-
Name
Chloric acid
- EINECS
- CAS No. 7790-93-4
- Article Data161
- CAS DataBase
- Density 1.2 g/mL at 25oC
- Solubility very soluble H2O [CRC10]
- Melting Point <-20℃ [CRC10]
- Formula ClH O3
- Boiling Point >100oC
- Molecular Weight 84.4591
- Flash Point
- Transport Information
- Appearance
- Safety A poison. A strong irritant by ingestion and inhalation. Dangerous fire hazard; ignites organic matter upon contact. A very powerful oxidizing agent. Violent or explosive reaction with oxidizable materials. Aqueous solutions decompose explosively during evaporation. Solutions greater than 40% are unstable. Reacts violently with NH3, Sb, Sb2S3, As2S3, Bi, CuS, PHI4, SnS2, SnS. Reaction with cellulose causes ignition after a delay period. Dangerous reaction with metal sulfides and metal chlorides (e.g., incandescent reaction with antimony trisulfide, arsenic trisulfide, tin(II)sulfide, tin(IV) sulfide, explosion on contact with copper sulfide). Reaction with metals (e.g., antimony, bismuth, iron) forms explosive products. When heated to decomposition it emits toxic fumes of Cl−. See also CHLORATES and CHLORINE.
- Risk Codes R8; R34
-
Molecular Structure
- Hazard Symbols Toxic by ingestion and inhalation. Strong oxidizer, ignites organic materials on contact.
- Synonyms Chloricacid (HClO3)
- PSA 54.37000
- LogP 0.27510
Synthetic route
-
-
14989-30-1
hypochloric acid

-
-
13898-47-0
hypochloric acid

-
A
-
7647-01-0
hydrogenchloride

-
B
-
10049-04-4, 25052-55-5
chlorine dioxide

-
C
-
7732-18-5
water

-
D
-
7790-93-4
chloric acid

| Conditions | Yield |
|---|---|
| In water reaction of HClO2 and HClO in weakly acidic or neutral soln. at ambient temp.;; removing of ClO2 with air;; | A n/a B 97% C n/a D n/a |
| In water reaction of HClO2 and HClO in aq. soln. at ambient temp.; influence of pH;; | |
| In water reaction of HClO2 and HClO in weakly acidic or neutral soln. at ambient temp.; acceleration on low concn. of ClO2(1-); no influence of ClO3(1-);; | |
| In water reaction of HClO2 and HClO in weakly acidic or neutral soln. at ambient temp.;; | |
| In water Kinetics; reaction of HClO2 and HClO in aq. soln. at ambient temp.;; |


| Conditions | Yield |
|---|---|
| With hydrogenchloride; platinum In not given byproducts: HCl; reaction by use of a 0.05 n-HCl soln. in presence of colloidal Pt;; | A 0% B n/a C 0% |


| Conditions | Yield |
|---|---|
| manganese(ll) chloride In water Kinetics; hydrolysis witout action of light with 0.02 m MnCl2 at 91°C;; | |
| In water Irradiation (UV/VIS); weak diffuse light; first HClO is formed; then HClO3;; | |
| In water hydrolysis without action of light at 91°C: thermochemical data;; |


| Conditions | Yield |
|---|---|
| In gas common condensing of educts;; |

-
-
10049-04-4, 25052-55-5
chlorine dioxide

-
A
-
7647-01-0
hydrogenchloride

-
B
-
7601-90-3
perchloric acid

-
C
-
7790-93-4
chloric acid

| Conditions | Yield |
|---|---|
| In water Irradiation (UV/VIS); decompn. in sunlight;; | |
| In water Irradiation (UV/VIS); reaction in sunlight;; | |
| In water Irradiation (UV/VIS); reaction in sunlight;; |


| Conditions | Yield |
|---|---|
| With water In not given Electrolysis; | |
| With H2O In not given Electrolysis; |

| Conditions | Yield |
|---|---|
| With water; chlorine In water Kinetics; byproducts: CH3CO2H; in buffered soln. (pH at begin 2.11 to 2.35) at 16.-+.1°C;; |

| Conditions | Yield |
|---|---|
| With chlorine In water oxidation in fuming HNO3 at 15 - 18°C about 1 month;; | A 0% B n/a |

| Conditions | Yield |
|---|---|
| With chlorine In not given 1 mo, at 15-18°C, in concd. HNO3; | |
| With Cl2 In not given 1 mo, at 15-18°C, in concd. HNO3; |

| Conditions | Yield |
|---|---|
| With bismuth In water frequently explosion, especially at dissolution of bismuth powder in hot HClO4;; | |
| With bismuth In water frequently explosion;; | |
| With Bi In water frequently explosion, especially at dissolution of bismuth powder in hot HClO4;; |

| Conditions | Yield |
|---|---|
| With water; chlorine In not given byproducts: HNO3; in the cold;; pptn. of AgCl;; |
-
-
7647-01-0
hydrogenchloride

-
A
-
7601-90-3
perchloric acid

-
B
-
80937-33-3
oxygen

-
C
-
7782-50-5
chlorine

-
D
-
7790-93-4
chloric acid

| Conditions | Yield |
|---|---|
| In water Electrolysis; anodic oxidation;; | |
| In water Electrolysis; electrolysis of dild. HCl; depending on concn. of HCl and temp.;; |


| Conditions | Yield |
|---|---|
| With H2O In water Electrolysis; at boiling heat; products fromed at anode;; |


| Conditions | Yield |
|---|---|
| With H2O In water Electrolysis; 1/30 n- to n-soln. of aq. HCl; HClO3 is formed beside small amounts of H2O2;; |
-
-
7647-01-0
hydrogenchloride

-
A
-
7722-84-1
dihydrogen peroxide

-
B
-
14989-30-1
hypochloric acid

-
C
-
7790-93-4
chloric acid

| Conditions | Yield |
|---|---|
| With H2O In water Electrolysis; 1/30 to 1 n-HCl is used; traces of HClO is formed at current densitys of 0.02, 0.2 and 2 amp/cm^2;; | A n/a B < 1 C n/a |

-
-
14989-30-1
hypochloric acid

-
A
-
7601-90-3
perchloric acid

-
B
-
80937-33-3
oxygen

-
C
-
7790-93-4
chloric acid

| Conditions | Yield |
|---|---|
| In water decompn. of aq. HClO;; | A 0% B n/a C n/a |

-
-
7782-50-5
chlorine

-
A
-
7601-90-3
perchloric acid

-
B
-
80937-33-3
oxygen

-
C
-
14989-30-1
hypochloric acid

-
D
-
7790-93-4
chloric acid

| Conditions | Yield |
|---|---|
| In water decompn. of chlorine water;; | A 0% B n/a C n/a D n/a |


| Conditions | Yield |
|---|---|
| In water Irradiation (UV/VIS); | |
| In water Irradiation (UV/VIS); |

-
-
7732-18-5
water

-
-
80937-33-3
oxygen

-
-
7782-50-5
chlorine

-
-
10028-15-6
ozone

-
A
-
7601-90-3
perchloric acid

-
B
-
7790-93-4
chloric acid

| Conditions | Yield |
|---|---|
| In neat (no solvent) Irradiation (UV/VIS); reaction by exposure of the mixture with radiation of a wave length of 3650 Angstrom; photochemical process;; |


| Conditions | Yield |
|---|---|
| With water In water acidifying;; |

-
-
7778-54-3
calcium hypochlorite

-
A
-
7647-01-0
hydrogenchloride

-
B
-
7782-50-5
chlorine

-
C
-
7790-93-4
chloric acid

| Conditions | Yield |
|---|---|
| With carbon dioxide In water byproducts: H2O; | |
| With carbon dioxide In water byproducts: H2O; |

-
-
10049-04-4, 25052-55-5
chlorine dioxide

-
-
7732-18-5
water

-
A
-
13898-47-0
hypochloric acid

-
B
-
7790-93-4
chloric acid

| Conditions | Yield |
|---|---|
| With dihydrogen peroxide In water hydrolysis of ClO2 in presence of H2O2;; | A n/a B 0% |
| With H2O2 In water |


| Conditions | Yield |
|---|---|
| With sulfur In water reaction of solid chlorate with S in presence of water;; | |
| With S In water reaction of solid chlorate with S in presence of water;; |

| Conditions | Yield |
|---|---|
| In water Irradiation (UV/VIS); aq. Cl2 soln. irridiated until colorless;; not isolated, identified as KClO3;; | |
| In water Irradiation (UV/VIS); aq. Cl2 soln. irridiated until colorless;; not isolated, identified as KClO3;; |

| Conditions | Yield |
|---|---|
| In water traces of ClO2 react in the dark;; | |
| With water In water | A 0% B 0% |
| In water traces of ClO2 react in the dark;; | |
| With H2O In water | A 0% B 0% |
-
-
10049-04-4, 25052-55-5
chlorine dioxide

-
A
-
7647-01-0
hydrogenchloride

-
B
-
13898-47-0
hypochloric acid

-
C
-
7790-93-4
chloric acid

| Conditions | Yield |
|---|---|
| In water very slow reaction;; | |
| In water very slow reaction;; |


| Conditions | Yield |
|---|---|
| With water; Nitrite In water byproducts: NO3(1-), H(1+); | |
| With NO2(1-); H2O In water byproducts: NO3(1-), H(1+); |
-
-
10049-04-4, 25052-55-5
chlorine dioxide

-
-
7790-93-4
chloric acid

| Conditions | Yield |
|---|---|
| With phosphoric acid In water Electrolysis; 12% aq. phosphoric acid is used; current density: 0.0125 to 0.0415 A/cm^2; at 8 to 10°C;; | |
| With sodium hypochlorite; water In water byproducts: NaCl; neutral soln.;; | |
| With NaClO; H2O In water byproducts: NaCl; neutral soln.;; | |
| With phosphoric acid In water Electrolysis; 12% aq. phosphoric acid is used; current density: 0.0125 to 0.0415 A/cm^2; at 8 to 10°C;; |

| Conditions | Yield |
|---|---|
| With water; platinum In not given decomposition in presence of platinated Pt at roomtemperature;; | |
| With water; chloride In not given Kinetics; decomposition at 0-60°C in presence of excess Cl-ions; mechanism discussed;; | |
| With water; chlorine In water in the dark at 18°C;; |

| Conditions | Yield |
|---|---|
| In water Irradiation (UV/VIS); | |
| In water byproducts: O; Irradiation (UV/VIS); reaction in sunlight in few hours, in diffuse light in several months;; | |
| In water Irradiation (UV/VIS); | |
| In water byproducts: O; Irradiation (UV/VIS); reaction in sunlight in few hours, in diffuse light in several months;; |

-
-
7790-93-4
chloric acid

| Conditions | Yield |
|---|---|
| In methanol; chloroform | 99.8% |
-
-
7782-99-2
sulphurous acid

-
-
7790-93-4
chloric acid

-
A
-
7664-93-9
sulfuric acid

-
B
-
10049-04-4, 25052-55-5
chlorine dioxide

-
C
-
7732-18-5
water

| Conditions | Yield |
|---|---|
| With air; sulfur dioxide In sulfuric acid byproducts: HClO, HCl, Cl2; Holst-process;; | A n/a B 90% C n/a |

-
-
131029-92-0
1-[2-[[2-(3-Chlorophenyl)-2-(2-methylphenyl)ethenyl]oxy]ethyl]-1,2,5,6-tetrahydro-3-pyridine carboxylic methyl ester

-
-
7790-93-4
chloric acid

| Conditions | Yield |
|---|---|
| With sodium hydroxide In ethanol; dichloromethane | 55% |
-
-
141125-02-2
2-(6-acetoxy-2-naphthoylamino)-4-butylphenol

-
-
7790-93-4
chloric acid

-
-
95-50-1
1,2-dichloro-benzene

-
-
141125-03-3
2-(6-hydroxy-2-naphthyl)-5-butylbenzooxazole

| Conditions | Yield |
|---|---|
| With potassium hydroxide In ethanol; water | 42.4% |

-
-
7790-93-4
chloric acid

-
-
10049-04-4, 25052-55-5
chlorine dioxide

| Conditions | Yield |
|---|---|
| With water Product distribution / selectivity; Polarised UV irradiation; | |
| With V(3+) | |
| With sulphurous acid In sulfuric acid byproducts: H2O4, H2O; passing SO2 (5 - 10 vol%) dild. with air or N2 over 3 - 4 mol/l NaClO3 in 7 - 8 val/l H2SO4;; |
-
-
174456-10-1
6-chloro-4-nitro-indoline-2,3-dione

-
-
7790-93-4
chloric acid

-
-
174456-26-9
4-chloro-6-nitro-anthranilic acid

| Conditions | Yield |
|---|---|
| With dihydrogen peroxide In sodium hydroxide |

-
-
188905-29-5
(2R,3R,4R)-3,4-dibenzyloxy-2-benzyloxymethyl-1-butylpyrrolidine

-
-
7790-93-4
chloric acid

-
-
188905-30-8
(2R,3R,4R)-1-Butyl-3,4-dihydroxy-2-hydroxymethylpyrrolidine, hydrochloride

| Conditions | Yield |
|---|---|
| palladium-carbon In ethanol |

-
-
626-55-1
3-Bromopyridine

-
-
150-19-6
O-methylresorcine

-
-
7790-93-4
chloric acid

-
-
73617-12-6
3-(3-methoxyphenoxy)pyridine

| Conditions | Yield |
|---|---|
| With potassium hydroxide; sodium hydroxide; copper In ethanol; water; toluene |


-
-
596-43-0
bromo-triphenyl-methane

-
-
7790-93-4
chloric acid

-
-
55444-53-6
4-cyclopentyl-1-iodo-3-triphenylmethoxy-1-trans-butene

| Conditions | Yield |
|---|---|
| In pyridine; hexane; water |


| Conditions | Yield |
|---|---|
| In water distg. of aq. HClO3;; | |
| With dihydrogen peroxide In water | 0% |
| With sulfuric acid In not given during 2 months;; | >99 |

| Conditions | Yield |
|---|---|
| In sulfuric acid |

| Conditions | Yield |
|---|---|
| In further solvent(s) 2n HClO3 soln.;; | |
| In further solvent(s) frim finely powdered As and 2n HClO3 soln.;; |
-
-
7790-93-4
chloric acid

-
-
12775-96-1, 15158-11-9, 15721-63-8, 16941-75-6, 17493-86-6, 19498-52-3, 20499-83-6, 20499-84-7, 20499-85-8, 20499-86-9, 20573-10-8, 20573-11-9, 21595-51-7, 21595-52-8, 22206-52-6, 26445-28-3, 28959-95-7, 37362-93-9, 39417-05-5, 54603-16-6, 54603-23-5, 54603-32-6, 54603-40-6, 54603-48-4, 54603-81-5, 54603-89-3, 56316-56-4, 95985-91-4, 122297-32-9, 7440-50-8
copper

| Conditions | Yield |
|---|---|
| In water at ambient temp. using 1 val/l HClO3-soln.;; | |
| In water byproducts: Cu-chloride, Cu-chlorate; at ambient temp. using a soln. of HClO3 with a concn. of 1 val/l;; |

| Conditions | Yield |
|---|---|
| In further solvent(s) Fe is dissolved by chloric acid in form of Fe(3+);; | |
| In water iron wire; highly exothermic reaction; at moderate temp. neither free chlorine nor lower oxygenic chlorine acids nor oxides of chlorine will form;; | |
| In further solvent(s) Fe is dissolved by chloric acid in form of Fe(3+);; |

| Conditions | Yield |
|---|---|
| In not given formation of H2 is hardly noticeable;; |

| Conditions | Yield |
|---|---|
| In hydrogenchloride dissolving finely powdered Te in mixture of aq. HCl and HClO3;; | |
| In water byproducts: Cl2; HNO3(50%) was added to Te, 1/4 of the HClO3 soln. was added, formed soln. was shaked, the soln. warmed up under Cl2 generation, the rest of HClO3 was added part by part, soln. was kept at reflux for 30 min, reduced in volume, cooled;; recrystn. from hot H2O, traces of Cl(1-) were removed as AgCl;; | |
| In hydrogenchloride byproducts: Cl2; addn. of part of aq. HClO3 soln. (from Ba(ClO3)2*H2O and concd. H2SO4) to Te moistened with concd. aq. HCl; no cooling because of formation of TeO2; further addn. of aq. HClO3 soln. in greater portions into boiling soln., shaking;; evapn.; crystn. on cooling with ice; washing with alc. and ether; drying in vac. desiccator; further pptn. on addn. of alc. to mother lye;; | >90 |
| In hydrogenchloride byproducts: Cl2; addn. of part of aq. HClO3 soln. (from Ba(ClO3)2*H2O and concd. H2SO4) to Te moistened with concd. aq. HCl; no cooling because of formation of TeO2; further addn. of aq. HClO3 soln. in greater portions into boiling soln., shaking;; recrystn. from water fro several times;; | |
| In water byproducts: Cl2; HNO3(50%) was added to Te, 1/4 of the HClO3 soln. was added, formed soln. was shaked, the soln. warmed up under Cl2 generation, the rest of HClO3 was added part by part, soln. was kept at reflux for 30 min, reduced in volume, cooled;; recrystn. from hot H2O, traces of Cl(1-) were removed as AgCl;; |

| Conditions | Yield |
|---|---|
| formation of H2 and redn. of chloric acid depends on concn. of acid and temp.;; | |
| In not given | |

| Conditions | Yield |
|---|---|
| In not given | |
| In not given |

| Conditions | Yield |
|---|---|
| With nitric acid In water byproducts: Cl2; dissolving of a suspension of Se in H2O by addn. of HNO3; concg.; addn. of HClO3; evapn.; concn. on passing of air through the soln. under reduced pressure; concn. at 150°C under reduced pressure;; diln. with H2O; filtration; concn. in vac.;; | 90-95 |
| In not given | |
| With HNO3 In water byproducts: Cl2; dissolving of a suspension of Se in H2O by addn. of HNO3; concg.; addn. of HClO3; evapn.; concn. on passing of air through the soln. under reduced pressure; concn. at 150°C under reduced pressure;; diln. with H2O; filtration; concn. in vac.;; | 90-95 |
| In not given |
CHLORIC ACID Chemical Properties
Molecular Formula:ClHO3
Molecular Weight:84.46
Properties:Colorless solution. Fairly stable in cold H2O up to 30%. Strong oxidant, stable as alkali metal salts. Mp: <20°C, bp: decomp @ 40°C d: 1.282 @ 14.2°C.
Occurrence:Only in aqueous solution.
Molecular Structure:
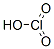
CHLORIC ACID Consensus Reports
CHLORIC ACID Safety Profile
CHLORIC ACID (7790-93-4) has hazard symbols: Toxic by ingestion and inhalation. Strong oxidizer, ignites organic materials on contact.
CHLORIC ACID Standards and Recommendations
DOT Classification: 5.1; Label: Oxidizer
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View