-
Name
LAPACHOL
- EINECS 201-563-7
- CAS No. 84-79-7
- Article Data37
- CAS DataBase
- Density 1.235 g/cm3
- Solubility
- Melting Point 141-143 ºC(lit.)
- Formula C15H14 O3
- Boiling Point 398.6 ºC at 760 mmHg
- Molecular Weight 242.274
- Flash Point 209 ºC
- Transport Information
- Appearance white to yellow powder
- Safety 26-36
- Risk Codes 20/21/22-36/37/38
-
Molecular Structure
-
Hazard Symbols
 Xn
Xn
- Synonyms 1,4-Naphthalenedione,2-hydroxy-3-(3-methyl-2-butenyl)- (9CI); 1,4-Naphthoquinone,2-hydroxy-3-(3-methyl-2-butenyl)- (7CI,8CI); Lapachol (6CI);2-Hydroxy-3-(3-methyl-2-butenyl)-1,4-naphthalenedione;2-Hydroxy-3-(3-methyl-2-butenyl)-1,4-naphthoquinone; Bethabarra Wood; C.I.75490; C.I. Natural Yellow 16; Greenhartin; Ipe-tobacco Wood; Lapachol Wood;NSC 11905; NSC 629756; Surinam Greenheart Wood; Taigu Wood; Taiguic acid;Tecomin
- PSA 54.37000
- LogP 3.23400
Synthetic route

| Conditions | Yield |
|---|---|
| With formic acid In ethanol; water at 200℃; under 155.149 - 155149 Torr; for 3h; chemoselective reaction; | 78% |
| With formic acid In ethanol; water at 200℃; for 3h; | 78% |

| Conditions | Yield |
|---|---|
| With oxalic acid In 1,4-dioxane at 100℃; for 6h; | 78% |
-
-
83-72-7
2-Hydroxy-1,4-naphthoquinone

-
-
870-63-3
prenyl bromide

-
A
-
84-79-7
Lapachol

-
B
-
64469-16-5
2-(1,1-dimethylprop-2-enyl)-3-hydroxy-naphthalene-1.4-dione

| Conditions | Yield |
|---|---|
| With tetrakis(triphenylphosphine) palladium(0); triethylamine In 1,4-dioxane at 20℃; for 4h; Heck Reaction; | A 61% B 16% |


| Conditions | Yield |
|---|---|
| With triethylamine; sodium iodide In dimethyl sulfoxide at 20 - 70℃; for 7h; Inert atmosphere; | 60% |
| With tetrakis(triphenylphosphine) palladium(0); triethylamine In 1,4-dioxane at 20℃; for 4h; Heck Reaction; | 23% |
| With lead(II) bromide; aluminium In acetonitrile for 15h; | 20% |
-
-
83-72-7
2-Hydroxy-1,4-naphthoquinone

-
-
870-63-3
prenyl bromide

-
A
-
42164-69-2
Lawsone 2-isopentenyl ether

-
B
-
84-79-7
Lapachol

| Conditions | Yield |
|---|---|
| With triethylamine; sodium iodide In N,N-dimethyl-formamide at 40℃; for 72h; | A 4.8% B 58.2% |
| With lithium hydride In dimethyl sulfoxide 45 deg C, 5 h; 25 deg C, 10 h; | A 30% B 40% |
| With zinc In tetrahydrofuran at 55 - 60℃; for 3h; Further byproducts given; | A 0.2 B 150 mg |


| Conditions | Yield |
|---|---|
| tetrakis(triphenylphosphine) palladium(0); 1-Adamantanecarboxylic acid for 1h; Heating; | 43% |
-
-
83-72-7
2-Hydroxy-1,4-naphthoquinone

-
-
563-45-1
3-Methyl-1-butene

-
A
-
84-79-7
Lapachol

-
B
-
4042-39-1
2-hydroxy-3-(3-methylbut-1-en-1-yl)naphthalene-1,4-dione

| Conditions | Yield |
|---|---|
| With copper diacetate; sodium acetate; palladium diacetate In tetrahydrofuran at 20℃; for 3h; Mechanism; Catalytic behavior; | A 20% B 33% |

-
-
36417-25-1
2-hydroxy-1,4-naphthoquinone silver salt

-
-
870-63-3
prenyl bromide

-
A
-
42164-69-2
Lawsone 2-isopentenyl ether

-
B
-
84-79-7
Lapachol


| Conditions | Yield |
|---|---|
| With 1,4-dioxane; oxalic acid und Behandeln des Reaktionsgemisches mit Luft; |
-
-
57620-99-2
2-acetyllapachol

-
-
84-79-7
Lapachol

| Conditions | Yield |
|---|---|
| With sodium carbonate |
-
-
17241-45-1
2-methoxylapachol

-
-
84-79-7
Lapachol

| Conditions | Yield |
|---|---|
| With sodium carbonate for 0.833333h; Heating; | 45 mg |
-
-
82214-83-3
2-chloro-3-(3-methylbut-2-enyl)naphthalene-1,4-dione

-
-
84-79-7
Lapachol

| Conditions | Yield |
|---|---|
| With sodium hydroxide for 0.416667h; Heating; | 30 mg |
-
-
79060-57-4
3-Bromo-α-lapachone

-
-
84-79-7
Lapachol

| Conditions | Yield |
|---|---|
| With acetic acid; zinc for 2h; Heating; | 10 mg |
-
-
79060-56-3
4-Bromo-iso-α-lapachone

-
-
84-79-7
Lapachol

| Conditions | Yield |
|---|---|
| With acetic acid; zinc for 2h; Heating; |
-
-
74693-34-8
4'-bromo-iso-β-lapachone

-
-
84-79-7
Lapachol

| Conditions | Yield |
|---|---|
| With zinc In acetic acid for 2h; Heating; | 40 mg |
-
-
96964-75-9
3-(2-methyl-2-buten-4-yl)-1,2-naphthalenedione

-
-
84-79-7
Lapachol

| Conditions | Yield |
|---|---|
| With sodium dithionite; potassium tert-butylate; oxygen 1.) benzene, 2 min, 2.) t-BuOH, 5 min.; Yield given. Multistep reaction; |
-
-
19336-09-5
1,2,4-triacetoxy-3-(3-methyl-but-2-enyl)-naphthalene

-
-
84-79-7
Lapachol

| Conditions | Yield |
|---|---|
| With sodium hydroxide In 1,4-dioxane |


-
-
84-79-7
Lapachol

| Conditions | Yield |
|---|---|
| With zinc |

-
-
84-79-7
Lapachol

| Conditions | Yield |
|---|---|
| With potassium hydroxide |

| Conditions | Yield |
|---|---|
| With acetone |


| Conditions | Yield |
|---|---|
| With diethyl ether at 0℃; |


| Conditions | Yield |
|---|---|
| With 1,4-dioxane; oxalic acid und Behandeln des Reaktionsgemisches mit Luft; |

-
-
111479-51-7
2-(3-Chloro-3-methylbutyl)-3-hydroxy-1,4-naphthoquinone

-
A
-
4707-32-8
2,2-dimethyl-3,4-dihydro-2H-benzo[h]chromene-5,6-dione

-
B
-
4707-33-9
α-lapachone

-
C
-
84-79-7
Lapachol

-
-
83-72-7
2-Hydroxy-1,4-naphthoquinone

-
-
115-18-4
2-methyl-3-buten-2-ol

-
A
-
84-79-7
Lapachol

-
B
-
64469-16-5
2-(1,1-dimethylprop-2-enyl)-3-hydroxy-naphthalene-1.4-dione

| Conditions | Yield |
|---|---|
| tetrakis(triphenylphosphine) palladium(0); 1-Adamantanecarboxylic acid Heating; Title compound not separated from byproducts.; |


| Conditions | Yield |
|---|---|
| With β-cellulase; water at 37℃; for 48h; Enzymatic reaction; | A n/a B 25.2 mg |
-
-
96964-74-8
2-hydroxy-3-(2-methyl-2-buten-4-yl)naphthalene

-
-
84-79-7
Lapachol

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 76 percent / CuCl, O2 / acetonitrile / 0.33 h / 5 - 10 °C 2: 1.) aq. Na2S2O4, 2.) t-BuOK, O2 / 1.) benzene, 2 min, 2.) t-BuOH, 5 min. View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: K2CO3 / acetone / 40 h / Heating 2: 70 mg / zinc dust / tetrahydrofuran / 24 h 3: 45 mg / aq.Na2CO3 / 0.83 h / Heating View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 300 mg / zinc dust / tetrahydrofuran / 4 h 2: 30 mg / 1percent aq.NaOH / 0.42 h / Heating View Scheme |

| Conditions | Yield |
|---|---|
| With sulfuric acid at 0 - 20℃; | 100% |
| With sulfuric acid at 0 - 20℃; | 100% |
| With sulfuric acid | 99% |

| Conditions | Yield |
|---|---|
| With lutidine In dichloromethane at 0℃; for 0.0833333h; | 100% |
| With lutidine In dichloromethane at 0℃; | 100% |
| With triethylamine In diethyl ether at 20℃; for 0.5h; |
-
-
84-79-7
Lapachol

| Conditions | Yield |
|---|---|
| With 10% palladium on activated carbon; hydrogen; acetic acid under 2844.39 Torr; for 6h; | 100% |
| With palladium 10% on activated carbon; hydrogen; acetic acid under 2844.39 Torr; for 6h; |

| Conditions | Yield |
|---|---|
| With lutidine In dichloromethane at 0℃; for 0.0833333h; | 99.7% |
| With lutidine In dichloromethane at 0℃; | 99% |
| With 2,6-dimethylpyridine In dichloromethane at 0 - 20℃; for 3h; Inert atmosphere; | 80% |
| With triethylamine In diethyl ether at 20℃; for 0.5h; |
-
-
84-79-7
Lapachol

-
-
33834-56-9
di(4-bromophenyl) selenide

| Conditions | Yield |
|---|---|
| With iodine; dimethyl sulfoxide at 50℃; for 0.166667h; Microwave irradiation; Sealed tube; Green chemistry; | 99% |

| Conditions | Yield |
|---|---|
| With hydrogen; acetic acid; palladium on activated charcoal under 3750.3 Torr; for 2h; Heating; | 98% |
| With 10% palladium on activated carbon; hydrogen In ethanol under 1551.49 Torr; for 0.25h; | 90% |
| With 5%-palladium/activated carbon; hydrogen In ethyl acetate at 20℃; for 12h; | 83% |
-
-
243643-58-5
mer-[(1,4-bis(diphenylphosphino)butane)aquatrichlororuthenium(III)]

-
-
84-79-7
Lapachol

-
-
1615718-36-9
RuCl2(lapachol)(1,4-bis(diphenylphosphine)butane)

| Conditions | Yield |
|---|---|
| With triethylamine In methanol; dichloromethane for 24h; Reflux; Inert atmosphere; | 98% |

-
-
84-79-7
Lapachol

-
-
1429209-71-1
chlorido[(3-(3-methylbut-2-enyl)-2-oxo-κO)-[1,4]-naphthoquinonato-κO](η6-pentamethylcyclopentadienyl)rhodium(III)

| Conditions | Yield |
|---|---|
| With sodium methylate In methanol; dichloromethane at 20℃; for 24h; Inert atmosphere; | 96% |

| Conditions | Yield |
|---|---|
| With lutidine In dichloromethane at 0℃; for 0.0833333h; | 95.7% |

| Conditions | Yield |
|---|---|
| With hydrogenchloride; acetic acid | 95% |
| With hydrogenchloride; acetic acid In water | 95% |
| With hydrogenchloride In water at 80℃; for 4h; | 85% |
-
-
84-79-7
Lapachol

-
-
41019-50-5
3'-bromo-β-lapachone

| Conditions | Yield |
|---|---|
| With bromine In dichloromethane for 24h; | 95% |
| With bromine; acetic acid Behandeln des Reaktionsprodukts mit Wasser; | |
| With chloroform; bromine Behandeln des Reaktionsprodukts mit Alkohol; |

| Conditions | Yield |
|---|---|
| With sodium acetate; acetic acid at 90 - 95℃; | 95% |

| Conditions | Yield |
|---|---|
| With potassium hydroxide at 0℃; Inert atmosphere; | 95% |
-
-
84-79-7
Lapachol

| Conditions | Yield |
|---|---|
| With selenium(IV) oxide In 1,4-dioxane Reflux; | 95% |

| Conditions | Yield |
|---|---|
| With triethylamine In water at 20℃; for 48h; | 95% |

| Conditions | Yield |
|---|---|
| Stage #1: isoniazid; Lapachol With triethylamine In water at 20℃; for 48h; Stage #2: With acetic acid In water | 95% |
-
-
159700-02-4, 21627-33-8
cis-[RuCl2(triphenylphospine)2(1,10-phenantroline)]

-
-
84-79-7
Lapachol

| Conditions | Yield |
|---|---|
| With triethylamine In methanol; dichloromethane for 72h; Reflux; Inert atmosphere; | 94% |

-
-
14835-66-6
Di-n-butyl selenide

-
-
84-79-7
Lapachol

| Conditions | Yield |
|---|---|
| With iodine; dimethyl sulfoxide at 50℃; for 0.166667h; Microwave irradiation; Sealed tube; Green chemistry; | 94% |
-
-
84-79-7
Lapachol

-
-
124-63-0
methanesulfonyl chloride

-
-
1376687-04-5
3-(3-methylbut-2-enyl)-1,4-dioxo-1,4-dihydronaphthalen-2-yl methanesulfonate

| Conditions | Yield |
|---|---|
| With potassium carbonate In dichloromethane at 0 - 20℃; for 48h; | 93.7% |

| Conditions | Yield |
|---|---|
| With lutidine In dichloromethane at 0℃; | 93% |

| Conditions | Yield |
|---|---|
| With tert-butylammonium hexafluorophosphate(V) In acetonitrile at 20℃; for 1h; Reagent/catalyst; Solvent; Time; Electrolysis; Green chemistry; | 93% |
| With iodine; dimethyl sulfoxide In neat (no solvent) at 50℃; for 0.166667h; Microwave irradiation; Sealed tube; Green chemistry; regioselective reaction; | 65% |
| With iodine; dimethyl sulfoxide at 50℃; for 0.166667h; Microwave irradiation; |

| Conditions | Yield |
|---|---|
| Stage #1: Lapachol With sodium hydride In N,N-dimethyl-formamide at 20℃; for 0.25h; Stage #2: 4,7-dichloroquinoline In N,N-dimethyl-formamide at 120℃; for 24h; | 93% |
-
-
52462-29-0
[ruthenium(II)(η6-1-methyl-4-isopropyl-benzene)(chloride)(μ-chloride)]2

-
-
84-79-7
Lapachol

-
-
1429209-69-7
chlorido[(3-(3-methylbut-2-enyl)-2-oxo-κO)-[1,4]-naphthoquinonato-κO](η6-p-cymene)ruthenium(II)

| Conditions | Yield |
|---|---|
| With sodium methylate In methanol; dichloromethane at 20℃; for 4.5h; Inert atmosphere; | 92% |

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetone at 20℃; for 3h; | 91% |
| With potassium carbonate In acetone at 20℃; for 2h; | 88% |
| With sodium hydroxide at 70 - 80℃; for 1h; | 139 mg |
| With potassium carbonate In acetone | |
| With potassium carbonate In acetone at 20℃; |
-
-
84-79-7
Lapachol

| Conditions | Yield |
|---|---|
| With sodium hydroxide; hydroxylamine hydrochloride for 2h; | 91% |

| Conditions | Yield |
|---|---|
| With tert-butylammonium hexafluorophosphate(V) In acetonitrile at 20℃; for 1h; Electrolysis; Green chemistry; | 91% |
-
-
84-79-7
Lapachol

-
-
855637-05-7
2,3-dihydro-3-[(3-methylbut-2-enyl)-1,4-naphthoquinon-2-yloxy]-3-(3-methylbut-2-enyl)-2-oxo-1,4-naphthoquinone

| Conditions | Yield |
|---|---|
| With lead dioxide In acetic acid Heating; | 90% |
| With lead dioxide; acetic acid | 90% |
| With lead dioxide; acetic acid Heating; |

| Conditions | Yield |
|---|---|
| With tert.-butylhydroperoxide; selenium(IV) oxide; salicylic acid In dichloromethane at 0 - 20℃; for 2h; | 90% |
| Multi-step reaction with 2 steps 1: selenium(IV) oxide / 1,4-dioxane / Reflux 2: cerium(III) chloride heptahydrate; sodium tetrahydroborate / methanol / 1 h / 0 - 20 °C View Scheme |

| Conditions | Yield |
|---|---|
| With zinc(II) chloride In dichloromethane at 42℃; for 4h; Reagent/catalyst; Temperature; | A n/a B 90% |

| Conditions | Yield |
|---|---|
| Stage #1: Lapachol With potassium hydroxide In methanol for 1h; Stage #2: 1,10-Phenanthroline; cobalt(II) acetate dihydrate In methanol at 20℃; for 3h; | 89.3% |

Lapachol Chemical Properties
Molecular Structure of Lapachol (CAS NO.84-79-7):
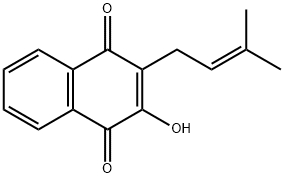
EINECS: 201-563-7
IUPAC Name: 4-Hydroxy-3-(3-methylbut-2-enyl)naphthalene-1,2-dione
Molecular Formula: C15H14O3
Molecular Weight: 242.269860 g/mol
XLogP3-AA: 2.8
H-Bond Donor: 1
H-Bond Acceptor: 3
Canonical SMILES: CC(=CCC1=C(C2=CC=CC=C2C(=O)C1=O)O)C
InChI: InChI=1S/C15H14O3/c1-9(2)7-8-12-13(16)10-5-3-4-6-11(10)14(17)15(12)18/h3-7,16H,8H2,1-2H3
InChIKey: CWPGNVFCJOPXFB-UHFFFAOYSA-N
Index of Refraction: 1.606
Molar Refractivity: 67.61 cm3
Molar Volume: 196 cm3
Surface Tension: 51.1 dyne/cm
Density: 1.235 g/cm3
Flash Point: 203.9 °C
Enthalpy of Vaporization: 67.46 kJ/mol
Boiling Point: 390.1 °C at 760 mmHg
Vapour Pressure: 8.73E-07 mmHg at 25 °C
Melting Point: 141-143 °C(lit.)
Water Solubility: 55.6 mg/L at 25 °C
BRN: 2051889
Lapachol Toxicity Data With Reference
| 1. | orl-rat LDLo:1200 mg/kg | TXAPA9 Toxicology and Applied Pharmacology. 17 (1970),1. | ||
| 2. | orl-mus LD50:487 mg/kg | TXAPA9 Toxicology and Applied Pharmacology. 17 (1970),1. | ||
| 3. | ipr-mus LD50:400 mg/kg | JMCMAR Journal of Medicinal Chemistry. 26 (1983),570. |
Lapachol Safety Profile
Safety Information of Lapachol (CAS NO.84-79-7):
Hazard Codes: Xn 
Risk Statements: 20/21/22-36/37/38
R20/21/22:Harmful by inhalation, in contact with skin and if swallowed
R36/37/38:Irritating to eyes, respiratory system and skin
Safety Statements: 26-36
S26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice
S36:Wear suitable protective clothing
WGK Germany: 3
RTECS: QL8750000
Poison by intraperitoneal route. Moderately toxic by ingestion. When heated to decomposition it emits acrid smoke and fumes.
Lapachol Specification
Lapachol with CAS registry number of 84-79-7 is also known as 2-Hydroxy-3-(3-methyl-2-butenyl)-1,4-naphthalenedione ; 2-Hydroxy-3-(3-methyl-2-butenyl)-1,4-naphthoquinone ; 4-08-00-02487 (Beilstein Handbook Reference) ; Bethabarra wood ; C.I. 75490 ; C.I. Natural Yellow 16 ; CCRIS 745 ; Greenharten ; Greenhartin ; IPE-tobacco wood ; Lapachic acid ; Lapachol wood ; Surinam greenheart wood ; Taigu wood ; Taiguic acid ; Tecomin ; Tecomin (VAN) ; Zlut prirodni 16 ; Zlut prirodni 16 [Czech] ; 1,4-Naphthalenedione, 2-hydroxy-3-(3-methyl-2-butenyl)- (9CI) ; 1,4-Naphthoquinone, 2-hydroxy-3-(3-methyl-2-butenyl)- . It can be isolated from the lapacho tree.
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View