-
Name
Tetraethyl orthocarbonate
- EINECS 201-082-2
- CAS No. 78-09-1
- Article Data27
- CAS DataBase
- Density 0.948 g/cm3
- Solubility insoluble in water
- Melting Point 222 °C
- Formula C9H20O4
- Boiling Point 159.5 °C at 760 mmHg
- Molecular Weight 192.255
- Flash Point 52.8 °C
- Transport Information UN 3272 3/PG 3
- Appearance clear colorless liquid
- Safety 37/39-26-16-36
- Risk Codes 10-36/37/38
-
Molecular Structure
-
Hazard Symbols
 Xi,
Xi, F
F
- Synonyms Tetraethylorthocarbonate;Orthocarbonicacid, tetraethyl ester (6CI,7CI,8CI);NSC 28574;Tetraethoxymethane;(triethoxymethoxy)ethane;Ethane, 1,1',1'',1'''-(methanetetrayltetrakis(oxy))tetrakis-;
- PSA 36.92000
- LogP 1.74370
Synthetic route

| Conditions | Yield |
|---|---|
| With sodium ethanolate at 65 - 70℃; for 3h; Temperature; Large scale; | 91% |
| With sodium Darst.; |
-
-
23213-96-9
ethyl 2,2,2-trichloroacetimidate

-
-
124-41-4
sodium methylate

-
-
78-09-1
orthocarbonic acid tetraethyl ester

| Conditions | Yield |
|---|---|
| In ethanol for 1h; Heating; | 85% |
-
-
141-52-6
sodium ethanolate

-
-
545-06-2
trichloroacetonitrile

-
-
78-09-1
orthocarbonic acid tetraethyl ester

| Conditions | Yield |
|---|---|
| In ethanol for 3.5h; Heating / reflux; | 83% |
| In ethanol Heating; |
-
-
56-23-5
tetrachloromethane

-
-
64-17-5
ethanol

-
-
141-52-6
sodium ethanolate

-
-
78-09-1
orthocarbonic acid tetraethyl ester

| Conditions | Yield |
|---|---|
| at 80 - 95℃; for 5h; Temperature; | 83% |


| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid Heating; | 71% |
-
-
68714-37-4
triethoxyacetonitrile

-
-
141-52-6
sodium ethanolate

-
A
-
78-09-1
orthocarbonic acid tetraethyl ester

-
B
-
81759-13-9
Triethylmethylorthocarbonat

-
C
-
81759-14-0
Trimethylethylorthocarbonat

| Conditions | Yield |
|---|---|
| In ethanol 1.) 5 min, 60 deg C, 2.) 30 min, to room temperature; Title compound not separated from byproducts; | A 70% B 58% C 2% |


| Conditions | Yield |
|---|---|
| With silver trifluoroacetate; triethylamine for 5h; Ambient temperature; | 61% |
| Yield given. Multistep reaction; |
-
-
68714-37-4
triethoxyacetonitrile

-
-
109-73-9
N-butylamine

-
A
-
78-09-1
orthocarbonic acid tetraethyl ester

-
B
-
81759-28-6
Diethyl(butylimido)carbonat

-
C
-
81759-30-0
1,3-Dibutyl-2-ethylisoharnstoff

| Conditions | Yield |
|---|---|
| at 120℃; Yield given. Yields of byproduct given. Title compound not separated from byproducts; | A n/a B n/a C 59% |
| at 120℃; Heating; Yields of byproduct given; | A n/a B n/a C 59% |


| Conditions | Yield |
|---|---|
| In ethanol at 57 - 64℃; for 2h; Heating / reflux; Industry scale; | 58.3% |
-
-
68714-37-4
triethoxyacetonitrile

-
-
124-41-4
sodium methylate

-
A
-
78-09-1
orthocarbonic acid tetraethyl ester

-
B
-
81759-13-9
Triethylmethylorthocarbonat

-
C
-
81759-14-0
Trimethylethylorthocarbonat

| Conditions | Yield |
|---|---|
| In methanol at 60℃; Title compound not separated from byproducts; | A 8 % Chromat. B 58% C 2 % Chromat. |

-
-
36749-09-4
3,3-diethoxypentane

-
A
-
78-09-1
orthocarbonic acid tetraethyl ester

-
B
-
1128-76-3
3-chloro-benzoic acid ethyl ester

-
C
-
105-37-3
Ethyl propionate

-
D
-
105-58-8
Diethyl carbonate

| Conditions | Yield |
|---|---|
| With 3-chloro-benzenecarboperoxoic acid In dichloromethane at 15 - 30℃; for 0.5h; Further byproducts given; | A 18% B n/a C n/a D 50% |

-
-
68714-37-4
triethoxyacetonitrile

-
-
100-46-9
benzylamine

-
A
-
78-09-1
orthocarbonic acid tetraethyl ester

-
B
-
6263-03-2
Diethyl(benzylimido)carbonat

-
C
-
81759-29-7
1,3-Dibenzyl-2-ethylisoharnstoff

| Conditions | Yield |
|---|---|
| at 120℃; | A n/a B 45% C 14% |
| at 120℃; Heating; | A n/a B 45% C 14% |

-
-
115-80-0
Triethyl orthopropionate

-
A
-
78-09-1
orthocarbonic acid tetraethyl ester

-
B
-
105-58-8
Diethyl carbonate

| Conditions | Yield |
|---|---|
| With 3-chloro-benzenecarboperoxoic acid In dichloromethane at 15 - 30℃; for 0.5h; | A 25% B 45% |
-
-
124-41-4
sodium methylate

-
-
545-06-2
trichloroacetonitrile

-
A
-
78-09-1
orthocarbonic acid tetraethyl ester

-
B
-
23213-96-9
ethyl 2,2,2-trichloroacetimidate

-
C
-
68714-37-4
triethoxyacetonitrile

-
D
-
77388-04-6
Ethyl-triethoxyacetimidat

| Conditions | Yield |
|---|---|
| In n-heptane at 94℃; for 2h; | A n/a B n/a C n/a D 5.5% |

-
-
56-23-5
tetrachloromethane

-
-
141-52-6
sodium ethanolate

-
A
-
78-09-1
orthocarbonic acid tetraethyl ester

-
B
-
122-51-0
orthoformic acid triethyl ester

-
-
60-29-7
diethyl ether

-
-
558-13-4
carbon tetrabromide

-
-
141-52-6
sodium ethanolate

-
-
78-09-1
orthocarbonic acid tetraethyl ester


| Conditions | Yield |
|---|---|
| With diethyl ether |
-
-
13029-05-5
trichloro-methanesulfenic acid ethyl ester

-
-
141-52-6
sodium ethanolate

-
-
78-09-1
orthocarbonic acid tetraethyl ester
-
-
60-29-7
diethyl ether

-
-
64-17-5
ethanol

-
-
141-52-6
sodium ethanolate

-
-
594-42-3
chlorothio-trichloro-methane

-
-
78-09-1
orthocarbonic acid tetraethyl ester

| Conditions | Yield |
|---|---|
| bei Einw.; |

-
-
141-52-6
sodium ethanolate

-
-
594-42-3
chlorothio-trichloro-methane

-
-
78-09-1
orthocarbonic acid tetraethyl ester

| Conditions | Yield |
|---|---|
| With diethyl ether |
-
-
75-15-0
carbon disulfide

-
-
3173-69-1, 14791-99-2, 24126-00-9, 96473-13-1
tin(II) ethoxide

-
-
78-09-1
orthocarbonic acid tetraethyl ester
-
-
64-17-5
ethanol

-
-
15146-36-8
N-(di-ethoxymethylene)-benzamide

-
-
78-09-1
orthocarbonic acid tetraethyl ester

| Conditions | Yield |
|---|---|
| With hydrogenchloride |

| Conditions | Yield |
|---|---|
| With pyridine Ambient temperature; |
-
-
631-57-2
Acetyl cyanide

-
-
73708-97-1
N,N-Dimethyl-orthocarbamidsaeure-triethylester

-
A
-
127-19-5
N,N-dimethyl acetamide

-
B
-
78-09-1
orthocarbonic acid tetraethyl ester

-
C
-
68714-37-4
triethoxyacetonitrile

-
D
-
34644-32-1
2,2-diethoxy-2-dimethylaminoacetonitrile

| Conditions | Yield |
|---|---|
| In diethyl ether for 5h; Ambient temperature; Yield given; | |
| In diethyl ether for 5h; Ambient temperature; |

-
-
124-41-4
sodium methylate

-
A
-
1850-14-2
tetramethoxymethane

-
B
-
78-09-1
orthocarbonic acid tetraethyl ester

-
C
-
81759-13-9
Triethylmethylorthocarbonat

-
D
-
81759-14-0
Trimethylethylorthocarbonat

| Conditions | Yield |
|---|---|
| In methanol for 12h; Product distribution; Ambient temperature; also with other sodium-alcoholates; |

-
-
58137-35-2
methyl 2-(trifluoroacetamido)acrylate

-
-
2678-54-8
ketene diethyl acetal

-
C
-
7737-40-8
ethyl 2-ethoxypropionate

-
D
-
78-09-1
orthocarbonic acid tetraethyl ester

| Conditions | Yield |
|---|---|
| In chloroform-d1 at 25℃; Product distribution; Michael-Dieckmann type reaction; |


| Conditions | Yield |
|---|---|
| at 160 - 170℃; for 10h; | 98% |
| at 120℃; for 3h; Product distribution; other times and temperatures; | 21% |
| at 160℃; |
-
-
1122-28-7
4,5-dicyano-1H-imidazole

-
-
78-09-1
orthocarbonic acid tetraethyl ester

-
-
133123-67-8
1-ethyl-4,5-dicyanoimidazole

| Conditions | Yield |
|---|---|
| 1) 0.5 h, 100 deg C 2) 2 h, 150 deg C; | 96% |
-
-
78-09-1
orthocarbonic acid tetraethyl ester

-
-
402-14-2
2-nitro-5-(trifluoromethyl)aniline

-
-
1060705-50-1
C10H9F3N2O

| Conditions | Yield |
|---|---|
| With 10% Pd/C; hydrogen; acetic acid In methanol at 20℃; under 760.051 Torr; | 96% |
-
-
78-09-1
orthocarbonic acid tetraethyl ester

-
-
95-54-5
1,2-diamino-benzene

-
-
22219-23-4
2-ethoxy-1H-benzo[d]imidazole

| Conditions | Yield |
|---|---|
| Stage #1: orthocarbonic acid tetraethyl ester; 1,2-diamino-benzene With acetic acid at 70℃; for 2h; Stage #2: With potassium hydroxide In water at 20℃; for 1h; | 94% |
| Stage #1: orthocarbonic acid tetraethyl ester; 1,2-diamino-benzene With acetic acid at 70℃; for 3h; Large scale; Stage #2: With potassium hydroxide for 1h; Large scale; | 94.2% |
| In neat (no solvent) at 70℃; for 2h; | 93% |

| Conditions | Yield |
|---|---|
| With 10% Pd/C; hydrogen; acetic acid In methanol at 20℃; under 760.051 Torr; | 94% |

| Conditions | Yield |
|---|---|
| With 10% Pd/C; hydrogen; acetic acid In methanol at 20℃; under 760.051 Torr; | 94% |
-
-
78-09-1
orthocarbonic acid tetraethyl ester

-
-
1236156-65-2
2-(((2'-(1H-tetrazol-5-yl)-[1,1'-biphenyl]-4-yl)methyl)amino)-3-aminobenzoic acid-1-(((cyclohexyloxy)carbonyl)oxy)ethyl ester

-
-
145040-37-5
candesartan cilexetil

| Conditions | Yield |
|---|---|
| With acetic acid In ethyl acetate at 30℃; Temperature; | 93.9% |
| With acetic acid In toluene at 60℃; for 2h; | 81% |
-
-
78-09-1
orthocarbonic acid tetraethyl ester

| Conditions | Yield |
|---|---|
| at 20℃; for 17h; | 93.4% |
-
-
78-09-1
orthocarbonic acid tetraethyl ester

-
-
1066-54-2
trimethylsilylacetylene

-
-
91022-26-3
trimethyl(3,3,3-triethoxyprop-1-yn-1-yl)silane

| Conditions | Yield |
|---|---|
| Stage #1: trimethylsilylacetylene With magnesium; isopropyl bromide In diethyl ether at 10℃; for 1h; Inert atmosphere; Cooling with ice; Stage #2: orthocarbonic acid tetraethyl ester In diethyl ether at 20℃; for 1h; Inert atmosphere; | 93% |
| Stage #1: trimethylsilylacetylene With ethylmagnesium bromide In diethyl ether at 20℃; Reflux; Stage #2: orthocarbonic acid tetraethyl ester In diethyl ether at 20℃; Reflux; | 77% |
| Stage #1: trimethylsilylacetylene With ethylmagnesium bromide In diethyl ether at 20℃; Inert atmosphere; Heating; Stage #2: orthocarbonic acid tetraethyl ester In diethyl ether at 20℃; Inert atmosphere; Reflux; | 70% |
-
-
78-09-1
orthocarbonic acid tetraethyl ester

-
-
61063-11-4
2-Amino-3-nitrobenzoic acid,ethyl ester

-
-
577957-02-9
ethyl 2-ethoxy-3H-benzimidazole-4-carboxylate

| Conditions | Yield |
|---|---|
| With 10% Pd/C; hydrogen; acetic acid In methanol at 20℃; under 760.051 Torr; | 93% |

| Conditions | Yield |
|---|---|
| With 10% Pd/C; hydrogen; acetic acid In methanol at 20℃; under 760.051 Torr; | 93% |

| Conditions | Yield |
|---|---|
| Stage #1: C30H32N6O5*C2H2O4 With potassium carbonate In ethyl acetate at 10 - 15℃; pH=6 - 7; Stage #2: orthocarbonic acid tetraethyl ester In acetic acid at 0 - 25℃; for 15h; | 92.3% |
-
-
78-09-1
orthocarbonic acid tetraethyl ester

-
-
136304-78-4
methyl 3-amino-2-[[(2'-cyanobiphenyl-4-yl)methyl]amino]benzoate

-
-
139481-44-0
methyl 1-[(2'-cyanobiphenyl-4-yl)methyl]-2-ethoxy-benzimidazole-7-carboxylate

| Conditions | Yield |
|---|---|
| With acetic acid at 80 - 90℃; for 0.666667h; | 91% |
| With acetic acid at 80℃; for 1h; | 86% |
| With acetic acid at 78 - 82℃; for 1 - 2h; Heating / reflux; Industry scale; | 84.8% |
| With acetic acid In toluene at 20℃; Reflux; |
-
-
78-09-1
orthocarbonic acid tetraethyl ester

-
-
88-74-4
2-nitro-aniline

-
-
22219-23-4
2-ethoxy-1H-benzo[d]imidazole

| Conditions | Yield |
|---|---|
| With 10% Pd/C; hydrogen; acetic acid In methanol at 20℃; under 760.051 Torr; | 91% |

| Conditions | Yield |
|---|---|
| With acetic acid at 80℃; for 4h; | 91% |
-
-
50-24-8
prednisolon

-
-
78-09-1
orthocarbonic acid tetraethyl ester

-
-
26129-79-3
prednisolone-17,21-diethylorthocarbonate

| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid In 1,4-dioxane at 22℃; for 17h; | 90% |
-
-
850154-78-8
2-cyano-3-methyl-benzenesulfonamide

-
-
78-09-1
orthocarbonic acid tetraethyl ester

-
-
937722-17-3
2-cyano-N-diethoxymethylene-3-methyl-benzenesulfonamide

| Conditions | Yield |
|---|---|
| at 160℃; | 90% |
-
-
78-09-1
orthocarbonic acid tetraethyl ester

-
-
6393-40-4
4-Amino 3-nitro-benzonitrile

-
-
952511-49-8
C10H9N3O

| Conditions | Yield |
|---|---|
| With 10% Pd/C; hydrogen; acetic acid In methanol at 20℃; under 760.051 Torr; | 90% |
-
-
22600-30-2
methyl 5-amino-2-furoate

-
-
78-09-1
orthocarbonic acid tetraethyl ester

-
-
110189-96-3
N-(5-methoxycarbonyl-2-furyl)imidocarbonate

| Conditions | Yield |
|---|---|
| With trifluoroacetic acid at 80℃; for 24h; | 88% |
-
-
78-09-1
orthocarbonic acid tetraethyl ester

-
-
169270-81-9
methyl 3-amino-2-<<(11-cyano-5-oxo-5H-dibenzocyclohepten-3-yl)methyl>amino>benzoate

-
-
169270-82-0
methyl 3-<(11-cyano-5-oxo-5H-dibenzocyclohepten-3-yl)methyl>-2-ethoxy-3H-benzimidazole-4-carboxylate

| Conditions | Yield |
|---|---|
| With acetic acid at 100℃; for 2h; | 88% |

| Conditions | Yield |
|---|---|
| With 10% Pd/C; hydrogen; acetic acid In methanol at 20℃; under 760.051 Torr; | 88% |
-
-
78-09-1
orthocarbonic acid tetraethyl ester

| Conditions | Yield |
|---|---|
| With sodium acetate In acetic acid | 87% |
-
-
78-09-1
orthocarbonic acid tetraethyl ester

-
-
37466-88-9
2,3-Diaminobenzoic acid,ethyl ester

-
-
577957-02-9
ethyl 2-ethoxy-3H-benzimidazole-4-carboxylate

| Conditions | Yield |
|---|---|
| With acetic acid at 0 - 20℃; for 3h; | 87% |
-
-
78-09-1
orthocarbonic acid tetraethyl ester

-
-
36692-49-6
Methyl 3,4-diaminobenzoate

| Conditions | Yield |
|---|---|
| With acetic acid at 0 - 20℃; for 4h; | 87% |
-
-
7677-24-9
trimethylsilyl cyanide

-
-
78-09-1
orthocarbonic acid tetraethyl ester

-
-
68714-37-4
triethoxyacetonitrile

| Conditions | Yield |
|---|---|
| tin(IV) chloride for 15h; Ambient temperature; | 86% |
-
-
625-72-9
(R)-3-hydroxybutyric acid

-
-
78-09-1
orthocarbonic acid tetraethyl ester

-
A
-
15890-55-8
β-valerolactone

| Conditions | Yield |
|---|---|
| In benzene | A n/a B 86% |

-
-
932034-83-8
4-(2-amino-4-methyl-phenylamino)-piperidine-1-carboxylic acid tert-butyl ester

-
-
78-09-1
orthocarbonic acid tetraethyl ester

-
-
932035-40-0
4-(2-ethoxy-5-methylbenzoimidazol-1-yl)-piperidine-1-carboxylic acid tert-butyl ester

| Conditions | Yield |
|---|---|
| With acetic acid at 20℃; for 4h; | 86% |
-
-
78-09-1
orthocarbonic acid tetraethyl ester

-
-
1258399-19-7
methyl {(3S)-6-[{(3R)-7-[(2-aminophenyl)amino]-2,3-dihydro-1-benzofuran-3-yl}(trifluoroacetyl)amino]-2,3-dihydro-1-benzofuran-3-yl}acetate

-
-
1258399-22-2
methyl [(3S)-6-{[(3R)-7-(2-ethoxy-1H-benzimidazol-1-yl)-2,3-dihydro-1-benzofuran-3-yl](trifluoroacetyl)amino}-2,3-dihydro-1-benzofuran-3-yl]acetate

| Conditions | Yield |
|---|---|
| Stage #1: orthocarbonic acid tetraethyl ester; methyl {(3S)-6-[{(3R)-7-[(2-aminophenyl)amino]-2,3-dihydro-1-benzofuran-3-yl}(trifluoroacetyl)amino]-2,3-dihydro-1-benzofuran-3-yl}acetate With acetic acid at 60℃; for 0.5h; Stage #2: With sodium hydrogencarbonate In water | 86% |
Tetraethyl orthocarbonate Specification
The Tetraethyl orthocarbonate, with the CAS registry number 78-09-1 and EINECS registry number 201-082-2, has the systematic name of (triethoxymethoxy)ethane. And the molecular formula of this chemical is C9H20O4. It is a kind of clear colorless liquid, and belongs to the following product categories: Acetals/Ketals/Ortho Esters; Organic Building Blocks; Oxygen Compounds. What's more, it should be stored in the refrigerator. In addition, it is used in the synthesis of medicine intermediate and cementing compound.
The physical properties of Tetraethyl orthocarbonate are as following: (1)ACD/LogP: 4.68; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): 4.68; (4)ACD/LogD (pH 7.4): 4.68; (5)ACD/BCF (pH 5.5): 2122.6; (6)ACD/BCF (pH 7.4): 2122.6; (7)ACD/KOC (pH 5.5): 8374.69; (8)ACD/KOC (pH 7.4): 8374.69; (9)#H bond acceptors: 4; (10)#H bond donors: 0; (11)#Freely Rotating Bonds: 8; (12)Polar Surface Area: 36.92 Å2; (13)Index of Refraction: 1.414; (14)Molar Refractivity: 50.69 cm3; (15)Molar Volume: 202.5 cm3; (16)Polarizability: 20.09×10-24cm3; (17)Surface Tension: 27.5 dyne/cm; (18)Density: 0.948 g/cm3; (19)Flash Point: 52.8 °C; (20)Enthalpy of Vaporization: 37.98 kJ/mol; (21)Boiling Point: 159.5 °C at 760 mmHg; (22)Vapour Pressure: 3.24 mmHg at 25°C.
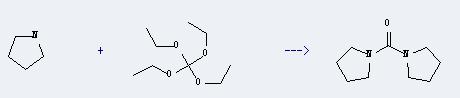
Uses of Tetraethyl orthocarbonate: It can react with pyrrolidine to produce 1,1'-carbonyl-bis-pyrrolidine. The reaction time is 5 days with heating, and the yield is about 39%.
You should be cautious while dealing with this chemical. It is a kind of flammable chemical which irritates eyes, respiratory system and skin. Therefore, you had better take the following instructions: Wear suitable protective clothing, gloves and eye/face protection, and in case of contacting with eyes, rinse immediately with plenty of water and seek medical advice; Keep away from sources of ignition - No smoking.
You can still convert the following datas into molecular structure:
(1)SMILES: O(CC)C(OCC)(OCC)OCC
(2)InChI: InChI=1/C9H20O4/c1-5-10-9(11-6-2,12-7-3)13-8-4/h5-8H2,1-4H3
(3)InChIKey: CWLNAJYDRSIKJS-UHFFFAOYAN
Related Products
- TETRAETHYL 1,1'-(1,1'-BIPHENYL-4,4'-DIYL)BIS(2,5-DIMETHYL-1 H-PYRROLE-3,4-DICARBOXYLATE)
- Tetraethyl 2,2-(1,4-phenylenedimethylidyne)bismalonate
- Tetraethyl ammonium chloride
- Tetraethyl butane-1,4-diylbis(phosphonate)
- TETRAETHYL GERMANE
- TETRAETHYL LEAD
- Tetraethyl methylenediphosphonate
- Tetraethyl orthocarbonate
- Tetraethyl orthosilicate
- TETRAETHYL PYROPHOSPHATE
- 78091-12-0
- 78092-53-2
- 78093-88-6
- 780-97-2
- 78101-74-3
- 78-10-4
- 78110-38-0
- 78111-17-8
- 78111-49-6
- 78114-46-2
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View