-
Name
Ethyl vinyl ether
- EINECS 203-718-4
- CAS No. 109-92-2
- Article Data95
- CAS DataBase
- Density 0.751 g/cm3
- Solubility 7.8 g/L (25 °C) in water
- Melting Point -116 °C(lit.)
- Formula C4H8O
- Boiling Point 32.5 °C at 760 mmHg
- Molecular Weight 72.1069
- Flash Point ?50°F
- Transport Information UN 1302 3/PG 1
- Appearance colourless liquid with an ether-like odour
- Safety 16-23-26-3/7-33-36
- Risk Codes 12-19-36/37/38
-
Molecular Structure
-
Hazard Symbols
 F+,
F+, Xi,
Xi, F
F
- Synonyms Ether, vinyl ethyl;1-Ethoxyethylene;Ether, ethyl vinyl (inhibited);Vinamar;Ethoxyethene;Ether, ethyl vinyl;Ethyl ethenyl ether;EVE;Ethene, ethoxy-;Ethene, ethoxy;1-Ethoxyethene;Ethylvinylether;supply ethyl vinyl ether;Ethoxyetylene, ethyl vinyl ether;
- PSA 9.23000
- LogP 1.16640
Synthetic route

| Conditions | Yield |
|---|---|
| With potassium hydroxide In dimethyl sulfoxide at 120℃; for 1.5h; | 92% |
| With sodium ethanolate at 145 - 165℃; | |
| With zinc(II) oxide at 310℃; |
-
-
64-17-5
ethanol

-
-
1112-54-5
vinyltriethylsilane

-
A
-
994-49-0
hexaethyl disiloxane

-
B
-
109-92-2
ethyl vinyl ether

| Conditions | Yield |
|---|---|
| With bromine for 4h; | A 25% B 55% |

-
-
54007-78-2
1-phenyl-4-ethoxy-2,3-dimethylthiobutanone

-
A
-
77787-36-1
2,4,4-Trimethyl-3-phenyl-tetrahydro-pyran-3-thiol

-
B
-
77787-34-9, 77787-35-0
4-Ethoxy-2,2-dimethyl-1-phenyl-cyclobutanethiol

-
C
-
109-92-2
ethyl vinyl ether

| Conditions | Yield |
|---|---|
| In benzene for 40h; Ambient temperature; Irradiation; through pyrex; | A 40% B 48% C n/a |
| With dimethylglyoxal In benzene Quantum yield; Rate constant; Ambient temperature; Irradiation; var. senzitizers and conc. of ones; sev. solvents of different polarities; |

-
-
925669-95-0
2,3-cis-epoxybutane

-
A
-
598-32-3
2-hydroxy-3-butene

-
B
-
74-85-1
ethene

-
C
-
78-84-2
isobutyraldehyde

-
D
-
109-92-2
ethyl vinyl ether

-
E
-
78-93-3
butanone

-
-
21490-63-1
2,3-trans-epoxybutane

| Conditions | Yield |
|---|---|
| at 757.9℃; Mechanism; Product distribution; Kinetics; reflected shocks in a pressurized driver single-pulse shock tube ; the temperature range: 900-1150 K.; | A 2.62% B 2.21% C 4.58% D 0.902% E 13.39% F 5.64% |


| Conditions | Yield |
|---|---|
| (CO)4(palladium)4(acetate)4 for 2h; Product distribution; effect of the catalyst to the production and the selectivity; | A 9% B n/a |

-
-
56-23-5
tetrachloromethane

-
-
592-55-2
2-Bromoethyl ethyl ether

-
A
-
112-36-7
3,6,9-trioxaundecane

-
B
-
109-92-2
ethyl vinyl ether

-
-
105-57-7
diethyl acetal

-
A
-
64-17-5
ethanol

-
B
-
74-85-1
ethene

-
C
-
75-07-0
acetaldehyde

-
D
-
109-92-2
ethyl vinyl ether

| Conditions | Yield |
|---|---|
| at 389 - 453℃; Kinetics; Pr.5: Kohlenmonoxid; Pr.6: Methan.Pyrolysis; |


| Conditions | Yield |
|---|---|
| With sulfuric acid at 350℃; | |
| With sodium hydrogen sulfate | |
| With platinum at 280 - 290℃; |

| Conditions | Yield |
|---|---|
| at 250℃; | |
| at 300 - 500℃; in Gegenwart von Katalysatoren; |

| Conditions | Yield |
|---|---|
| With mercury(II) diacetate | |
| With sodium hydroxide; kieselguhr at 310 - 320℃; |

| Conditions | Yield |
|---|---|
| With pyridine at 25℃; |

| Conditions | Yield |
|---|---|
| With sodium hydroxide | |
| With potassium hydroxide at 110 - 120℃; |

| Conditions | Yield |
|---|---|
| With alkali hydroxide |

| Conditions | Yield |
|---|---|
| at 100 - 120℃; | |
| unter Druck; reagiert analog mit anderen Alkoholaten; |
-
-
109393-74-0
3-ethoxy-2,2-diphenylcyclobutanone

-
A
-
525-06-4
diphenyl ketene

-
B
-
109-92-2
ethyl vinyl ether

| Conditions | Yield |
|---|---|
| bei gelindem Erwaermen oder laengerem Aufbewahren; |

| Conditions | Yield |
|---|---|
| at 100℃; unter Druck; |

| Conditions | Yield |
|---|---|
| With magnesium at 100 - 110℃; |

| Conditions | Yield |
|---|---|
| With sulfuric acid; mercury(II) sulfate Behandeln des entstandenen Reaktionsgemisch mit Alkohol; |

| Conditions | Yield |
|---|---|
| With sodium at 130 - 140℃; | |
| With diethyl ether; sodium at 0 - 20℃; |

| Conditions | Yield |
|---|---|
| With pyridine at 120℃; | |
| With potassium tert-butylate Kinetics; |

| Conditions | Yield |
|---|---|
| In various solvent(s) Product distribution; Irradiation; infrared multiphoton induced photolysis; the luminescence of the reaction mixture was also investigated; | |
| Irradiation; luminescence during the photodecompsition; |

| Conditions | Yield |
|---|---|
| In various solvent(s) Product distribution; Irradiation; infrared multiphoton induced photolysis; the luminescence of the reaction mixture was also investigated; | |
| Irradiation; luminescence during the photodecompsition; |
-
-
629-14-1
monoethylene glycol diethyl ether

-
A
-
22056-82-2
ethoxyacetaldehyde

-
B
-
16484-86-9
1,2-diethoxyethylene

-
C
-
120188-10-5
(2R,3R,4S,6R)-3,5-Diethoxy-6-ethoxymethyl-tetrahydro-pyran-2,4-diol

-
-
90201-72-2, 120188-09-2
2,4-di-O-ethylthreose

-
E
-
109-92-2
ethyl vinyl ether

| Conditions | Yield |
|---|---|
| Rhodium(I) compound In chloroform-d1 for 72h; Product distribution; |

-
-
80060-53-3
acetaldehyde ethyl 1-propenyl acetal

-
A
-
123-38-6
propionaldehyde

-
B
-
109-92-2
ethyl vinyl ether

| Conditions | Yield |
|---|---|
| (Ph3P)CH3COPdCl2 In dichloromethane Product distribution; Ambient temperature; |
-
-
107-19-7
propargyl alcohol

-
-
109-92-2
ethyl vinyl ether

-
-
18669-04-0
acetaldehyde ethyl propargyl acetal

| Conditions | Yield |
|---|---|
| With trifluoroacetic acid Inert atmosphere; | 100% |
| With trifluoroacetic acid at 20 - 60℃; for 0.333333h; | 100% |
| With toluene-4-sulfonic acid at 0 - 15℃; | 92% |
-
-
56-23-5
tetrachloromethane

-
-
109-92-2
ethyl vinyl ether

-
-
1561-41-7
Ethyl-(1,3,3,3-tetrachlor-propyl)-ether

| Conditions | Yield |
|---|---|
| With dibenzoyl peroxide | 100% |
| With dibenzoyl peroxide for 2h; Heating; | 90.3% |
| With pyridine; dibenzoyl peroxide |
-
-
109-92-2
ethyl vinyl ether

-
-
115-19-5
2-methyl-but-3-yn-2-ol

-
-
39807-00-6
2-methyl-3-butyn-2-ol 1-ethoxyethyl ether

| Conditions | Yield |
|---|---|
| With pyridinium p-toluenesulfonate In dichloromethane at 20℃; for 1h; | 100% |
| With sulfuric acid for 0.5h; | 77% |
| With hydrogenchloride |
-
-
687-47-8
(S)-Ethyl lactate

-
-
109-92-2
ethyl vinyl ether

-
-
184110-35-8
(2S,1'R/S)-Ethyl-2-(1'-ethoxyethoxy)propanoate

| Conditions | Yield |
|---|---|
| toluene-4-sulfonic acid | 100% |
| With toluene-4-sulfonic acid at -20℃; for 0.75h; | 98% |
| With pyridinium p-toluenesulfonate In dichloromethane 1.) 0 deg C, 50 min, 2.) 1 h, room temp.; | 97% |
-
-
121-33-5
vanillin

-
-
109-92-2
ethyl vinyl ether

-
-
35615-21-5
3-methoxy-4-(ethoxyethylidene-oxy)-benzaldehyde

| Conditions | Yield |
|---|---|
| With pyridinium p-toluenesulfonate In tetrahydrofuran at 20℃; for 30h; Etherification; | 100% |
| With pyridinium p-toluenesulfonate; sodium hydrogencarbonate In dichloromethane for 6h; Ambient temperature; | 90% |
| With pyridinium p-toluenesulfonate In dichloromethane |
-
-
937-32-6
4-nitrobenzenesulfenyl chloride

-
-
109-92-2
ethyl vinyl ether

-
-
81146-73-8
p-nitrophenyl 2-ethoxy-2-chloroethyl sulfide

| Conditions | Yield |
|---|---|
| In benzene at 20℃; for 0.5h; | 100% |
-
-
24915-95-5
Ethyl (R)-3-hydroxybutanoate

-
-
109-92-2
ethyl vinyl ether

| Conditions | Yield |
|---|---|
| trifluoroacetic acid | 100% |
-
-
617-55-0
dimethyl (S)-malate

-
-
109-92-2
ethyl vinyl ether

-
-
72229-30-2
(S)-(-)-dimethyl 2-O-(1-ethoxyethyl)malate

| Conditions | Yield |
|---|---|
| With pyridinium p-toluenesulfonate In dichloromethane for 36h; | 100% |
| With trifluoroacetic acid for 48h; Ambient temperature; Yield given; |
-
-
691-84-9
Diethyl (S)-malate

-
-
109-92-2
ethyl vinyl ether

-
-
929110-09-8
(S)-(-)-Aepfelsaeurediaethylester α-Aethoxyaethylaether

| Conditions | Yield |
|---|---|
| With trifluoroacetic acid at 20℃; for 120h; | 100% |
| With trifluoroacetic acid for 48h; Ambient temperature; | 98.8% |
| With hydrogenchloride; silver 1) 0 deg C to r.t., overnight, 2) 1 h; | 92.8% |
-
-
24573-30-6
3-chloro-2-hydroxy-1-propyl acetate

-
-
109-92-2
ethyl vinyl ether

-
-
85328-35-4
3-Chloro-2-(1-ethoxyethoxy)-1-propyl acetate

| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid at 35 - 45℃; for 20h; | 100% |
| With toluene-4-sulfonic acid at 35 - 40℃; for 18h; | |
| With pyridinium p-toluenesulfonate In dichloromethane | 44.0 g (98.0%) |
-
-
32837-87-9
3-hydroxy-3-methyl-1-cyano-1-butyne

-
-
109-92-2
ethyl vinyl ether

-
-
82819-73-6
3-methyl-3-(α-ethoxyethoxy)-1-cyano-1-butyne

| Conditions | Yield |
|---|---|
| With trifluoroacetic acid at 20 - 25℃; | 100% |
| With hydrogenchloride at 30℃; for 3h; | 65% |
-
-
59694-08-5
3-hydroxylbutyl benzoate

-
-
109-92-2
ethyl vinyl ether

-
-
345624-84-2
Benzoesaeure-<3-(1'-aethoxyaethoxy)-butyl>ester

| Conditions | Yield |
|---|---|
| With trifluoroacetic acid for 48h; Ambient temperature; | 100% |
-
-
99002-57-0
4-(2-Hydroxyethyl)-<2.2>paracyclophan

-
-
109-92-2
ethyl vinyl ether

-
-
123413-79-6
1-(<2.2>Paracyclophan-4-ethoxy)-1-ethoxy-ethan

| Conditions | Yield |
|---|---|
| With hydrogenchloride In dichloromethane for 5h; Ambient temperature; | 100% |
-
-
960-82-7, 13132-42-8, 51048-23-8, 18142-87-5
rac-methyl(1-naphthyl)phenylchlorosilane

-
-
109-92-2
ethyl vinyl ether

-
-
138380-27-5
(1-ethoxyethenyl)methyl(naphth-1-yl)phenylsilane

| Conditions | Yield |
|---|---|
| With tert.-butyl lithium In tetrahydrofuran; pentane 1) -78 to 23 deg C 2) 23 deg C, 30 min; | 100% |
-
-
473-61-0, 1196-00-5, 2734-31-8, 19889-94-2, 19889-95-3, 24041-60-9, 25465-65-0, 25465-95-6, 27779-29-9, 35997-96-7, 35998-01-7, 36129-11-0, 51152-11-5, 73366-08-2, 125473-76-9
(1S*,2R*,3R*)-2,6,6-Trimethylbicyclo<3.1.1>heptan-3-ol

-
-
109-92-2
ethyl vinyl ether

-
-
138905-21-2
(1S*,2R*,3R*)-3-(1-ethoxyethoxy)-2,6,6-trimethylbicyclo<3.1.1>heptane

| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid for 0.666667h; Ambient temperature; | 100% |
-
-
52267-81-9, 53906-54-0, 54142-65-3, 59685-87-9, 59685-90-4, 72029-56-2, 93302-71-7, 93302-72-8
<3aα,4α,5β,6aα>-(+/-)-hexahydro-5-hydroxy-4-<(E)-4-(3-chlorophenoxy)-(3α)-hydroxy-1-butenyl>-2H-cyclopentafuran-2-one

-
-
109-92-2
ethyl vinyl ether

-
-
109323-19-5
(1SR,5RS,6RS,7RS)-6-<(3SR)-4-(3-chlorophenoxy)-3-(1-ethoxy-ethoxy)-1(E)-butenyl>-7-(1-ethoxy-ethoxy)-2-oxabicyclo<3.3.0>octan-3-one

| Conditions | Yield |
|---|---|
| With pyridinium p-toluenesulfonate In dichloromethane at 20℃; for 0.5h; | 100% |
-
-
109431-72-3
(+)-(R)-2-cyclopentene-1-ol

-
-
109-92-2
ethyl vinyl ether

-
-
87453-50-7
1-(1-ethoxy-2-bromoethoxy)-2-cyclopentene

| Conditions | Yield |
|---|---|
| With N-Bromosuccinimide In dichloromethane 1) -40 to -50 deg C, 2h, 2) RT, 16h; | 100% |
-
-
32345-63-4, 82807-42-9
1-(3-methoxyphenyl)-1,2-ethanediol

-
-
109-92-2
ethyl vinyl ether

-
-
136178-90-0
1-ethoxyethyl 2-methoxy-2-(3-methoxyphenyl)ethyl ether

| Conditions | Yield |
|---|---|
| With trifluoroacetic acid | 100% |
-
-
127940-47-0, 131530-60-4
(4R*,4aS*,8aR*)-4,4a,5,8-Tetrahydro-4-hydroxy-4a-methoxycarbonyl-1(8aH)-naphthalenone

-
-
109-92-2
ethyl vinyl ether

-
-
127940-63-0
(4R*,4aS*,8aR*)-4-(1-Ethoxy)ethoxy-4,4a,5,8-tetrahydro-4a-methoxycarbonyl-1(8aH)-naphthalenone

| Conditions | Yield |
|---|---|
| With pyridinium p-toluenesulfonate In dichloromethane for 6h; Ambient temperature; | 100% |
| pyridinium p-toluenesulfonate In dichloromethane Ambient temperature; Yield given; |
-
-
110994-93-9
(S)-(-)-hydroxy-2 octanoate d'ethyle

-
-
109-92-2
ethyl vinyl ether

-
-
121404-65-7
ethyl-2-(1-ethoxyethoxy)octanoate

| Conditions | Yield |
|---|---|
| With pyridinium p-toluenesulfonate In dichloromethane for 1h; Ambient temperature; | 100% |
-
-
132127-34-5
(3R,4S)-3-hydroxy-4-phenylazetidin-2-one

-
-
109-92-2
ethyl vinyl ether

-
-
201856-48-6
(3R,4S)-3-<((R*)-1'-ethoxyethyl)oxy>-4-phenyl-2-azetidinone

| Conditions | Yield |
|---|---|
| With hydrogen cation | 100% |
| In tetrahydrofuran at 0℃; for 2h; | 100% |
| With toluene-4-sulfonic acid In tetrahydrofuran for 0.5h; | 95% |
| With methanesulfonic acid In tetrahydrofuran at 0℃; |
-
-
136178-92-2
2-(2-methoxyethoxy)-2-(3-methoxyphenyl)ethanol

-
-
109-92-2
ethyl vinyl ether

-
-
136178-91-1
1-ethoxyethyl 2-(2-methoxyethoxy)-2-(3-methoxyphenyl)ethyl ether

| Conditions | Yield |
|---|---|
| With trifluoroacetic acid | 100% |
-
-
90388-37-7
N-(2',4'-dichloro-6'-oxo-2',4'-cyclohexadien-1'-ylidene)-4-nitrobenzamide

-
-
109-92-2
ethyl vinyl ether

-
-
90368-45-9
(5,7-Dichloro-2-ethoxy-2,3-dihydro-benzo[1,4]oxazin-4-yl)-(4-nitro-phenyl)-methanone

| Conditions | Yield |
|---|---|
| In acetone at 20℃; Product distribution; Kinetics; Thermodynamic data; rate constants in various solvents, in benzene and 1,4-dioxane at different temperatures, ΔH(excit.), ΔS(excit.); | 100% |
-
-
79343-57-0
2-(1-acetyl-2-oxoindolin-3-ylidene)1,3-indandione

-
-
109-92-2
ethyl vinyl ether

-
-
79343-61-6, 79343-74-1
spiro<2,3,4,5-tetrahydro-2-ethoxy-5-ketoindano<1,2-b>pyran-4-3'(1'-acetyl-2'-oxo)indoline>

| Conditions | Yield |
|---|---|
| at 100℃; for 2h; Paar bomb; | 100% |
-
-
109950-62-1
1,3,4,4a,7,8,8a,9-octahydro-8aβ-hydroxymethyl-1α,4aβ-dimethyl-2,6-phenanthrenedione 2-(ethylene acetal)

-
-
109-92-2
ethyl vinyl ether

| Conditions | Yield |
|---|---|
| With pyridinium p-toluenesulfonate In dichloromethane for 1h; Ambient temperature; | 100% |
| With pyridinium p-toluenesulfonate In dichloromethane |
-
-
79343-56-9
1-acetyl-2-oxoindolin-3-ylidene diethylmalonate

-
-
109-92-2
ethyl vinyl ether

-
-
79343-60-5
2-ethoxy-4,4-dicarbethoxy-9-acetyl-2,3-dihydropyran<2,3-b>indole

| Conditions | Yield |
|---|---|
| In acetonitrile for 336h; Ambient temperature; | 100% |
-
-
139599-50-1
4α-hydroxy-1β-methylcyclohex-2-enecarbaldehyde

-
-
109-92-2
ethyl vinyl ether

-
-
139599-51-2
(1S,4R)-4-(1-Ethoxy-ethoxy)-1-methyl-cyclohex-2-enecarbaldehyde

| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid a) 0 deg C, 45 min, b) 0 deg C to RT, 2 h; | 100% |
-
-
76453-38-8
4-Hydroxymethyl-2-oxo-imidazolidine-1-carboxylic acid benzyl ester

-
-
109-92-2
ethyl vinyl ether

-
-
76453-39-9
4-(1-Ethoxy-ethoxymethyl)-2-oxo-imidazolidine-1-carboxylic acid benzyl ester

| Conditions | Yield |
|---|---|
| With trichloroacetic acid In 1,2-dimethoxyethane at 0℃; for 4h; | 100% |
-
-
144963-77-9, 144963-78-0
trans-6-benzyl-3-methyl-2-cyclohexen-1-ol

-
-
109-92-2
ethyl vinyl ether

-
-
144963-79-1
trans-4-benzyl-3-<(ξ)-1-ethoxy-2-iodoethoxy>-1-methylcyclohexene

| Conditions | Yield |
|---|---|
| With N-Bromosuccinimide In dichloromethane at -40℃; | 100% |
-
-
147378-92-5
2-<(R)-7-Hydroxyoctyl>-1,3-dithian

-
-
109-92-2
ethyl vinyl ether

-
-
1026008-60-5
2-<(R)-7-(2-Ethoxyethyl)octyl>-1,3-dithian

| Conditions | Yield |
|---|---|
| With trifluoroacetic acid at 0℃; for 48h; | 100% |
Ethyl vinyl ether Chemical Properties
Structure of Ethyl vinyl ether (CAS NO.109-92-2):
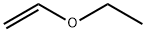
IUPAC Name: ethoxyethene
Empirical Formula: C4H8O
Molecular Weight: 72.1057
EINECS: 203-718-4
Index of Refraction: 1.376
Molar Refractivity: 22.05 cm3
Molar Volume: 96 cm3
Polarizability: 8.74×10-24cm3
Surface Tension: 18.8 dyne/cm
Density: 0.751 g/cm3
Enthalpy of Vaporization: 26.63 kJ/mol
Melting Point: −116 °C(lit.)
Boiling Point: 32.5 °C at 760 mmHg
Vapour Pressure: 581 mmHg at 25°C
Water Solubility: 7.8 g/L (25 ºC)
Physical Appearance: colourless liquid with an ether-like odour
Stability: Stable. Highly flammable. Incompatible with strong oxidizing agents. May form peroxides in storage - check for their formation before use.
Product Categories: Pharmaceutical Intermediates
Ethyl vinyl ether Uses
In medicine, Ethyl vinyl ether (CAS NO.109-92-2) is used as anesthetics, analgesics, but also as intermediates for fine chemical, intermediates for sulfadiazine. It makes up the monomer of copolymer, organic synthetic materials, spices amd lubricating oil additives.
Ethyl vinyl ether Production
Ethyl vinyl ether (CAS NO.109-92-2) was produced by the pressurized reaction of acetylene and ethanol in the presence of potassium hydroxide catalyst.
Ethyl vinyl ether Toxicity Data With Reference
| 1. | skn-rbt 500 mg open MLD | UCDS** Union Carbide Data Sheet. (Industrial Medicine and Toxicology Dept., Union Carbide Corp., 270 Park Ave., New York, NY 10017) 11/15 ,1971. | ||
| 2. | sce-ham:ovr 17,900 ppm | ANESAV Anesthesiology. 50 (1979),426. | ||
| 3. | orl-rat LD50:6153 mg/kg | AIHAAP American Industrial Hygiene Association Journal. 30 (1969),470. |
Ethyl vinyl ether Consensus Reports
Reported in EPA TSCA Inventory.
Ethyl vinyl ether Safety Profile
Hazard Codes:  F+,
F+, Xi,
Xi, F
F
Risk Statements: 12-19-36/37/38
R12:Extremely flammable.
R19:May form explosive peroxides.
R36/37/38:Irritating to eyes, respiratory system and skin.
Safety Statements: 16-23-26-3/7-33-36
S16:Keep away from sources of ignition.
S23:Do not breathe vapour.
S3:Keep in a cool place.
S7:Keep container tightly closed.
S33:Take precautionary measures against static discharges.
S36:Wear suitable protective clothing.
RIDADR: UN 1302 3/PG 1
WGK Germany: 1
RTECS: KO0710000
F: 19
HazardClass: 3
PackingGroup: I
HS Code: 29091900
Mildly toxic by ingestion. Mutation data reported. A skin irritant. A very dangerous fire and explosion hazard when exposed to heat or flame; can react vigorously with oxidizing materials. To fight fire, use alcohol foam, foam, CO2, dry chemical. Explosive polymerization is catalyzed by methane sulfonic acid. When heated to decomposition it emits acrid smoke and irritating fumes. See also ETHERS.
Ethyl vinyl ether Standards and Recommendations
DOT Classification: 3; Label: Flammable Liquid
Related Products
- Ethyl (13-cis)-9-(4-methoxy-2,3,6-trimethylphenyl)-3,7-dimethyl-2,4,6,8-nonatetraenoate
- ethyl (1R,2R)-1-phenyl-2-(trideuteriomethylamino)cyclohex-3-ene-1-carboxylate,hydrochloride
- Ethyl (1S,2R)-2-(dimethylamino)-1-phenylcyclohex-3-ene-1-carboxylate hydrochloride
- Ethyl (2,4,6-trimethylbenzoyl) phenylphosphinate
- Ethyl (2-amino-4-hydroxy-6-methyl-5-pyrimidinyl)acetate
- Ethyl (2-bromopropionamido)acetate
- Ethyl (2-cyanoimino-5,6-dichloro-1,2,3,4-tetrahydroquinazolin-3-yl)acetate
- ETHYL (2E,4Z)-DECADIENOATE
- Ethyl (2-hydroxyethyl)dimethyl-ammonium benzilate chloride
- Ethyl (2-mercaptoethyl) carbamate S-ester with O,O-dimethyl phosphorodithioate
- 109925-10-2
- 109926-11-6
- 109-94-4
- 109945-04-2
- 1099-45-2
- 109954-48-5
- 109-95-5
- 109-96-6
- 109966-30-5
- 1099687-03-2
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View