
289-06-5
1,3,4-thiadiazole

-
-
77-78-1
dimethyl sulfate

-
-
114395-38-9
3-Methyl-1,3,4-thiadiazolium-methylsulfat

| Conditions | Yield |
|---|---|
| In dichloromethane for 0.5h; Heating; | 100% |
-
-
4394-85-8
4-morpholinecarboxaldehyde

-
-
77-78-1
dimethyl sulfate

-
-
5780-15-4
4-(Methoxymethylene)-morpholinium methyl sulfate

| Conditions | Yield |
|---|---|
| at 60 - 70℃; for 3h; | 100% |
| at 70℃; for 2h; |
-
-
719-59-5
2-Amino-5-chlorobenzophenone

-
-
77-78-1
dimethyl sulfate

-
-
1022-13-5
5-chloro-2-(methylamino)benzophenone

| Conditions | Yield |
|---|---|
| With sodium hydroxide; tetrabutylammomium bromide In tetrahydrofuran at 60℃; for 1h; | 100% |
| With sodium hydroxide; tetrabutylammomium bromide; potassium carbonate In tetrahydrofuran at 30 - 35℃; for 2.5h; | 96% |
-
-
58488-39-4
2,5-bis(1,3-benzodithiol-2-yl)pyrrole

-
-
77-78-1
dimethyl sulfate

-
-
153850-73-8
2,5-bis(1,3-benzodithiol-2-yl)-1-methylpyrrole

| Conditions | Yield |
|---|---|
| With sodium hydroxide; tetraethylammonium bromide In dichloromethane Heating; | 100% |
-
-
68-12-2, 33513-42-7
N,N-dimethyl-formamide

-
-
77-78-1
dimethyl sulfate

-
-
4637-24-5
N,N-dimethyl-formamide dimethyl acetal

| Conditions | Yield |
|---|---|
| With nitrogen at 80℃; for 2h; | 100% |
| With sodium 1.) 3 h, 60-80 degC: 2.) 0 degC, methanol; Yield given. Multistep reaction; | |
| at 60℃; for 3h; |
-
-
54287-99-9
5-hydroxy-2,2-dimethyl-2H-chromene-6-carbaldehyde

-
-
77-78-1
dimethyl sulfate

-
-
79571-17-8
5-(methoxy)-2,2-dimethyl-2H-chromene-6-carbaldehyde

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetone at 20℃; for 20h; Inert atmosphere; | 100% |
| With potassium carbonate In acetone at 20℃; for 20h; Inert atmosphere; | 100% |
| With potassium carbonate In acetone at 20℃; | 100% |
| With potassium carbonate In acetone for 1h; Heating; | 52 mg |
| With potassium carbonate In acetone at 20℃; for 20h; Inert atmosphere; | 1.5 g |
Dimethyl sulfate History
Dimethyl sulfate Consensus Reports
Dimethyl sulfate Standards and Recommendations
ACGIH TLV: TWA 0.1 ppm (skin); Animal Carcinogen
DFG MAK: DFG TRK: Production: 0.02 ppm; Use: 0.04 ppm; Animal Carcinogen, Suspected Human Carcinogen
DOT Classification: 3; Label: Poison, Corrosive
Dimethyl sulfate Analytical Methods
Dimethyl sulfate Specification
The IUPAC name of this chemical is Dimethyl sulfate. With the CAS registry number 77-78-1 and EINECS registry number 201-058-1, it is also named as Sulfuricacid, dimethyl ester. In addition, the molecular formula is C2H6O4S and the molecular weight is 126.13. It is a kind of colourless liquid and belongs to the class of Pharmaceutical Intermediates.
Physical properties about this chemical are: (1)ACD/LogP: 0.12; (2)ACD/LogD (pH 5.5): 0.121; (3)ACD/LogD (pH 7.4): 0.121; (4)ACD/BCF (pH 5.5): 1; (5)ACD/BCF (pH 7.4): 1; (6)ACD/KOC (pH 5.5): 27.723; (7)ACD/KOC (pH 7.4): 27.723; (8)#H bond acceptors: 4; (9)#Freely Rotating Bonds: 2; (10)Polar Surface Area: 60.98 Å2; (11)Index of Refraction: 1.41; (12)Molar Refractivity: 23.597 cm3; (13)Molar Volume: 95.306 cm3; (14)Polarizability: 9.355 ×10-24cm3; (15)Surface Tension: 36.492 dyne/cm; (16)Density: 1.323 g/cm3; (17)Flash Point: 83.333 °C; (18)Enthalpy of Vaporization: 40.684 kJ/mol; (19)Boiling Point: 187.999 °C at 760 mmHg; (20)Vapour Pressure: 0.845 mmHg at 25°C.
Preparation of Dimethyl sulfate: it can be prepared by methanol with sulfuric acid. The methanol react with sulfuric acid to give methyl hydrogen sulfate at first. Then the methyl hydrogen sulfate can react with methanol to get dimethyl ether. At last, dimethyl ether react with sulfur trioxide to prepare the product.
.png)
Uses of Dimethyl sulfate: it can be used as a reagent for the methylation of phenols, amines, and thiols. And it can effect the base-specific cleavage of guanine in DNA by rupturing the imidazole rings present in guanine. This process can be used to determine base sequencing, cleavage on the DNA chain, and other applications. In addition, it can react with 1H-benzoimidazole to get 1-methyl-1H-benzoimidazole. This reaction will need reagents K2CO3 and triethylbenzylammonium chloride and solvent benzene. The reaction time is 0.5 hour at reaction temperature of 20 °C. The yield is about 50%.
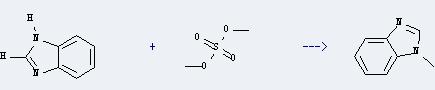
When you are using this chemical, please be cautious about it as the following:
This chemical is toxic if swallowed and very toxic by inhalation. And it can cause burns and may cause sensitization by skin contact. Moreover, it may cause cancer and has risk of irreversible effects possibly. You should avoid exposure - obtain special instruction before use. During using it, in case of accident or if you feel unwell, seek medical advice immediately (show label where possible).
You can still convert the following datas into molecular structure:
(1)SMILES: COS(=O)(=O)OC
(2)InChI: InChI=1/C2H6O4S/c1-5-7(3,4)6-2/h1-2H3
(3)InChIKey: VAYGXNSJCAHWJZ-UHFFFAOYAK
The toxicity data is as follows:
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| guinea pig | LC50 | inhalation | 32ppm/1H (32ppm) | "Prehled Prumyslove Toxikologie; Organicke Latky," Marhold, J., Prague, Czechoslovakia, Avicenum, 1986Vol. -, Pg. 1079, 1986. | |
| human | LCLo | inhalation | 97ppm/10M (97ppm) | "Toxicology of Drugs and Chemicals," Deichmann, W.B., New York, Academic Press, Inc., 1969Vol. -, Pg. 226, 1969. | |
| mouse | LC50 | inhalation | 280mg/m3 (280mg/m3) | SENSE ORGANS AND SPECIAL SENSES: OTHER CHANGES: OLFACTION BEHAVIORAL: SOMNOLENCE (GENERAL DEPRESSED ACTIVITY) LUNGS, THORAX, OR RESPIRATION: RESPIRATORY DEPRESSION | Gigiena Truda i Professional'nye Zabolevaniya. Labor Hygiene and Occupational Diseases. Vol. 23(3), Pg. 28, 1979. |
| mouse | LD50 | oral | 140mg/kg (140mg/kg) | Gigiena Truda i Professional'nye Zabolevaniya. Labor Hygiene and Occupational Diseases. Vol. 23(3), Pg. 28, 1979. | |
| rabbit | LDLo | intravenous | 50mg/kg (50mg/kg) | BEHAVIORAL: MUSCLE CONTRACTION OR SPASTICITY) | Archiv fuer Experimentelle Pathologie und Pharmakologie. Vol. 47, Pg. 113, 1902. |
| rabbit | LDLo | oral | 45mg/kg (45mg/kg) | BEHAVIORAL: FOOD INTAKE (ANIMAL) | Archiv fuer Experimentelle Pathologie und Pharmakologie. Vol. 47, Pg. 113, 1902. |
| rabbit | LDLo | subcutaneous | 53mg/kg (53mg/kg) | BEHAVIORAL: MUSCLE CONTRACTION OR SPASTICITY) SKIN AND APPENDAGES (SKIN): "DERMATITIS, OTHER: AFTER SYSTEMIC EXPOSURE" | Archiv fuer Experimentelle Pathologie und Pharmakologie. Vol. 47, Pg. 113, 1902. |
| rat | LC50 | inhalation | 45mg/m3/4H (45mg/m3) | LUNGS, THORAX, OR RESPIRATION: DYSPNEA LUNGS, THORAX, OR RESPIRATION: CYANOSIS BLOOD: HEMORRHAGE | Gigiena Truda i Professional'nye Zabolevaniya. Labor Hygiene and Occupational Diseases. Vol. 24(11), Pg. 55, 1980. |
| rat | LD50 | oral | 205mg/kg (205mg/kg) | Gigiena Truda i Professional'nye Zabolevaniya. Labor Hygiene and Occupational Diseases. Vol. 23(3), Pg. 28, 1979. | |
| rat | LD50 | subcutaneous | 100mg/kg (100mg/kg) | Zeitschrift fuer Krebsforschung. Vol. 74, Pg. 241, 1970. |
Related Products
- Dimethyl ( )-2,3-O-Isopropylidene-D-tartrate
- Dimethyl ((1-methyl-5-nitro-1H-imidazol-2-yl)methylene)propanedioate
- Dimethyl (2-oxo-3,3-difluoroheptyl)phosphonate
- Dimethyl (2-oxo-4-phenylbutyl)phosphonate
- Dimethyl (2-oxoheptyl)phosphonate
- Dimethyl (2S, 2'S)-1, 1'-((2S, 2'S)-2, 2'-(4, 4'-(biphenyl-4, 4'-diyl)bis(1H-imidazole-4, 2-diyl))bis(pyrrolidine-2, 1-diyl))bis(3-methyl-1-oxobutane-2, 1-diyl)dicarbamate
- Dimethyl (3-phenoxy-2-oxopropyl)phosphonate
- Dimethyl (R)-(+)-methylsuccinate
- Dimethyl (S)-(-)-methylsuccinate
- Dimethyl (S)-3-hydroxy-L-aspartate
- 7778-18-9
- 7778-39-4
- 77784-22-6
- 7778-43-0
- 7778-49-6
- 7778-50-9
- 7778-53-2
- 7778-54-3
- 7778-66-7
- 7778-74-7
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View