-
Name
Tetrakis(hydroxymethyl)phosphonium chloride
- EINECS 204-707-7
- CAS No. 124-64-1
- Article Data10
- CAS DataBase
- Density 1.341 g/mL at 25 °C
- Solubility water: >=10 g/100 mL at 20 °C
- Melting Point 154 °C
- Formula C4H12O4P.Cl
- Boiling Point 115°C/760mmHg
- Molecular Weight 190.564
- Flash Point > 96oC
- Transport Information UN 2810 6.1/PG 3
- Appearance Clear light pink or yellow-green solution
- Safety 22-26-36/37/39-45-60-61
- Risk Codes 21-25-38-41-42/43-51/53
-
Molecular Structure
-
Hazard Symbols
 T,
T, N
N
- Synonyms Phosphonium,tetrakis(hydroxymethyl)-, chloride (8CI,9CI);NSC 30698;Pyroset TKC;THPC;Tetrakis(hydroxymethyl)phosphochloride;Tetramethylolphosphonium chloride;
- PSA 94.51000
- LogP -4.19280
Synthetic route

| Conditions | Yield |
|---|---|
| Stage #1: formaldehyd With phosphorus; bis(tri-n-butyltin)oxide; 1,1'-azobis(1-cyanocyclohexanenitrile) In ethanol at 20℃; for 16h; Stage #2: With hydrogenchloride In 1,4-dioxane; ethanol at 20℃; for 4h; Catalytic behavior; Reagent/catalyst; Cooling with liquid nitrogen; | 80% |
| With hydrogenchloride; phosphorus hydrogen; water at 80℃; | |
| With hydrogenchloride; phosphan |
-
-
7647-01-0
hydrogenchloride

-
-
50-00-0
formaldehyd

-
-
688-73-3
tri-n-butyl-tin hydride

-
A
-
1461-22-9
tributyltin chloride

-
B
-
124-64-1
tetrakis(hydroxymethyl)phosphonium chloride

| Conditions | Yield |
|---|---|
| Stage #1: phosphorus; tri-n-butyl-tin hydride In toluene at 20℃; for 16h; Schlenk technique; Irradiation; Stage #2: formaldehyd In ethanol at 20℃; for 16h; Stage #3: hydrogenchloride In 1,4-dioxane; ethanol at 20℃; for 2h; Cooling with liquid nitrogen; | A 96% B 75% |


| Conditions | Yield |
|---|---|
| With hydrogenchloride In water passing PH3 in aq. formaldehyde soln. containing HCl at ambient temp.;; | |
| With hydrogenchloride In water passing PH3 in aq. formaldehyde soln. containing HCl at 80 °C;; | |
| With HCl In water passing PH3 in aq. formaldehyde soln. containing HCl at 80 °C;; | |
| With HCl In water passing PH3 in aq. formaldehyde soln. containing HCl at ambient temp.;; |

| Conditions | Yield |
|---|---|
| With water |

-
-
50-00-0
formaldehyd

-
-
2767-80-8
tris(hydroxymethyl)phosphine

-
-
124-64-1
tetrakis(hydroxymethyl)phosphonium chloride

| Conditions | Yield |
|---|---|
| With hydrogenchloride |

| Conditions | Yield |
|---|---|
| Stage #1: tetrakis(hydroxymethyl)phosphonium chloride With potassium hydroxide In methanol; water at 15 - 20℃; for 0.166667h; Stage #2: With zinc(II) chloride; sodium sulfite In methanol; water at 25℃; Reagent/catalyst; Solvent; | 98.3% |
| With triethylamine at 60℃; for 4h; Elimination; | 95% |
| With sodium hydroxide |

| Conditions | Yield |
|---|---|
| In ethanol at 20℃; for 2h; | 95% |
| In ethanol for 2h; |

| Conditions | Yield |
|---|---|
| In ethanol at 20℃; for 2h; | 100% |

| Conditions | Yield |
|---|---|
| In ethanol at 20℃; for 2h; | 91% |

| Conditions | Yield |
|---|---|
| In ethanol at 20℃; for 2h; | 85% |

| Conditions | Yield |
|---|---|
| In ethanol at 20℃; for 2h; | 97% |

| Conditions | Yield |
|---|---|
| In ethanol at 20℃; for 2h; | 97% |

| Conditions | Yield |
|---|---|
| In ethanol at 20℃; for 2h; | 94% |

| Conditions | Yield |
|---|---|
| In ethanol at 20℃; for 2h; | 92% |

| Conditions | Yield |
|---|---|
| In ethanol at 20℃; for 2h; | 91% |

| Conditions | Yield |
|---|---|
| In ethanol at 20℃; for 2h; | 90% |

| Conditions | Yield |
|---|---|
| In ethanol at 20℃; for 2h; | 97% |

| Conditions | Yield |
|---|---|
| In ethanol at 20℃; for 2h; | 93% |

| Conditions | Yield |
|---|---|
| In ethanol at 20℃; for 2h; | 85% |

| Conditions | Yield |
|---|---|
| In ethanol at 20℃; for 2h; | 87% |

| Conditions | Yield |
|---|---|
| In ethanol at 20℃; for 2h; | 83% |

| Conditions | Yield |
|---|---|
| In ethanol at 20℃; for 2h; | 83% |
-
-
124-64-1
tetrakis(hydroxymethyl)phosphonium chloride

-
-
74-88-4
methyl iodide

-
-
5958-52-1
[hydroxymethyl(methyl)phosphanyl]methanol

| Conditions | Yield |
|---|---|
| Stage #1: tetrakis(hydroxymethyl)phosphonium chloride With triethylamine at 20℃; for 18h; Stage #2: methyl iodide In tetrahydrofuran at -40 - 20℃; | 33% |

| Conditions | Yield |
|---|---|
| In methanol at 20℃; for 2h; | 72% |

| Conditions | Yield |
|---|---|
| Stage #1: tetrakis(hydroxymethyl)phosphonium chloride With triethylamine In N,N-dimethyl-formamide; toluene at 60℃; for 1h; Inert atmosphere; Stage #2: With octasulfur In N,N-dimethyl-formamide; toluene Inert atmosphere; | 94% |

-
-
124-64-1
tetrakis(hydroxymethyl)phosphonium chloride

| Conditions | Yield |
|---|---|
| Stage #1: tetrakis(hydroxymethyl)phosphonium chloride With potassium hydroxide In methanol Stage #2: potassium tetrachloropalladate(II) In methanol for 336h; Inert atmosphere; | 37% |
-
-
129826-46-6
4‐bromobutylferrocene

-
-
124-64-1
tetrakis(hydroxymethyl)phosphonium chloride

-
-
1599441-98-1
Fc(CH2)4P(CH2OH)2

| Conditions | Yield |
|---|---|
| Stage #1: tetrakis(hydroxymethyl)phosphonium chloride With potassium hydroxide In methanol for 1h; Schlenk technique; Inert atmosphere; Stage #2: 4‐bromobutylferrocene In methanol for 21h; Schlenk technique; Inert atmosphere; Reflux; Stage #3: With triethylamine In diethyl ether; water for 1h; Schlenk technique; Inert atmosphere; |
-
-
136237-36-0
ω-bromohexane ferrocene

-
-
124-64-1
tetrakis(hydroxymethyl)phosphonium chloride

-
-
1599442-01-9
Fc(CH2)6P(CH2OH)2

| Conditions | Yield |
|---|---|
| Stage #1: tetrakis(hydroxymethyl)phosphonium chloride With potassium hydroxide In methanol for 1h; Schlenk technique; Inert atmosphere; Stage #2: ω-bromohexane ferrocene In methanol; ethanol for 20.5h; Schlenk technique; Inert atmosphere; Reflux; Stage #3: With triethylamine In diethyl ether; water for 2h; Schlenk technique; Inert atmosphere; |

-
-
124-64-1
tetrakis(hydroxymethyl)phosphonium chloride

-
-
1599442-03-1
Fc(CH2)11P(CH2OH)2

| Conditions | Yield |
|---|---|
| Stage #1: tetrakis(hydroxymethyl)phosphonium chloride With potassium hydroxide In methanol for 1h; Schlenk technique; Inert atmosphere; Stage #2: 11-bromo-n-undecylferrocene In methanol for 20.5h; Schlenk technique; Inert atmosphere; Reflux; Stage #3: With triethylamine In diethyl ether; water for 2h; Schlenk technique; Inert atmosphere; |

-
-
124-64-1
tetrakis(hydroxymethyl)phosphonium chloride

-
-
1599442-18-8
RcCH2P(CH2OH)2

| Conditions | Yield |
|---|---|
| Stage #1: tetrakis(hydroxymethyl)phosphonium chloride With potassium hydroxide In methanol for 1h; Schlenk technique; Inert atmosphere; Stage #2: N-trimethyl-N-ruthenocenylammonium iodide In methanol for 20h; Schlenk technique; Inert atmosphere; Reflux; |
-
-
124-64-1
tetrakis(hydroxymethyl)phosphonium chloride

-
-
460-00-4
1-Bromo-4-fluorobenzene

-
A
-
18437-79-1
tri(4-fluorophenyl)phosphine oxide

| Conditions | Yield |
|---|---|
| With 3 A molecular sieve; potassium carbonate; [(o-Tol)2PC6H4CH2Pd(OAc)]2 In N,N-dimethyl acetamide at 130℃; for 17h; | A 10% B 40% |
| With 3 A molecular sieve; potassium carbonate; [(o-Tol)2PC6H4CH2Pd(OAc)]2 In N,N-dimethyl acetamide at 130℃; for 17h; | A 23 % Chromat. B 13 % Chromat. |


| Conditions | Yield |
|---|---|
| With sodium hydroxide In methanol; water at 50℃; for 2h; Elimination; | 100% |
| With barium carbonate In water | |
| With sodium hydroxide | |
| With sodium hydroxide; dihydrogen peroxide at 70 - 80℃; for 1h; | |
| With sodium hydroxide at 20℃; Rate constant; |
Tetrakis(hydroxymethyl)phosphonium chloride Chemical Properties
Molar mass: 155.1089 g/mol
Index of Refraction: n20/D 1.512
Boiling Point: 115°C/760mmHg
Vapour Pressure: 1.16 mmHg at 25°C
Melting point: 154 °C
Water solubility: >=10 g/100 mL at 20°C
Appearance: Clear slightly viscous,colorless to yellow liquid
Structure of Tetrakis(hydroxymethyl)phosphonium chloride(124-64-1):
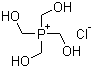
Synonyms of Tetrakis(hydroxymethyl)phosphonium chloride(124-64-1): THPC;pyrosettkc;Tetramethylolphosphonium chloride;Phosphonium,tetrakis(hydroxymethyl)-,chloride
Product Categories of Tetrakis(hydroxymethyl)phosphonium chloride(124-64-1): Phosphonium Compounds:Phosphonium Salts; Catalysis;Greener Alternatives
Tetrakis(hydroxymethyl)phosphonium chloride Toxicity Data With Reference
| 1. | otr-mus:emb 5000 ppm | JTEHD6 Journal of Toxicology and Environmental Health. 6 (1980),259. | ||
| 2. | otr-ham:kdy 5000 ppm | JTEHD6 Journal of Toxicology and Environmental Health. 6 (1980),259. | ||
| 3. | cyt-ham:lng 87 mg/L | GMCRDC Gann Monograph on Cancer Research. 27 (1981),95. | ||
| 4. | msc-ham:lng 5000 ppm | JTEHD6 Journal of Toxicology and Environmental Health. 6 (1980),259. | ||
| 5. | orl-rat LD50:161 mg/kg | NTPTR* National Toxicology Program Technical Report Series. (Research Triangle Park, NC 27709) No. NTP-TR-296 ,1987. | ||
| 6. | ipr-mus LDLo:125 mg/kg | CBCCT* “Summary Tables of Biological Tests“ National Research Council Chemical-Biological Coordination Center. 7 (1955),789. |
Tetrakis(hydroxymethyl)phosphonium chloride Consensus Reports
Tetrakis(hydroxymethyl)phosphonium chloride Safety Profile
Hazard Codes: T
 ,N
,N
Risk Statements:
21: Harmful in contact with skin
25: Toxic if swallowed
38: Irritating to the skin
41: Risk of serious damage to eyes
42: May cause sensitization by inhalation
43: May cause sensitization by skin contact
51: Toxic to aquatic organisms
53: May cause long-term adverse effects in the aquatic environment
Safety Statements:
22: Do not breathe dust
26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice
36: Wear suitable protective clothing
37: Wear suitable gloves
39: Wear eye/face protection
45: In case of accident or if you feel unwell, seek medical advice immediately (show label where possible)
60: This material and/or its container must be disposed of as hazardous waste
61: Avoid release to the environment. Refer to special instructions safety data sheet
Related Products
- Tetrakis(hydroxymethyl)phosphonium acetate
- Tetrakis(hydroxymethyl)phosphonium chloride
- Tetrakis(hydroxymethyl)phosphonium chloride urea polymer
- TETRAKIS(HYDROXYMETHYL)PHOSPHONIUM NITRATE
- Tetrakis(hydroxymethyl)phosphonium phosphate
- Tetrakis(hydroxymethyl)phosphonium sulfate
- Tetrakis(hydroxymethyl)phosphonium sulfate urea polymer
- 1246454-25-0
- 124646-10-2
- 124-65-2
- 1246525-60-9
- 124655-09-0
- 1246551-22-3
- 124655-17-0
- 1246552-49-7
- 1246553-27-4
- 124655-63-6
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View