-
Name
Ethylparaben
- EINECS 204-399-4
- CAS No. 120-47-8
- Article Data191
- CAS DataBase
- Density 1.168 g/cm3
- Solubility Soluble in ethanol, ether and acetone, slightly soluble in water
- Melting Point 114-117 °C(lit.)
- Formula C9H10O3
- Boiling Point 297.5 °C at 760 mmHg
- Molecular Weight 166.177
- Flash Point 120.3 °C
- Transport Information
- Appearance white crystalline powder
- Safety 26-36-24/25
- Risk Codes 36/37/38
-
Molecular Structure
-
Hazard Symbols
 Xi
Xi
- Synonyms Benzoicacid, p-hydroxy-, ethyl ester (6CI,8CI);4-(Ethoxycarbonyl)phenol;4-Carbethoxyphenol;4-Hydroxybenzoic acid ethyl ester;Aseptin A;Aseptine A;Aseptoform E;Bonomold OE;E 214;Easeptol;Ethyl 4-hydroxybenzoate;EthylButex;Ethyl nipagin;Ethyl p-hydroxybenzoate;Ethyl parasept;Ethylparaben;Mekkings E;Mycocten;p-(Ethoxycarbonyl)phenol;p-Carbethoxyphenol;p-Hydroxybenzoate ethyl ester;
- PSA 46.53000
- LogP 1.56890
Synthetic route

| Conditions | Yield |
|---|---|
| With sodium; diphenyldisulfane In 1-methyl-pyrrolidin-2-one at 90℃; for 0.5h; | 100% |
| With potassium carbonate In 1-methyl-pyrrolidin-2-one at 100℃; for 3h; | 88% |
| With potassium fluoride; thiophenol In 1-methyl-pyrrolidin-2-one at 100℃; for 1h; | 82% |
-
-
56441-55-5
ethyl 4-(benzyloxy)benzoate

-
-
120-47-8
Ethyl 4-hydroxybenzoate

| Conditions | Yield |
|---|---|
| With palladium diacetate; sodium hydride In N,N-dimethyl acetamide at 50℃; for 5h; Inert atmosphere; | 99% |
| With palladium diacetate; sodium hydride In N,N-dimethyl acetamide at 50℃; for 5h; Inert atmosphere; | 99% |

| Conditions | Yield |
|---|---|
| With ammonium formate; palladium on activated charcoal In methanol for 0.5h; Heating; | 98% |

| Conditions | Yield |
|---|---|
| With ethanol; sulfuric acid at 100℃; for 24h; Neat (no solvent); chemoselective reaction; | 97.22% |

| Conditions | Yield |
|---|---|
| With sulfuric acid for 5h; Reflux; | 97% |
| With sulfuric acid for 24h; Reflux; | 96% |
| With [1-(3-sulfonic acid)]propyl-3-methylimidazolium hydrogen sulfate at 85 - 100℃; for 3h; | 95.8% |
-
-
13031-45-3
ethyl 4-acetoxybenzoate

-
-
120-47-8
Ethyl 4-hydroxybenzoate

| Conditions | Yield |
|---|---|
| With Candida cylindracea lipase In di-isopropyl ether; butan-1-ol at 28 - 30℃; for 14h; | 97% |
| With sodium tetrahydroborate; cobalt(II) chloride In ethanol at 0 - 25℃; for 10h; | 94% |
| With sodium; diphenyldisulfane In 1-methyl-pyrrolidin-2-one at 90℃; for 0.5h; | 87% |
-
-
153025-65-1
ethyl (4-tert-butyldimethylsilyloxyphenyl)glyoxylate

-
-
120-47-8
Ethyl 4-hydroxybenzoate

| Conditions | Yield |
|---|---|
| With potassium hydroxide In ethanol at 20℃; for 1.5h; | 97% |
| With sodium phosphate dodecahydrate In N,N-dimethyl-formamide at 20℃; for 2h; chemoselective reaction; | 86% |
| With copper(ll) sulfate pentahydrate In methanol at 100℃; for 0.25h; Microwave irradiation; |

| Conditions | Yield |
|---|---|
| With dihydrogen peroxide In water at 30℃; Green chemistry; | 97% |
| With sodium chlorite In water at 20℃; for 0.5h; | 90% |
| With 10-methylacridine-3(10H)-one; oxygen; N-ethyl-N,N-diisopropylamine In water at 20℃; for 24h; Reagent/catalyst; Time; Irradiation; Green chemistry; | 88% |
| With water; oxygen; copper dichloride at 60℃; under 760.051 Torr; for 24h; | 67% |
-
-
23676-09-7
ethyl 4-ethoxybenzoate

-
-
120-47-8
Ethyl 4-hydroxybenzoate

| Conditions | Yield |
|---|---|
| With aluminium trichloride In dichloromethane; ethanethiol for 9.5h; Ambient temperature; | 95.5% |
| With aluminium trichloride In dichloromethane; ethanethiol for 9.5h; Ambient temperature; | 95.5% |
| With aluminium trichloride; ethanethiol |

| Conditions | Yield |
|---|---|
| With 4 A molecular sieve; (Ph3P)PdCl2 In tetrahydrofuran at 100℃; under 22800 Torr; for 24h; | 95% |
| bis-triphenylphosphine-palladium(II) chloride In various solvent(s) at 100℃; under 22800 Torr; for 24h; | 99 % Spectr. |


| Conditions | Yield |
|---|---|
| With perchloric acid; sodium percarbonate; vanadia for 3h; Cooling; | 95% |
| With urea-2,2-dihydroperoxypropane; hydrogen bromide; acetic acid at 20℃; for 12h; | 81% |
| With dihydrogen peroxide at 60℃; for 3h; | |
| With dihydrogen peroxide at 70℃; for 24h; | 29 %Chromat. |
-
-
153025-65-1
ethyl (4-tert-butyldimethylsilyloxyphenyl)glyoxylate

-
-
120-47-8
Ethyl 4-hydroxybenzoate

| Conditions | Yield |
|---|---|
| In water; N,N-dimethyl-formamide at 20℃; for 1h; | 95% |

| Conditions | Yield |
|---|---|
| In dichloromethane for 0.5h; Reflux; | 95% |
| With triethylamine at 50℃; Temperature; |

| Conditions | Yield |
|---|---|
| With [(2-di-tert-butylphosphino-3-methoxy-6-methyl-2,4,6-triisopropyl-1,1-biphenyl)-2-(2-aminobiphenyl)]palladium(II) methanesulfonate; caesium carbonate; Benzaldoxime In N,N-dimethyl-formamide at 80℃; for 18h; Inert atmosphere; Glovebox; Sealed tube; | 95% |
| With tris(6,6'-diamino-2,2'-bipyridine); 4,4-diphenyl-1,3,5,7,8-pentamethyl-2,6-diethyl-4-bora-3a,4a-diaza-s-indacene; Br2Ni*3H2O; water; N-ethyl-N,N-diisopropylamine In N,N-dimethyl-formamide; acetonitrile at 20℃; for 24h; Glovebox; Irradiation; Inert atmosphere; | 90% |
| Stage #1: Ethyl 4-bromobenzoate With nickel(II) chloride hexahydrate; water; cadmium(II) sulphide In N,N-dimethyl acetamide for 0.00277778h; Schlenk technique; Sonication; Stage #2: In N,N-dimethyl acetamide at 55℃; for 6h; Schlenk technique; Irradiation; | 87% |

| Conditions | Yield |
|---|---|
| With carbon tetrabromide; triphenylphosphine In 1,2-dichloro-ethane at 40℃; | 94% |

| Conditions | Yield |
|---|---|
| Stage #1: 4-pyrone; diethyl malonate With potassium tert-butylate In tert-butyl alcohol for 3h; Reflux; Stage #2: With hydrogenchloride In water; tert-butyl alcohol for 1h; Reflux; | 94% |
-
-
118827-08-0
ethyl 4-(tetrahydro-2H-pyran-2-yloxy)benzoate

-
-
120-47-8
Ethyl 4-hydroxybenzoate

| Conditions | Yield |
|---|---|
| With CuCl2*H2O In ethanol for 3h; Hydrolysis; Heating; | 92% |
| With titanium(III) chloride In acetonitrile at 20℃; for 2.5h; | 88% |
| With cerium (IV) sulfate tetrahydrate In methanol at 130℃; for 0.333333h; Temperature; Microwave irradiation; | 86% |
| With hydrogen; palladium on activated charcoal In ethanol at 20℃; for 3h; | 81% |

-
-
120-47-8
Ethyl 4-hydroxybenzoate

| Conditions | Yield |
|---|---|
| With hydrogen; palladium on activated charcoal In ethanol at 20℃; for 3h; | 92% |

| Conditions | Yield |
|---|---|
| microwave irradiation; | 91% |
-
-
1085453-61-7
ethyl 4-(triisopropylsilyloxy)benzoate

-
-
120-47-8
Ethyl 4-hydroxybenzoate

| Conditions | Yield |
|---|---|
| With potassium acetate In water; N,N-dimethyl-formamide at 25℃; for 7h; | 89% |
| With copper(ll) sulfate pentahydrate In methanol at 100℃; for 0.25h; Microwave irradiation; |

-
-
120-47-8
Ethyl 4-hydroxybenzoate

| Conditions | Yield |
|---|---|
| With magnesium In methanol at 20℃; | 88% |

| Conditions | Yield |
|---|---|
| With sodium hydrogencarbonate; 3-butyl-1-methyl-1H-imidazol-3-ium hexafluorophosphate at 40℃; for 0.666667h; | 88% |

| Conditions | Yield |
|---|---|
| With cesium fluoride In acetonitrile for 5h; Heating; | 87% |

| Conditions | Yield |
|---|---|
| With acetylhydroxamic acid; potassium carbonate In dimethyl sulfoxide at 80℃; for 16h; Reagent/catalyst; Solvent; Sealed tube; | 87% |

-
-
120-47-8
Ethyl 4-hydroxybenzoate

| Conditions | Yield |
|---|---|
| With magnesium In methanol at 20℃; for 3h; | 86% |
-
-
51934-41-9
4-iodobenzoic acid ethyl ester

-
-
120-47-8
Ethyl 4-hydroxybenzoate

| Conditions | Yield |
|---|---|
| With nickel(II) bromide trihydrate; water; 1-(2,2-diphenyl-2λ4,3λ4-[1,3,2]diazaborolo[4,5,1-ij]quinolin-1(2H)-yl)-3-phenylpropan-1-one; N-ethyl-N,N-diisopropylamine; 4,4'-di-tert-butyl-2,2'-bipyridine In dimethyl sulfoxide at 25℃; for 22h; Irradiation; | 84% |
-
-
109-87-5
Dimethoxymethane

-
-
56441-84-0
ethyl 4-((4-methoxybenzyl)oxy)-benzoate

-
A
-
120-47-8
Ethyl 4-hydroxybenzoate

-
B
-
5942-30-3
ethyl 4-(methoxymethoxy)benzoate

| Conditions | Yield |
|---|---|
| With bromethyl methyl ether; tin(II) bromide In 1,2-dichloro-ethane for 24h; Ambient temperature; | A 80% B 13% |


| Conditions | Yield |
|---|---|
| With copper(I) oxide; sodium methylate In methanol at 185℃; for 12h; Autoclave; | 80% |
| With 2-(diethylamino)ethanethiol hydrochloride; sodium t-butanolate In N,N-dimethyl-formamide for 1h; Heating; | 69% |

| Conditions | Yield |
|---|---|
| With tetrachlorosilane for 11h; Heating; | 79% |

| Conditions | Yield |
|---|---|
| With sodium carbonate; N,N-dimethyl-formamide at 80℃; for 12h; Inert atmosphere; | 100% |
| With potassium carbonate In acetone for 20h; Heating; | |
| With potassium carbonate In butanone Heating; | |
| With potassium carbonate In acetone; toluene | 13.5 gm (61%) |
| With sodium hydroxide |
-
-
120-47-8
Ethyl 4-hydroxybenzoate

-
-
3618-04-0, 75877-66-6, 17159-80-7
4-hydroxycyclohexyl carboxylic acid ethyl ester

| Conditions | Yield |
|---|---|
| With hydrogen; Rh/Al2O3 In ethanol at 20℃; under 3800 Torr; for 20h; Hydrogenation; | 100% |
| With hydrogen; nickel In ethanol at 100℃; under 76000 Torr; for 48h; | 88% |
| With hydrogen; nickel In ethanol at 145 - 150℃; under 83256.7 Torr; for 18h; | 61% |
-
-
120-47-8
Ethyl 4-hydroxybenzoate

-
-
160295-23-8
2-(5-chloropentanoyl)-3-methylthiophene

-
-
160295-29-4
4-[5-(3-Methyl-thiophen-2-yl)-5-oxo-pentyloxy]-benzoic acid ethyl ester

| Conditions | Yield |
|---|---|
| With potassium carbonate; sodium iodide In acetonitrile for 72h; Heating; | 100% |
-
-
120-47-8
Ethyl 4-hydroxybenzoate

-
-
776-74-9
Bromodiphenylmethane

-
-
204840-46-0
ethyl 4-(1,1-diphenylmethoxy)benzoate

| Conditions | Yield |
|---|---|
| With CsCO3 In N,N-dimethyl-formamide at 60℃; for 18h; | 100% |
| Stage #1: Ethyl 4-hydroxybenzoate With sodium ethanolate In N,N-dimethyl-formamide at 0 - 20℃; Stage #2: Bromodiphenylmethane In N,N-dimethyl-formamide at 20℃; Product distribution / selectivity; |

| Conditions | Yield |
|---|---|
| With caesium carbonate In N,N-dimethyl-formamide | 100% |
| With potassium carbonate at 100℃; for 6h; Etherification; | 98% |
| With Aliquat 336; potassium carbonate at 100℃; for 6h; | 98% |
-
-
120-47-8
Ethyl 4-hydroxybenzoate

-
-
106-95-6
allyl bromide

-
-
5443-37-8
ethyl 4-(n-prop-2'-enyloxy)benzoate

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetone for 3h; Heating; | 100% |
| With potassium carbonate In acetone | 99% |
| With potassium carbonate In toluene for 10h; Reflux; | 98% |
-
-
120-47-8
Ethyl 4-hydroxybenzoate

-
-
98-09-9
benzenesulfonyl chloride

-
-
1286990-00-8
ethyl 4-((phenylsulfonyl)oxy)benzoate

| Conditions | Yield |
|---|---|
| With potassium carbonate; tetra(n-butyl)ammonium hydrogensulfate In water; ethyl acetate at 13 - 35℃; for 3h; Reflux; | 100% |
| With tetra(n-butyl)ammonium hydrogensulfate; potassium carbonate In water; ethyl acetate at 13 - 35℃; Reflux; | 100% |
-
-
120-47-8
Ethyl 4-hydroxybenzoate

-
-
100-97-0
hexamethylenetetramine

-
-
329904-04-3
3,5-diformyl-4-hydroxybenzoic acid ethyl ester

| Conditions | Yield |
|---|---|
| Stage #1: Ethyl 4-hydroxybenzoate; hexamethylenetetramine With trifluoroacetic acid Reflux; Stage #2: With water at 20 - 80℃; for 2h; | 100% |
| With trifluoroacetic acid | |
| With trifluoroacetic acid Duff Aldehyde Synthesis; Reflux; | |
| With trifluoroacetic acid Duff Aldehyde Synthesis; |
-
-
120-47-8
Ethyl 4-hydroxybenzoate

-
-
76-05-1
trifluoroacetic acid

-
-
329904-04-3
3,5-diformyl-4-hydroxybenzoic acid ethyl ester

| Conditions | Yield |
|---|---|
| With hexamethylenetetraamine at 80℃; for 2h; Reflux; Inert atmosphere; | 100% |

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide at 65℃; for 24h; | 100% |
| With potassium carbonate In N,N-dimethyl-formamide at 65℃; for 24h; | 100% |
| With potassium carbonate In N,N-dimethyl-formamide at 65℃; for 52h; | 6.13 g |

| Conditions | Yield |
|---|---|
| With Cp2Ti(OSO2C8F17)2 at 20℃; for 1.5h; Neat (no solvent); | 99% |
| With magnesium(II) perchlorate | 92% |
| With magnesium(II) perchlorate | 92% |
-
-
120-47-8
Ethyl 4-hydroxybenzoate

-
-
98-59-9
p-toluenesulfonyl chloride

-
-
98634-20-9
ethyl 4-(tosyloxy)benzoate

| Conditions | Yield |
|---|---|
| With pyridine at 25℃; for 20h; | 99% |
| With potassium carbonate In tetrahydrofuran; water at 0 - 20℃; for 2h; Reagent/catalyst; Green chemistry; | 99% |
| In pyridine at 45℃; for 15h; | 94% |

| Conditions | Yield |
|---|---|
| With potassium carbonate In ethanol at 60℃; for 2h; | 99% |
| With potassium carbonate In ethanol at 60℃; for 2h; | 99% |
| With potassium carbonate In acetone for 72h; Reflux; | 91.8% |
-
-
120-47-8
Ethyl 4-hydroxybenzoate

-
-
112-29-8
1-bromo dodecane

-
-
154845-74-6
4-decyloxy-benzoic acid ethyl ester

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetonitrile Reflux; | 99% |
| With 18-crown-6 ether; potassium carbonate In acetone at 60℃; for 12h; Inert atmosphere; | 97% |
| With potassium carbonate In N,N-dimethyl-formamide at 80℃; for 4h; Inert atmosphere; | 90.3% |
-
-
120-47-8
Ethyl 4-hydroxybenzoate

-
-
124-63-0
methanesulfonyl chloride

-
-
902148-89-4
ethyl 4-(methanesulfonyloxy)benzoate

| Conditions | Yield |
|---|---|
| With triethylamine In ethyl acetate at 0 - 20℃; for 0.166667h; Green chemistry; | 99% |
| With pyridine at 0 - 20℃; |

| Conditions | Yield |
|---|---|
| With Hg; 1,1,1,3,3,3-hexamethyl-disilazane | 99% |
-
-
120-47-8
Ethyl 4-hydroxybenzoate

-
-
13154-24-0
triisopropylsilyl chloride

-
-
1085453-61-7
ethyl 4-(triisopropylsilyloxy)benzoate

| Conditions | Yield |
|---|---|
| With 1H-imidazole at 0 - 20℃; for 16h; | 99% |
| With 1H-imidazole In dichloromethane at 25℃; for 4h; | 91.1% |
-
-
120-47-8
Ethyl 4-hydroxybenzoate

-
-
70419-06-6
(S)-nonan-2-ol

-
-
1241902-61-3
ethyl 4-[(R)-1-methyloctoxy]benzoate

| Conditions | Yield |
|---|---|
| With di-isopropyl azodicarboxylate; triphenylphosphine In tetrahydrofuran at 0℃; for 1h; Mitsunobu coupling; Inert atmosphere; | 99% |

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetonitrile Reflux; | 99% |
-
-
120-47-8
Ethyl 4-hydroxybenzoate

-
-
640723-20-2, 2699-79-8, 12769-73-2
fluorosulfonyl fluoride

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane at 20℃; | 99% |
-
-
120-47-8
Ethyl 4-hydroxybenzoate

| Conditions | Yield |
|---|---|
| With [(4-acetamidophenyl)(fluorosulfonyl)amino]sulfonyl fluoride; 1,8-diazabicyclo[5.4.0]undec-7-ene In tetrahydrofuran at 20℃; for 0.166667h; | 99% |
| Stage #1: Ethyl 4-hydroxybenzoate With triethylamine In dichloromethane Stage #2: With potassium fluoride; N,N`-sulfuryldiimidazole; trifluoroacetic acid In water at 20℃; for 18h; | 97% |
| With fluorosulfonyl fluoride; triethylamine In dichloromethane at 20℃; for 18h; | 95% |
-
-
192132-77-7
2-(2-(tert-butoxycarbonylamino)ethoxy)ethyl 4-methylbenzenesulfonate

-
-
120-47-8
Ethyl 4-hydroxybenzoate

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide at 60℃; for 14h; Inert atmosphere; | 99% |

| Conditions | Yield |
|---|---|
| With triethylamine In acetonitrile at 0℃; for 1h; Inert atmosphere; | 99% |

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetone at 70℃; for 16h; | 99% |
-
-
120-47-8
Ethyl 4-hydroxybenzoate

-
-
59142-68-6
2-bromo-4-fluorobenzaldehyde

-
-
1196474-68-6
ethyl 4-(3-bromo-4-formylphenoxy)benzoate

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide at 100℃; for 17h; Inert atmosphere; | 98.8% |
| With caesium carbonate In N,N-dimethyl-formamide at 80℃; |
-
-
120-47-8
Ethyl 4-hydroxybenzoate

-
-
824-94-2
p-methoxybenzyl chloride

-
-
56441-84-0
ethyl 4-((4-methoxybenzyl)oxy)-benzoate

| Conditions | Yield |
|---|---|
| With potassium carbonate; sodium iodide In N,N-dimethyl-formamide at 50℃; Inert atmosphere; | 98% |
| With sodium hydroxide | |
| With sodium hydride 1.) DMF, RT, 30 min, 2.) DMF, 5 h; Multistep reaction; | |
| With potassium carbonate; N,N-dimethyl-formamide; sodium iodide at 20 - 50℃; Inert atmosphere; | |
| Stage #1: Ethyl 4-hydroxybenzoate With potassium hydroxide In acetonitrile for 0.5h; Reflux; Stage #2: p-methoxybenzyl chloride In acetonitrile for 3h; Reflux; |
-
-
120-47-8
Ethyl 4-hydroxybenzoate

-
-
358-23-6
trifluoromethylsulfonic anhydride

-
-
125261-30-5
p-(ethoxycarbonyl)phenyl triflate

| Conditions | Yield |
|---|---|
| With pyridine In dichloromethane at 0 - 20℃; for 1h; Inert atmosphere; | 98% |
| With pyridine In dichloromethane at 0 - 20℃; for 5h; Schlenk technique; Inert atmosphere; | 94% |
| With pyridine In dichloromethane at 0℃; for 3h; Inert atmosphere; | 94% |
Ethylparaben Chemical Properties
Empirical Formula: C9H10O3
Molecular Weight: 166.1739 g/mol
EINECS: 204-399-4
Index of Refraction: 1.538
Density: 1.168 g/cm3
Flash Point: 120.3 °C
Melting point: 115-118 °C(lit.)
Storage tempreture: 0-6 °C
Appearance: White crystalline powder
Enthalpy of Vaporization: 55.89 kJ/mol
Boiling Point: 297.5 °C at 760 mmHg
Vapour Pressure: 0.000759 mmHg at 25 °C
Solubility: Soluble in ethanol, ether and acetone, slightly soluble in water
Structure of Ethylparaben (CAS NO.120-47-8):
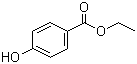
IUPAC Name: Ethyl 4-hydroxybenzoate
Product Category of Ethylparaben (CAS NO.120-47-8): Pharmaceutical Raw Materials;Aromatic Esters
Ethylparaben Uses
Ethylparaben (CAS NO.120-47-8) is used as a food preservative, and is also used in the pharmaceutical, leather and other industries.
Ethylparaben Production
Ethylparaben (CAS NO.120-47-8) can be obtained from the right-hydroxy benzoic acid and ethanol in the presence of sulfuric acid as catalyst for esterification, after that having them in water for crystallization, and then through filter and pickling to get the product.
Ethylparaben Toxicity Data With Reference
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| dog | LD50 | oral | 5gm/kg (5000mg/kg) | FAO Nutrition Meetings Report Series. Vol. 53A, Pg. 81, 1974. | |
| guinea pig | LD50 | oral | 2gm/kg (2000mg/kg) | FAO Nutrition Meetings Report Series. Vol. 53A, Pg. 81, 1974. | |
| mouse | LD50 | intraperitoneal | 520mg/kg (520mg/kg) | Drug Standards. Vol. 20, Pg. 89, 1952. | |
| mouse | LD50 | oral | 3gm/kg (3000mg/kg) | Bromatologia i Chemia Toksykologiczna. Vol. 14, Pg. 301, 1981. | |
| rabbit | LD50 | oral | 5gm/kg (5000mg/kg) | FAO Nutrition Meetings Report Series. Vol. 53A, Pg. 81, 1974. | |
| rat | LD | oral | > 200mg/kg (200mg/kg) | Acta Obstetrica et Gynaecologica Japonica, English Edition. Vol. 22, Pg. 94, 1975. |
Ethylparaben Safety Profile
Hazard Codes:  Xi
Xi
Risk Statements: 36/37/38
R36/37/38:Irritating to eyes, respiratory system and skin.
Safety Statements: 26-36-24/25
S24/25:Avoid contact with skin and eyes.
S26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice.
S36:Wear suitable protective clothing.
Ethylparaben Specification
Ethylparaben , its cas register number is 120-47-8. It also can be called Ethyl paraben ; 4-(Ethoxycarbonyl)phenol ; Ethyl 4-hydroxybenzoate ; p-Hydroxybenzoate ethyl ester ; Ethyl butex ; 4-Hydroxybenzoic acid ethyl ester ; and Ethyl p-hydroxybenzoate . It is hazardous, so the first aid measures and others should be known. Such as: When on the skin: first, should flush skin with plenty of water immediately for at least 15 minutes while removing contaminated clothing. Secondly, get medical aid. Or in the eyes: Flush eyes with plenty of water for at least 15 minutes, occasionally lifting the upper and lower eyelids. Then get medical aid soon. While, it's inhaled: Remove from exposure and move to fresh air immediately. Give artificial respiration while not breathing. When breathing is difficult, give oxygen. And as soon as to get medical aid. Then you have the ingesting of the product: Wash mouth out with water, and get medical aid immediately. Notes to physician: Treat supportively and symptomatically.
In addition, Ethylparaben (CAS NO.120-47-8) is not compatible with strong oxidizing agents, and you must not take it with incompatible materials. And also prevent it to broken down into hazardous decomposition products: carbon monoxide, carbon dioxide.
Related Products
- Ethylparaben
- 1204-78-0
- 120478-49-1
- 120482-96-4
- 120484-50-6
- 12048-51-0
- 1204-85-9
- 1204-86-0
- 120486-24-0
- 120496-69-7
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View