-
Name
Sodium cyanide
- EINECS 205-599-4
- CAS No. 143-33-9
- Article Data128
- CAS DataBase
- Density 1.6 g/cm3
- Solubility 37 g/100mL (20 °C) in water
- Melting Point 563.7 °C
- Formula NaCN
- Boiling Point 1497 °C
- Molecular Weight 49.0075
- Flash Point 1500°C
- Transport Information UN 1689 6.1/PG 1
- Appearance white crystals
- Safety 7-28-29-45-60-61-28A
- Risk Codes 26/27/28-32-50/53
-
Molecular Structure
-
Hazard Symbols
 T+,
T+, N
N
- Synonyms Sodiumcyanide (8CI);Cyanasalt H;Cyanasalt S;Cymag;NSC 77379;Natrium cyanide;Sodium cyanide (Na(CN));sodium cyanide 98%;
- PSA 23.79000
- LogP 0.01678
Synthetic route
-
-
107126-37-4
α-benzyloxy-α-phenyl-as-triazine-5-acetonitrile

-
A
-
143-33-9
sodium cyanide

-
B
-
107126-34-1
5-benzoyl-6-methyl-3-phenyl-as-triazine

-
C
-
65-85-0
benzoic acid

| Conditions | Yield |
|---|---|
| With sodium hydroxide In water; dimethyl sulfoxide | A n/a B 54% C n/a |


| Conditions | Yield |
|---|---|
| at 22 - 27℃; | |
| at 22 - 27℃; |
-
-
60-29-7
diethyl ether

-
-
506-68-3
bromocyane

-
-
996-82-7
sodium diethylmalonate

-
A
-
4513-67-1
diethyl 2-cyanomalonate

-
B
-
6174-95-4
1,1,2,2-tetracarboethoxy-ethylene

-
C
-
632-56-4
tetraethyl ethane-1,1,2,2-tetracarboxylate

-
D
-
143-33-9
sodium cyanide

-
-
506-68-3
bromocyane

-
-
64-17-5
ethanol

-
-
996-82-7
sodium diethylmalonate

-
A
-
4513-67-1
diethyl 2-cyanomalonate

-
B
-
6174-95-4
1,1,2,2-tetracarboethoxy-ethylene

-
C
-
632-56-4
tetraethyl ethane-1,1,2,2-tetracarboxylate

-
D
-
143-33-9
sodium cyanide

-
-
506-68-3
bromocyane

-
-
64-17-5
ethanol

-
-
996-82-7
sodium diethylmalonate

-
A
-
6174-95-4
1,1,2,2-tetracarboethoxy-ethylene

-
B
-
143-33-9
sodium cyanide


| Conditions | Yield |
|---|---|
| With sodium hydroxide | |
| With sodium hydroxide; ethanol | |
| With sodium hydroxide at 200 - 300℃; |
-
-
13534-15-1, 88770-07-4
diethyl oximidate

-
-
141-52-6
sodium ethanolate

-
A
-
2812-77-3
diethyl imidocarbonate

-
B
-
143-33-9
sodium cyanide

-
-
816-27-3
ethyl 2-ethoxy-2-iminoacetate

-
-
141-52-6
sodium ethanolate

-
A
-
143-33-9
sodium cyanide

-
B
-
105-58-8
Diethyl carbonate

-
-
939-56-0
benzoyloxyacetonitrile

-
-
141-52-6
sodium ethanolate

-
A
-
532-32-1
sodium benzoate

-
B
-
93-89-0
benzoic acid ethyl ester

-
C
-
143-33-9
sodium cyanide

-
-
100400-22-4
1-hydroxy-2-isopropyl-5-methyl-cyclohexanecarbonitrile

-
A
-
143-33-9
sodium cyanide

-
B
-
10458-14-7
Menthone


| Conditions | Yield |
|---|---|
| With sodium amide at 620℃; |

-
A
-
143-33-9
sodium cyanide

-
B
-
19981-17-0, 20611-81-8, 123092-13-7, 17292-62-5
sodium cyanamide

| Conditions | Yield |
|---|---|
| With ammonia at 500℃; |
-
-
61706-59-0
sodium formaldoximate

-
-
143-33-9
sodium cyanide

| Conditions | Yield |
|---|---|
| bei der Explosion; |
-
-
556-64-9
methyl thiocyanate

-
-
2303-76-6, 118080-94-7
sodium diethyl phosphite

-
-
108-88-3
toluene

-
A
-
152-20-5
O,O,S-trimethyl phosphorothioate

-
B
-
143-33-9
sodium cyanide

-
-
141-52-6
sodium ethanolate

-
-
623-49-4
ethyl cyanoformate

-
A
-
143-33-9
sodium cyanide

-
B
-
105-58-8
Diethyl carbonate

-
-
19432-60-1
4-cyano-1,4-dihydro-N-methylpyridine-3-carboxamide

-
A
-
143-33-9
sodium cyanide

-
B
-
1005-24-9
1-methylnicotinamide chloride

| Conditions | Yield |
|---|---|
| at 25℃; Equilibrium constant; reaction in reversed-micellar system H2O/ HTAB/ CHCl3-isooctane, diff. molar ratios H2O/ HTAB, other temp.; |

| Conditions | Yield |
|---|---|
| With iodide In ethanol at 30℃; Equilibrium constant; buffer solvent (CH3CO2Na, CH3CO2H); |
-
-
51210-89-0
1,3-diphenyl-2-oximino-1,3-propanedione

-
A
-
93-89-0
benzoic acid ethyl ester

-
B
-
143-33-9
sodium cyanide

-
C
-
65-85-0
benzoic acid

| Conditions | Yield |
|---|---|
| With sodium ethanolate In ethanol Product distribution; |

-
-
74784-26-2
hexadecyltrimethylammonium cyanide

-
A
-
143-33-9
sodium cyanide

-
B
-
57-09-0
cetyltrimethylammonim bromide

| Conditions | Yield |
|---|---|
| With sodium bromide In water at 20℃; Equilibrium constant; |

| Conditions | Yield |
|---|---|
| at 750 - 850℃; beim Zusammenschmelzen; | |
| at 750 - 850℃; beim Zusammenschmelzen; |

-
-
6011-99-0
benzo[d]thiazol-2-yl thiocyanate

-
-
2492-26-4
sodium 2-mercaptobenzothiazole

-
A
-
4074-77-5
2,2'-thiobis(benzothiazole)

-
B
-
120-78-5
di(benzothiazol-2-yl)disulfide

-
C
-
143-33-9
sodium cyanide

-
D
-
540-72-7
sodium thiocyanide


| Conditions | Yield |
|---|---|
| at 250℃; |

| Conditions | Yield |
|---|---|
| at 750 - 850℃; beim Zusammenschmelzen; |


-
-
143-33-9
sodium cyanide

-
-
1585-16-6
2-chloromethyl-1,3,5-trimethylbenzene

-
-
34688-71-6
mesitylacetonitrile

| Conditions | Yield |
|---|---|
| In dimethyl sulfoxide at 80℃; for 0.5h; Substitution; | 100% |
| Stage #1: sodium cyanide In ethanol; water Heating / reflux; Stage #2: 2-chloromethyl-1,3,5-trimethylbenzene In ethanol; water for 3h; Heating / reflux; | 72% |
| In ethanol |
-
-
143-33-9
sodium cyanide

-
-
102-46-5
4-(chloromethyl)-1,2-dimethylbenzene

-
-
3020-06-2
3,4-dimethylphenylacetonitrile

| Conditions | Yield |
|---|---|
| In N,N-dimethyl-formamide at 100℃; for 16h; | 100% |
| In ethanol | |
| In ethanol Heating; |
-
-
98-01-1
furfural

-
-
143-33-9
sodium cyanide

-
-
541-41-3
chloroformic acid ethyl ester

-
-
20893-23-6
carbonic acid, cyano(2-furyl)methyl ethyl ester

| Conditions | Yield |
|---|---|
| With tetrabutyl-ammonium chloride In dichloromethane; water Heating; | 100% |

-
-
54475-84-2
10-Chloro-5H-benzoxazole<3,2-a>quinolin-5-one

-
-
143-33-9
sodium cyanide

-
-
80990-60-9
1-(5-chloro-2-hydroxyphenyl)-1,4-dihydro-4-oxo-2-quinolinecarbonitrile

| Conditions | Yield |
|---|---|
| In dimethyl sulfoxide at 120℃; for 0.5h; | 100% |

| Conditions | Yield |
|---|---|
| With benzyltri(n-butyl)ammonium chloride In water at 80℃; for 3h; | 100% |
-
-
7500-58-5, 20246-03-1, 20518-14-3, 32970-96-0, 60894-68-0
trans-4,5-bis(hydroxymethyl)cyclohexene bis(toluene-p-sulfonate)

-
-
143-33-9
sodium cyanide

-
-
119595-00-5
(+/-)-(trans-cyclohex-4-ene-1,2-diyl)-di-acetonitrile

| Conditions | Yield |
|---|---|
| In ethanol for 72h; Heating; | 100% |
-
-
143-33-9
sodium cyanide

-
-
6616-69-9, 16964-48-0, 16964-51-5, 52195-11-6, 60761-09-3, 63790-37-4, 138383-58-1
dl-6aα,7,10,10aα-Tetrahydro-1-hydroxy-6,6-dimethyl-3-pentyl-6H-dibenzo[b,d]pyran-9(8H)-on

-
-
105539-71-7, 105539-72-8, 121657-71-4
(trans-rac)-6a,7,8,9,10,10a-hexahydro-1,9-dihydroxy-6,6-dimethyl-3-pentyl-6H-dibenzopyran-9-carbonitrile

| Conditions | Yield |
|---|---|
| In methanol for 2h; Ambient temperature; | 100% |
-
-
143-33-9
sodium cyanide

-
-
117571-35-4
1-(bromomethyl)-4-methyl-2-(phenylmethoxy)benzene

-
-
117571-36-5
<4-methyl-2-(phenylmethoxy)phenyl>acetonitrile

| Conditions | Yield |
|---|---|
| With tetrabutylammomium bromide In dichloromethane; water at 20℃; for 1h; | 100% |
| tetrabutylammomium bromide In dichloromethane; water at 20℃; for 15h; |
-
-
143-33-9
sodium cyanide

-
-
6522-22-1, 14697-77-9
6-chloro-9-thiabicyclo<3.3.1>non-2-ene

-
-
6435-98-9, 74830-38-9
9-thiabicyclo<3.3.1>non-2-ene-6-carbonitrile

| Conditions | Yield |
|---|---|
| With 18-crown-6 ether In chloroform for 4h; Heating; | 100% |
| In water; acetone for 20h; Heating; | 71% |
-
-
143-33-9
sodium cyanide

-
-
80314-64-3
2-(chloromethyl)-1-methoxy-4-(2-methoxyethyl)benzene

-
-
80314-59-6
<2'-methoxy-5'-(2''-methoxyethyl)phenyl>acetonitrile

| Conditions | Yield |
|---|---|
| In dimethyl sulfoxide | 100% |
| In dimethyl sulfoxide at 42.5℃; for 3h; | 100% |
-
-
143-33-9
sodium cyanide

-
-
73454-44-1
6-benzyloxy-4-methyl-4Z-hexen-1-ol p-toluenesulfonate

-
-
73454-49-6
(Z)-7-benzyloxy-5-methyl-5-heptenenitrile

| Conditions | Yield |
|---|---|
| In N,N-dimethyl-formamide at 98℃; for 24h; | 100% |
-
-
143-33-9
sodium cyanide

-
-
122807-12-9
ethyl N-<2-(bromoacetyl)phenyl>carbamate

-
-
138768-58-8
1,4-bis(2-ethoxycarbonylaminophenyl)-2-cyano-1,4-butanedione

| Conditions | Yield |
|---|---|
| In N,N-dimethyl-formamide for 1h; Ambient temperature; | 100% |
-
-
143-33-9
sodium cyanide

-
-
80535-58-6
8-methoxy-5-<2'-(2''-methyl-1'',3''-dioxolan-2''-yl)ethyl>-3,4-dihydronaphthalen-2(1H)-one

-
-
80535-46-2
2-hydroxy-8-methoxy-5-<2'-(2"-methyl-1",3"-dioxolan-2"-yl)ethyl>-1,2,3,4-tetrahydronaphthalene-2-carbonitrile

| Conditions | Yield |
|---|---|
| With hydrogenchloride In tetrahydrofuran; water | 100% |
-
-
143-33-9
sodium cyanide

-
-
76235-43-3
4-cyanomethyl-1,1-ethylenedithio-2-methylcyclohexane

| Conditions | Yield |
|---|---|
| In dimethyl sulfoxide | 100% |
-
-
143-33-9
sodium cyanide

-
-
115094-15-0
Toluene-4-sulfonic acid (5E,7E)-(R)-4-methyl-undeca-5,7-dienyl ester

-
-
115094-13-8
(R)-1-cyano-4-methyl-(E,E)-5,7-undecadiene

| Conditions | Yield |
|---|---|
| In dimethyl sulfoxide at 80 - 85℃; for 3.5h; | 100% |
-
-
143-33-9
sodium cyanide

-
-
82757-18-4
2-(2-formamidophenyl)adamantan-2-ol

-
-
82757-22-0
2-(2-aminophenyl)-2-cyanoadamantane

| Conditions | Yield |
|---|---|
| In N,N-dimethyl-formamide at 80℃; for 120h; | 100% |
| In N,N-dimethyl-formamide at 80℃; | 100% |
-
-
143-33-9
sodium cyanide

-
-
90664-12-3
(2R,3S,4E)-(-)-(3-Methoxy-2-methyl-5-phenyl-4-pentenyl)-p-toluolsulfonat

-
-
90664-13-4
(3R,4R,5E)-(+)-4-Methoxy-3-methyl-6-phenyl-5-hexennitril

| Conditions | Yield |
|---|---|
| In N,N-dimethyl-formamide at 115℃; for 0.666667h; | 100% |
-
-
143-33-9
sodium cyanide

-
-
141546-69-2
(3S,4R)-5-benzyloxy-4-methoxy-3-methylpentanenitrile

| Conditions | Yield |
|---|---|
| In N,N-dimethyl-formamide at 50℃; for 6h; | 100% |
-
-
143-33-9
sodium cyanide

-
-
93645-46-6
11,12-dicyano-9,10-anthraquinodimethane

-
-
93645-47-7
9-(dicyanomethyl)-10-cyanomethylanthracene

| Conditions | Yield |
|---|---|
| In 1,4-dioxane; ethanol for 24h; Heating; | 100% |
-
-
143-33-9
sodium cyanide

-
-
79557-82-7
1-[4-(9-Bromo-nonyloxy)-2-hydroxy-3-propyl-phenyl]-ethanone

-
-
92518-11-1
10-(4-Acetyl-3-hydroxy-2-propylphenoxy)decane nitrile

| Conditions | Yield |
|---|---|
| In N,N-dimethyl-formamide at 75 - 85℃; for 17h; | 100% |
-
-
143-33-9
sodium cyanide

-
-
106627-35-4
1-[4-(10-Bromo-decyloxy)-2-hydroxy-3-propyl-phenyl]-ethanone

-
-
92518-12-2
11-(4-Acetyl-3-hydroxy-2-propyl-phenoxy)-undecanenitrile

| Conditions | Yield |
|---|---|
| In N,N-dimethyl-formamide at 75 - 85℃; for 17h; | 100% |
-
-
143-33-9
sodium cyanide

-
-
141546-68-1
4-O-benzoyl-2-deoxy-1-O-methylsulfonyl-2-C-methyl-3-O-methyl-L-threitol

-
-
141546-69-2
(3S,4R)-5-benzyloxy-4-methoxy-3-methylpentanenitrile

| Conditions | Yield |
|---|---|
| In N,N-dimethyl-formamide at 50℃; for 6h; | 100% |
-
-
143-33-9
sodium cyanide

-
-
154954-44-6
(+/-)-2,3-dihydro-5-methoxy-4,6,7-trimethyl-2-benzofuranethanol p-toluenesulfonate

-
-
154954-45-7
(+/-)-β-(2,3-dihydro-5-methoxy-4,6,7-trimethyl-2-benzofuranyl)propionitrile

| Conditions | Yield |
|---|---|
| In ethanol; N,N-dimethyl-formamide for 1.5h; Heating; | 100% |
-
-
143-33-9
sodium cyanide

-
-
102492-60-4
2-(bromomethyl)-11H-dibenzo<1,4>dioxepin

-
-
102492-61-5
11H-dibenzo<1,4>dioxepin-2-acetonitrile

| Conditions | Yield |
|---|---|
| In dimethyl sulfoxide at 60℃; for 1h; | 100% |

| Conditions | Yield |
|---|---|
| With tetrabutyl-ammonium chloride In dichloromethane; water Ambient temperature; | 100% |

-
-
143-33-9
sodium cyanide

-
-
108-24-7
acetic anhydride

-
-
129867-98-7
(R)-N-(isopropoxycarbonyl)phenylalaninal

-
-
129867-99-8, 129868-00-4
(3-R)-2-acetoxy-3-isopropoxycarbonylamino-4-phenylbutyronitrile

| Conditions | Yield |
|---|---|
| With benzyltri(n-butyl)ammonium chloride In dichloromethane; water at 0℃; for 3h; | 100% |

-
-
79027-28-4
(2S)-3-(benzyloxy)-2-methylpropanal

-
-
143-33-9
sodium cyanide

-
-
2393-23-9
4-methoxy-benzylamine

-
-
130533-04-9
(3R)-2-(4-methoxybenzylamino)-3-benzyloxymethylbutyronitrile

| Conditions | Yield |
|---|---|
| With hydrogenchloride In methanol; water for 2h; Ambient temperature; | 100% |

-
-
143-33-9
sodium cyanide

-
-
107097-23-4
p-Toluenesulfonic acid 3-(indol-2-yl)propyl ester

-
-
165532-78-5
4-(1H-Indol-2-yl)butanenitrile

| Conditions | Yield |
|---|---|
| In dimethyl sulfoxide Ambient temperature; | 100% |
-
-
143-33-9
sodium cyanide

-
-
165532-83-2
1-Benzenesulfonyl-2-(4-bromobutanyl)-1H-indole

-
-
165532-84-3
5-(1-Benzenesulfonyl-1H-indol-2-yl)pentanenitrile

| Conditions | Yield |
|---|---|
| In dimethyl sulfoxide at 80℃; for 12h; | 100% |
-
-
143-33-9
sodium cyanide

-
-
159886-35-8
2-[4-(3-Bromopropylcarbamoyl)-5-(trifluoromethyl)pyrazol-1-yl]-4-(3-trifluoromethylphenyl)thiazole

-
-
1026655-81-1
5-Trifluoromethyl-1-[4-(3-trifluoromethyl-phenyl)-thiazol-2-yl]-1H-pyrazole-4-carboxylic acid (3-cyano-propyl)-amide

| Conditions | Yield |
|---|---|
| In N,N-dimethyl-formamide | 100% |
Sodium Cyanide Consensus Reports
Sodium Cyanide Standards and Recommendations
ACGIH TLV: CL 5 mg(CN)/m3 (skin)
DFG MAK: 5 mg(CN)/m3
NIOSH REL: CL 5 mg(CN)/m3/10M
DOT Classification: 6.1; Label: Poison
Sodium Cyanide Analytical Methods
Sodium Cyanide Specification
The sodium cyanide. With the CAS registry number 143-33-9, it is also named as Hydrocyanic acid, sodium salt. The product's categories are Sodium Cyanide; Inorganics; Peroxidase Inhibitors; Essential Chemicals; Reagent Grade; Routine Reagents; Molecular Biology; Molecular Biology Reagents; Superoxide Dismutase. The structure of solid NaCN is related to that of sodium chloride. It is white crystals with faint odor of bitter almond, which is soluble in water, ammonia, ethanol and methanol. When contact with acid and water, it liberates very toxic gas. Additionally, this chemical should be sealed in the container and stored in the cool, ventilate and dry place. Keep Sodium Cyanide separate from alkali, acids and food additives.
Physical properties about sodium cyanide are:
(1)H-Bond Donor: 0; (2)H-Bond Acceptor: 2; (3)Rotatable Bond Count: 0; (4)Exact Mass: 48.992844; (5)MonoIsotopic Mass: 48.992844; (6)Topological Polar Surface Area: 23.8; (7)Heavy Atom Count: 3; (8)Formal Charge: 0; (9)Complexity: 12.8; (10)Covalently-Bonded Unit Count: 2.
Preparation of Sodium Cyanide:
It can be obtained by treating hydrogen cyanide with sodium hydroxide:
HCN + NaOH → NaCN + H2O
Uses of Sodium Cyanide:
It is mainly used to extract gold and other precious metals in mining.
4 Au + 8 NaCN + O2 + 2 H2O → 4 Na[Au(CN)2] + 4 NaOH
It is also used to kill or stun rapidly such as in illegal cyanide fishing and in collecting jars used by entomologists. What's more, this chemica is used in plastics, pharmaceuticals, pesticides, dyes, metallurgy, electroplating, mineral processing and other industries. In addition, it can react with cycloheptanone to get 4-cyanocycloheptanone. This reaction needs reagents Na2S2O8, Na2SO4 and solvent water at temperature of 80 °C. The reaction time is 5 hours. The yield is 35%.
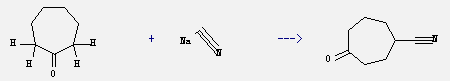
Safety Information of Sodium Cyanide:
It is not only very toxic by inhalation, in contact with skin and if swallowed, but also very toxic to aquatic organisms, may cause long-term adverse effects in the aquatic environment. After contact with skin, wash immediately with plenty of soap-suds. In case of accident or if you feel unwell, seek medical advice immediately (show the label whenever possible.) This material and its container must be disposed of as hazardous waste. And people should avoid release it to the environment. Refer to special instructions / safety data sheets.
You can still convert the following datas into molecular structure:
1. SMILES:[C-]#N.[Na+]
2. InChI:InChI=1/CN.Na/c1-2;/q-1;+1
3. InChIKey:MNWBNISUBARLIT-UHFFFAOYAG
The toxicity data of Sodium Cyanide is as follows:
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| bird - wild | LD50 | oral | 4mg/kg (4mg/kg) | BEHAVIORAL: TREMOR BEHAVIORAL: ATAXIA LUNGS, THORAX, OR RESPIRATION: DYSPNEA | Journal of Wildlife Diseases. Vol. 22, Pg. 538, 1986. |
| chicken | LD50 | oral | 21mg/kg (21mg/kg) | SENSE ORGANS AND SPECIAL SENSES: OTHER: EYE LUNGS, THORAX, OR RESPIRATION: OTHER CHANGES GASTROINTESTINAL: CHANGES IN STRUCTURE OR FUNCTION OF SALIVARY GLANDS | Journal of Wildlife Diseases. Vol. 22, Pg. 538, 1986. |
| child | LDLo | oral | 100mg/kg (100mg/kg) | GASTROINTESTINAL: OTHER CHANGES | Forensic Science International. Vol. 38, Pg. 173, 1988. |
| dog | LD50 | subcutaneous | 5360ug/kg (5.36mg/kg) | JAMA, Journal of the American Medical Association. Vol. 149, Pg. 113, 1952. | |
| dog | LDLo | intravenous | 1300ug/kg (1.3mg/kg) | Federation Proceedings, Federation of American Societies for Experimental Biology. Vol. 6, Pg. 349, 1947. | |
| domestic animals - goat/sheep | LD50 | oral | 4mg/kg (4mg/kg) | Toxicology and Applied Pharmacology. Vol. 45, Pg. 359, 1978. | |
| duck | LD50 | oral | 2500ug/kg (2.5mg/kg) | National Technical Information Service. Vol. OTS0530251, | |
| frog | LDLo | parenteral | 60mg/kg (60mg/kg) | PERIPHERAL NERVE AND SENSATION: SPASTIC PARALYSIS WITH OR WITHOUT SENSORY CHANGE BEHAVIORAL: CHANGES IN MOTOR ACTIVITY (SPECIFIC ASSAY) BEHAVIORAL: CONVULSIONS OR EFFECT ON SEIZURE THRESHOLD | Naunyn-Schmiedeberg's Archiv fuer Experimentelle Pathologie und Pharmakologie. Vol. 166, Pg. 437, 1932. |
| guinea pig | LD50 | subcutaneous | 5800ug/kg (5.8mg/kg) | Medicina del Lavoro. Industrial Medicine. Vol. 46, Pg. 221, 1955. | |
| human | LDLo | oral | 2800ug/kg (2.8mg/kg) | National Technical Information Service. Vol. OTS0528336, | |
| human | LDLo | oral | 2857ug/kg (2.857mg/kg) | "Toxicology of Drugs and Chemicals," Deichmann, W.B., New York, Academic Press, Inc., 1969Vol. -, Pg. 191, 1969. | |
| mammal (species unspecified) | LD50 | oral | 8mg/kg (8mg/kg) | Bulletin of Environmental Contamination and Toxicology. Vol. 23, Pg. 211, 1979. | |
| man | LDLo | oral | 6557ug/kg (6.557mg/kg) | BEHAVIORAL: FLUID INTAKE GASTROINTESTINAL: GASTRITIS | Acta Pharmacologica et Toxicologica. Vol. 1, Pg. 18, 1945. |
| man | LDLo | unreported | 2206ug/kg (2.206mg/kg) | "Poisoning; Toxicology, Symptoms, Treatments," 2nd ed., Arena, J.M., Springfield, IL, C.C. Thomas, 1970Vol. 2, Pg. 73, 1970. | |
| man | TDLo | oral | 714ug/kg (0.714mg/kg) | BEHAVIORAL: "HALLUCINATIONS, DISTORTED PERCEPTIONS" BEHAVIORAL: MUSCLE WEAKNESS | Acta Pharmacologica et Toxicologica. Vol. 20, Pg. 291, 1963. |
| mouse | LD50 | intraperitoneal | 4900ug/kg (4.9mg/kg) | National Technical Information Service. Vol. OTS0528336, | |
| mouse | LD50 | subcutaneous | 3600ug/kg (3.6mg/kg) | American Journal of Physiology. Vol. 179, Pg. 60, 1954. | |
| mouse | LD50 | unreported | 10mg/kg (10mg/kg) | Igaku No Ayumi. Progress in Medicine. Vol. 112, Pg. 861, 1980. | |
| quail | LD50 | oral | 8500ug/kg (8.5mg/kg) | BEHAVIORAL: TREMOR BEHAVIORAL: ATAXIA LUNGS, THORAX, OR RESPIRATION: DYSPNEA | Journal of Wildlife Diseases. Vol. 22, Pg. 538, 1986. |
| rabbit | LD50 | intramuscular | 1666ug/kg (1.666mg/kg) | Journal of the American College of Toxicology. Vol. 1(3), Pg. 120, 1982. | |
| rabbit | LD50 | ocular | 5048ug/kg (5.048mg/kg) | Journal of the American College of Toxicology. Vol. 1(3), Pg. 120, 1982. | |
| rabbit | LD50 | skin | 10400ug/kg (10.4mg/kg) | BEHAVIORAL: SOMNOLENCE (GENERAL DEPRESSED ACTIVITY) LUNGS, THORAX, OR RESPIRATION: DYSPNEA BEHAVIORAL: TREMOR | National Technical Information Service. Vol. OTS0536153, |
| rabbit | LDLo | subcutaneous | 2200ug/kg (2.2mg/kg) | JAMA, Journal of the American Medical Association. Vol. 100, Pg. 1920, 1933. | |
| rat | LD50 | intraperitoneal | 4300ug/kg (4.3mg/kg) | Gigiena i Sanitariya. For English translation, see HYSAAV. Vol. 43(12), Pg. 90, 1978. | |
| rat | LD50 | oral | 6440ug/kg (6.44mg/kg) | "Sbornik Vysledku Toxixologickeho Vysetreni Latek A Pripravku," Marhold, J.V., Institut Pro Vychovu Vedoucicn Pracovniku Chemickeho Prumyclu Praha, Czechoslovakia, 1972Vol. -, Pg. 13, 1972. | |
| women | LDLo | oral | 40mg/kg (40mg/kg) | GASTROINTESTINAL: OTHER CHANGES | Forensic Science International. Vol. 38, Pg. 173, 1988. |
Related Products
- Sodium
- Sodium (+)-10-camphorsulfonate
- Sodium 1,3-benzothiazole-2-thiolate
- Sodium 1-amine-2-naphthol-4-sulfonate
- Sodium 1-amino-4-((4-butylphenyl)amino)-9,10-dihydro-9,10-dioxoanthracene-2-sulphonate
- Sodium 1-butanesulfonate
- Sodium 1-dodecanesulfonate
- Sodium 1-menaphthyl sulfate
- Sodium 1-naphthalenesulfonate
- Sodium 1-Nonanesulfonate
- 143339-58-6
- 14334-40-8
- 14335-33-2
- 143355-56-0
- 143360-00-3
- 143361-87-9
- 14336-71-1
- 14337-43-0
- 14338-01-3
- 143382-03-0
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View