-
Name
Methyl 2-bromohexanoate
- EINECS 226-643-9
- CAS No. 5445-19-2
- Article Data16
- CAS DataBase
- Density 1.297 g/cm3
- Solubility
- Melting Point < -10ºC
- Formula C7H13BrO2
- Boiling Point 195.9 °C at 760 mmHg
- Molecular Weight 209.083
- Flash Point 80.6 °C
- Transport Information
- Appearance Clear, colourless liquid
- Safety 26-36
- Risk Codes 36/37/38
-
Molecular Structure
-
Hazard Symbols
 Xi
Xi
- Synonyms 2-Bromohexanoic acid methyl ester;Methyl 2-bromocaproate;Methyl a-bromocaproate;NSC 21976;
- PSA 26.30000
- LogP 2.11310
Synthetic route

| Conditions | Yield |
|---|---|
| Stage #1: hexanal With ammonium cerium (IV) nitrate; lithium bromide In neat liquid Stage #2: methanol In neat liquid at 35 - 40℃; for 3.5h; | A 80% B 10 %Chromat. |

-
-
73621-73-5
methyl 2-acetyl-2-bromohexanoate

-
-
5445-19-2
methyl 2-bromohexanoate

| Conditions | Yield |
|---|---|
| With lithium perchlorate; triethylamine In diethyl ether for 5h; Ambient temperature; | 72% |
-
-
32798-42-8
methyl α-butylacetoacetate

-
-
5445-19-2
methyl 2-bromohexanoate

| Conditions | Yield |
|---|---|
| With sodium acetate; sodium bromide In methanol at 15℃; Electrochemical reaction; | 70% |
| Multi-step reaction with 2 steps 1: MgBr2, H2O2 2: 72 percent / LiClO4, Et3N / diethyl ether / 5 h / Ambient temperature View Scheme |
-
-
42768-46-7
2-bromohexanoyl chloride

-
-
5445-19-2
methyl 2-bromohexanoate

| Conditions | Yield |
|---|---|
| With triethylamine; mercury In methanol; benzene | 69.7% |

| Conditions | Yield |
|---|---|
| Stage #1: methyl hexanoate With n-butyllithium; diisopropylamine In tetrahydrofuran; hexane at -78 - 0℃; for 0.583333h; Stage #2: With N-Bromosuccinimide In tetrahydrofuran at -78 - 20℃; | 51% |
-
-
186581-53-3, 908094-01-9
diazomethane

-
-
616-05-7
2-bromohexanoic acid

-
-
5445-19-2
methyl 2-bromohexanoate

| Conditions | Yield |
|---|---|
| With diethyl ether |

| Conditions | Yield |
|---|---|
| (i) (bromination), (ii) /BRN= 1098229/; Multistep reaction; |
-
-
106-70-7
methyl hexanoate

-
A
-
5445-19-2
methyl 2-bromohexanoate

-
B
-
41796-82-1
methyl 5-bromohexanoate

-
C
-
78019-66-6
Methyl 4-bromohexanoate

| Conditions | Yield |
|---|---|
| With N-hydroxyphthalimide; bromine; nitric acid; copper diacetate In acetic acid at 80℃; for 5h; Title compound not separated from byproducts.; |

-
-
106-70-7
methyl hexanoate

-
A
-
5445-19-2
methyl 2-bromohexanoate

-
B
-
14273-90-6
methyl 6-bromohexanoate

-
C
-
41796-82-1
methyl 5-bromohexanoate

-
D
-
78019-66-6
Methyl 4-bromohexanoate

| Conditions | Yield |
|---|---|
| With N-bromo-N-(t-butyl)-3,5-bis(trifluoromethyl)benzamide In benzene at 20℃; for 4h; Reagent/catalyst; Sealed tube; Irradiation; Inert atmosphere; Overall yield = 56.1 %Chromat.; |


| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid at 65℃; for 12h; |
-
-
5445-19-2
methyl 2-bromohexanoate

-
-
81629-62-1
α-Azidohexansaeure-methylester

| Conditions | Yield |
|---|---|
| With sodium azide; Aliquat 336 In water at 60℃; for 14h; | 99% |
-
-
5445-19-2
methyl 2-bromohexanoate

-
-
90-02-8
salicylaldehyde

-
-
138320-26-0
methyl 2-(2-formylphenoxy)hexanoate

| Conditions | Yield |
|---|---|
| Stage #1: salicylaldehyde With potassium carbonate In N,N-dimethyl-formamide; toluene at 60 - 70℃; for 0.5h; Stage #2: methyl 2-bromohexanoate In N,N-dimethyl-formamide; toluene at 80 - 100℃; for 2h; | 99% |
| With potassium carbonate; potassium iodide In DMF (N,N-dimethyl-formamide) at 80℃; for 4h; Product distribution / selectivity; | 98.6% |
| With potassium carbonate In DMF (N,N-dimethyl-formamide) at 80℃; for 4h; Product distribution / selectivity; | 98.9% |

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetonitrile for 16h; Heating / reflux; | 99% |
-
-
5445-19-2
methyl 2-bromohexanoate

-
-
38622-91-2, 36635-61-7
[(p-methylphenyl)sulfonylmethyl]isonitrile

-
-
71512-23-7
methyl 2-tosylhexanoate

| Conditions | Yield |
|---|---|
| With water; copper(II) bis(trifluoromethanesulfonate); caesium carbonate In 1,4-dioxane at 70℃; for 9h; Schlenk technique; Inert atmosphere; | 98% |

| Conditions | Yield |
|---|---|
| With 1-methyl-1H-imidazole; titanium tetrachloride; triphenylphosphine In dichloromethane at -50 - -45℃; Ti-crossed Claisen condensation; | 96% |

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide at 92 - 94℃; for 4h; Williamson synthesis; | 96% |
-
-
5445-19-2
methyl 2-bromohexanoate

-
-
1404435-50-2
7-hydroxy-6-iodo-2,2-dimethyl-4H-benzo[d][1,3]dioxin-4-one

| Conditions | Yield |
|---|---|
| With potassium carbonate In dimethyl sulfoxide at 20℃; | 96% |

| Conditions | Yield |
|---|---|
| With sodium hydroxide In ethanol | 95% |
| With sodium hydroxide In ethanol for 18h; | 95% |
-
-
5445-19-2
methyl 2-bromohexanoate

-
-
42806-90-6
2-(4-hydroxyphenoxy)-2-methyl propionic acid ethyl ester

-
-
860262-39-1
C19H28O6

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetonitrile Heating / reflux; | 95% |

| Conditions | Yield |
|---|---|
| With 1-methyl-1H-imidazole; titanium tetrachloride; triphenylphosphine In dichloromethane at -50 - -45℃; Ti-crossed Claisen condensation; | 94% |
-
-
5445-19-2
methyl 2-bromohexanoate

-
-
635-93-8
5-chlorosalicyclaldehyde

-
-
1323367-31-2
methyl 2-(4-chloro-2-formyl-phenoxy)hexanoate

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide at 20℃; for 2h; Inert atmosphere; | 94% |
-
-
38460-95-6
undec-10-enoyl chloride

-
-
5445-19-2
methyl 2-bromohexanoate

| Conditions | Yield |
|---|---|
| With 1-methyl-1H-imidazole; titanium tetrachloride; triphenylphosphine In dichloromethane at -50 - -45℃; Ti-crossed Claisen condensation; | 93% |

| Conditions | Yield |
|---|---|
| With 1-methyl-1H-imidazole; titanium tetrachloride; triphenylphosphine In dichloromethane at -50 - -45℃; Ti-crossed Claisen condensation; | 93% |
-
-
5445-19-2
methyl 2-bromohexanoate

-
-
845790-87-6
4-fluoro-3-methyl benzenethiol

-
-
1043450-31-2
2-(4-fluoro-3-methyl-phenylsulfanyl)-hexanoic acid methyl ester

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane at 20℃; for 46h; | 92% |
-
-
5445-19-2
methyl 2-bromohexanoate

-
-
41624-92-4
methyl 8-chloro-8-oxooctanoate

| Conditions | Yield |
|---|---|
| With 1-methyl-1H-imidazole; titanium tetrachloride; triphenylphosphine In dichloromethane at -50 - -45℃; Ti-crossed Claisen condensation; | 91% |

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide at 92 - 94℃; for 4h; Williamson synthesis; | 91% |
-
-
5445-19-2
methyl 2-bromohexanoate

-
-
1393481-89-4
1-(phenylethynyl)-2-(vinyloxy)-benzene

| Conditions | Yield |
|---|---|
| With tert.-butylhydroperoxide; tetrakis(actonitrile)copper(I) hexafluorophosphate; silver carbonate In N,N-dimethyl acetamide at 120℃; for 24h; Schlenk technique; Inert atmosphere; | 88% |
-
-
5445-19-2
methyl 2-bromohexanoate

-
-
97-51-8
5-Nitrosalicylaldehyde

-
-
335153-23-6
methyl 2-(2-formyl-4-nitrophenoxy)hexanoate

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide at 92 - 94℃; for 3.5h; | 87% |
| With potassium carbonate In N,N-dimethyl-formamide |
-
-
530-48-3
1,1-Diphenylethylene

-
-
5445-19-2
methyl 2-bromohexanoate

-
-
1380341-61-6
methyl 2-(2,2-diphenylvinyl)hexanoate

| Conditions | Yield |
|---|---|
| With potassium phosphate; tetrakis(triphenylphosphine)nickel(0); 1,3-bis-(diphenylphosphino)propane In toluene at 100℃; for 16h; Heck type reaction; Inert atmosphere; | 87% |
-
-
5445-19-2
methyl 2-bromohexanoate

-
-
1310063-64-9
9-isopropyl-6-phenyl-9H-purine

| Conditions | Yield |
|---|---|
| With [Ru(O2CAd)2(p-cymene)]; potassium carbonate; triphenylphosphine In 1,4-dioxane at 40℃; for 20h; | 87% |
| With potassium phosphate; {bis(triphenylphosphine)ruthenium diacetate} In 1,4-dioxane at 100℃; for 18h; Inert atmosphere; Schlenk technique; chemoselective reaction; | 60% |

| Conditions | Yield |
|---|---|
| With Ru(mesitylCO2)2(p-cymene); potassium carbonate; triphenylphosphine In 1,4-dioxane at 60℃; for 20h; | 86% |
-
-
5445-19-2
methyl 2-bromohexanoate

-
-
126775-18-6
4-(2-methoxybenzyl)-6-methylpyridazin-3(2H)-one

| Conditions | Yield |
|---|---|
| With sodium hydride In tetrahydrofuran; mineral oil at 50℃; for 5h; | 86% |
-
-
5445-19-2
methyl 2-bromohexanoate

-
-
17028-61-4
5-nitro-o-vanilline

-
-
959416-85-4
methyl 2-(2-formyl-6-methoxy-4-nitrophenoxy)hexanoate

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide at 92 - 94℃; for 4h; | 85% |
| With potassium carbonate In N,N-dimethyl-formamide at 93 - 95℃; for 4h; |

| Conditions | Yield |
|---|---|
| With [Ru(O2CAd)2(p-cymene)]; potassium carbonate; triphenylphosphine In 1,4-dioxane at 40℃; for 20h; | 85% |

| Conditions | Yield |
|---|---|
| With [Ru(O2CAd)2(p-cymene)]; potassium carbonate; triphenylphosphine In 1,4-dioxane at 60℃; for 20h; | 84% |
-
-
5445-19-2
methyl 2-bromohexanoate

-
-
1431563-05-1
methyl 2-(2-(phenylethynyl)benzyl)acrylate

| Conditions | Yield |
|---|---|
| With tris[2-phenylpyridinato-C2,N]iridium(III); sodium carbonate In acetonitrile at 20℃; for 20h; Inert atmosphere; Photolysis; | 83% |

| Conditions | Yield |
|---|---|
| With potassium phosphate; tetrakis(triphenylphosphine)nickel(0) In toluene at 80℃; for 16h; | 82% |

| Conditions | Yield |
|---|---|
| With [Ru(O2CAd)2(p-cymene)]; potassium carbonate; triphenylphosphine In 1,4-dioxane at 40℃; for 20h; | 82% |

| Conditions | Yield |
|---|---|
| With tris(bipyridine)ruthenium(II) dichloride hexahydrate; potassium carbonate; copper(l) chloride In acetonitrile at 20℃; for 2h; Inert atmosphere; Irradiation; Schlenk technique; | 82% |

-
-
5445-19-2
methyl 2-bromohexanoate

-
-
122-52-1
triethyl phosphite

-
-
94014-69-4
methyl 2-diethylphosphonohexanoate

| Conditions | Yield |
|---|---|
| at 150℃; for 16h; | 81% |
Methyl 2-bromohexanoate Specification
The Methyl 2-bromohexanoate, with the CAS registry number 5445-19-2 and EINECS registry number 226-643-9, is also called hexanoic acid, 2-bromo-, methyl ester. It belongs to the product category of Acid based bromo compounds. And the molecular formula of this chemical is C7H13BrO2.
The physical properties of Methyl 2-bromohexanoate are as following: (1)ACD/LogP: 2.78; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): 2.78; (4)ACD/LogD (pH 7.4): 2.78; (5)ACD/BCF (pH 5.5): 76.31; (6)ACD/BCF (pH 7.4): 76.31; (7)ACD/KOC (pH 5.5): 774.72; (8)ACD/KOC (pH 7.4): 774.72; (9)#H bond acceptors: 2; (10)#H bond donors: 0; (11)#Freely Rotating Bonds: 5; (12)Polar Surface Area: 26.3 Å2; (13)Index of Refraction: 1.457; (14)Molar Refractivity: 43.94 cm3; (15)Molar Volume: 161.1 cm3; (16)Polarizability: 17.42×10-24cm3; (17)Surface Tension: 32.5 dyne/cm; (18)Density: 1.297 g/cm3; (19)Flash Point: 80.6 °C; (20)Enthalpy of Vaporization: 43.2 kJ/mol; (21)Boiling Point: 195.9 °C at 760 mmHg; (22)Vapour Pressure: 0.411 mmHg at 25°C.
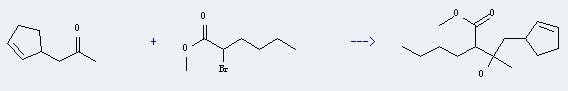
Uses of Methyl 2-bromohexanoate: It can react with cyclopent-2-enyl-acetone to produce 2-(2-cyclopent-2-enyl-1-hydroxy-1-methyl-ethyl)-hexanoic acid methyl ester. And the yield is about 70%.
You should be cautious while dealing with this chemical. It irritates eyes, respiratory system and skin. Therefore, you had better take the following instructions: Wear suitable protective clothing, and in case of contacting with eyes, rinse immediately with plenty of water and seek medical advice.
You can still convert the following datas into molecular structure:
(1)SMILES: BrC(C(=O)OC)CCCC
(2)InChI: InChI=1/C7H13BrO2/c1-3-4-5-6(8)7(9)10-2/h6H,3-5H2,1-2H3
(3)InChIKey: YGLPDRIMFIXNBI-UHFFFAOYAZ
Related Products
- Methyl 1-Benzyl-5-oxopyrrolidine-3-carboxylate
- Methyl (((methoxymethylphosphinothioyl)thio)acetyl)methylcarbamate
- Methyl (+)-(3R)-7-[4-(4-fluorophenyl)-6-isopropyl-2-(N-methyl-N-methanesulfonylamino)pyrimidin-5-yl]-3-hydroxy-5-oxo-(6E)-heptenoate
- Methyl (2-amino-5-methyl-1,3-thiazol-4-yl)acetate
- Methyl (2-chloromethyl)oxazole-4-carboxylate
- Methyl (2E)-3-(4-methylphenyl)propenoate
- Methyl (2E)-3-cyclohexylprop-2-enoate
- Methyl (2R)-2-[(tert-butoxycarbonyl)amino]-3-iodopropanoate
- Methyl (2R)-2-[4-(2,4-dichlorophenoxy)phenoxy]propanoate
- Methyl (2R)-2-amino-2-cyclohexylethanoate hydrochloride
- 5445-22-7
- 5445-24-9
- 5445-26-1
- 5445-29-4
- 54453-91-7
- 54453-93-9
- 54454-10-3
- 54454-12-5
- 5445-44-3
- 5445-51-2
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View