-
Name
1,2,4-Trichlorobenzene
- EINECS 204-428-0
- CAS No. 120-82-1
- Article Data140
- CAS DataBase
- Density 1.448 g/cm3
- Solubility Insoluble in water
- Melting Point 16 °C(lit.)
- Formula C6H3Cl3
- Boiling Point 211.4 °C at 760 mmHg
- Molecular Weight 181.449
- Flash Point 126.7 °C
- Transport Information UN 2321 6.1/PG 3
- Appearance colorless liquid
- Safety 23-37/39-60-61-45-36/37-16
- Risk Codes 22-38-50/53-52/53-39/23/24/25-23/24/25-11
-
Molecular Structure
-
Hazard Symbols
 Xn,
Xn,  N,
N,  Xi,
Xi,  T,
T,  F
F
- Synonyms 1,2,4-Trichlorobenzol;1,2,5-Trichlorobenzene;1,3,4-Trichlorobenzene;HostetexL-PEC;NSC 406697;unsym-Trichlorobenzene;
- PSA 0.00000
- LogP 3.64680
Synthetic route
-
-
634-90-2
1,2,3,5-tetrachlorobenzene

-
A
-
108-70-3
1,3,5-trichlorobenzene

-
B
-
120-82-1
1,2,4-Trichlorobenzene

| Conditions | Yield |
|---|---|
| With radical anion of p,p'-di-tert-butylbiphenyl In tetrahydrofuran at -25℃; Irradiation; | A 2.8% B 97.2% |

-
-
120-82-1
1,2,4-Trichlorobenzene

| Conditions | Yield |
|---|---|
| In dimethyl sulfoxide Ambient temperature; | 97% |

-
-
120-82-1
1,2,4-Trichlorobenzene

| Conditions | Yield |
|---|---|
| In dimethyl sulfoxide Ambient temperature; | 93% |
-
-
58-89-9
LINDANE

-
A
-
108-70-3
1,3,5-trichlorobenzene

-
B
-
120-82-1
1,2,4-Trichlorobenzene

-
C
-
87-61-6
1,2,3-trichlorobenzene

| Conditions | Yield |
|---|---|
| With ammonia at 25℃; Product distribution; Further Variations:; Reagents; Dehalogenation; | A 5% B 87% C 8% |
| at 25℃; pH=8.32; Kinetics; Product distribution; Further Variations:; pH-values; | |
| With (5,10,15,20-tetrakis(pentafluorophenyl)porphyrinato)iron(III) chloride; tetrabutylammonium perchlorate In N,N-dimethyl-formamide for 4h; Electrolysis; |


| Conditions | Yield |
|---|---|
| With Dichlorophenylphosphine In various solvent(s) at 170℃; for 5h; | 86% |
-
-
95-94-3
1,2,4,5-tetrachlorobenzene

-
A
-
106-46-7
para-dichlorobenzene

-
B
-
95-50-1
1,2-dichloro-benzene

-
C
-
541-73-1
1,3-Dichlorobenzene

-
D
-
120-82-1
1,2,4-Trichlorobenzene

| Conditions | Yield |
|---|---|
| With carbon dioxide; tetrabutylammomium bromide In N,N-dimethyl-formamide at -5 - 0℃; Dehalogenation; Electrochemical reaction; Further byproducts given; | A 9% B 1% C 2% D 8% |
| With [2,2]bipyridinyl; nickel dichloride; zinc In water; N,N-dimethyl-formamide at 80℃; for 6h; Title compound not separated from byproducts; | A 15.2 % Spectr. B 5.8 % Spectr. C 3.9 % Spectr. D 66.0 % Spectr. |

-
-
110-86-1
pyridine

-
-
319-84-6
alpha-hexachlorocyclohexane

-
A
-
108-70-3
1,3,5-trichlorobenzene

-
B
-
120-82-1
1,2,4-Trichlorobenzene

-
C
-
87-61-6
1,2,3-trichlorobenzene

-
-
91-22-5
quinoline

-
-
319-84-6
alpha-hexachlorocyclohexane

-
A
-
108-70-3
1,3,5-trichlorobenzene

-
B
-
120-82-1
1,2,4-Trichlorobenzene

-
C
-
87-61-6
1,2,3-trichlorobenzene

| Conditions | Yield |
|---|---|
| at 105 - 110℃; |


| Conditions | Yield |
|---|---|
| Diazotization; |
-
-
64-17-5
ethanol

-
-
319-86-8
delta-lindane

-
A
-
108-70-3
1,3,5-trichlorobenzene

-
B
-
120-82-1
1,2,4-Trichlorobenzene

-
C
-
87-61-6
1,2,3-trichlorobenzene

| Conditions | Yield |
|---|---|
| Product distribution; |

-
-
64-17-5
ethanol

-
-
58-89-9
LINDANE

-
A
-
108-70-3
1,3,5-trichlorobenzene

-
B
-
120-82-1
1,2,4-Trichlorobenzene

-
C
-
87-61-6
1,2,3-trichlorobenzene

| Conditions | Yield |
|---|---|
| Product distribution; |

-
-
64-17-5
ethanol

-
-
319-85-7
beta-hexachlorocyclohexane

-
A
-
108-70-3
1,3,5-trichlorobenzene

-
B
-
120-82-1
1,2,4-Trichlorobenzene

-
C
-
87-61-6
1,2,3-trichlorobenzene

| Conditions | Yield |
|---|---|
| Product distribution; |

-
-
64-17-5
ethanol

-
-
319-84-6
alpha-hexachlorocyclohexane

-
A
-
108-70-3
1,3,5-trichlorobenzene

-
B
-
120-82-1
1,2,4-Trichlorobenzene

-
C
-
87-61-6
1,2,3-trichlorobenzene

| Conditions | Yield |
|---|---|
| Product distribution; |

-
-
608-73-1, 119911-70-5
1,2,3,4,5,6-hexachlorocyclohexane

-
-
120-82-1
1,2,4-Trichlorobenzene

| Conditions | Yield |
|---|---|
| With kieselguhr at 295℃; | |
| With aluminium trichloride at 160℃; | |
| With nickel diacetate at 305℃; |

| Conditions | Yield |
|---|---|
| at 360 - 460℃; Leiten ueber Kupfer-Verbindungen enthaltende Katalysatoren; | |
| at 300 - 400℃; Leiten ueber Palladium/Aluminiumoxid; |

| Conditions | Yield |
|---|---|
| With phosphorus pentachloride at 200 - 210℃; |

| Conditions | Yield |
|---|---|
| With thionyl chloride at 160 - 180℃; im Rohr; | |
| With thionyl chloride at 160 - 180℃; im Rohr; |

| Conditions | Yield |
|---|---|
| With copper dichloride |

| Conditions | Yield |
|---|---|
| With potassium hydroxide unter Stickstoff; |
-
-
68-12-2, 33513-42-7
N,N-dimethyl-formamide

-
-
120-82-1
1,2,4-Trichlorobenzene

-
-
4659-47-6
2,3,6-trichlorobenzaldehyde

| Conditions | Yield |
|---|---|
| Stage #1: 1,2,4-Trichlorobenzene With n-butyllithium In tetrahydrofuran Stage #2: N,N-dimethyl-formamide In tetrahydrofuran at -65℃; for 1.5h; | 100% |

| Conditions | Yield |
|---|---|
| With hydrogen | 100% |


| Conditions | Yield |
|---|---|
| 99.6% |

| Conditions | Yield |
|---|---|
| With potassium phosphate; palladium bis(dibenzylideneacetone)palladium(0); catacxium A In toluene at 110℃; for 24h; Suzuki-Miyaura Coupling; Inert atmosphere; Sealed tube; | 99% |
| With potassium phosphate; [NiCl(Ph2PCH2CH2OH)2(H2O)]Cl In isopropyl alcohol at 80℃; for 24h; Suzuki coupling reaction; | 58% |

| Conditions | Yield |
|---|---|
| Stage #1: tert-butyl 2-hydroxyacetate With potassium ethoxide at 70℃; Stage #2: 1,2,4-Trichlorobenzene at 120℃; | 99% |
-
-
5426-48-2
n-heptyl glycolate

-
-
120-82-1
1,2,4-Trichlorobenzene

-
-
1917-96-0
n-heptyl 2,4-dichlorophenoxyacetate

| Conditions | Yield |
|---|---|
| Stage #1: n-heptyl glycolate With magnesium methanolate at 90℃; Stage #2: 1,2,4-Trichlorobenzene at 140℃; | 99% |
-
-
37160-54-6
di-n-hexyl glycolate

-
-
120-82-1
1,2,4-Trichlorobenzene

-
-
1917-95-9
n-hexyl 2,4-dichlorophenoxyacetate

| Conditions | Yield |
|---|---|
| Stage #1: di-n-hexyl glycolate With calcium ethoxide at 80℃; Stage #2: 1,2,4-Trichlorobenzene at 130℃; | 98.7% |
-
-
120-82-1
1,2,4-Trichlorobenzene

-
-
13075-01-9
1,2,4-tribromo-3,5,6-trichlorobenzene

| Conditions | Yield |
|---|---|
| With aluminum tri-bromide; bromine for 3h; Heating; | 98% |
| With aluminium trichloride; bromine | |
| With bromine; iron at 134.84 - 139.84℃; for 1h; | |
| With bromine; iron at 135℃; for 1h; |
-
-
5720-07-0
4-methoxyphenylboronic acid

-
-
120-82-1
1,2,4-Trichlorobenzene

-
-
136612-96-9
4,4″-dimethoxy-4′-(4-methoxyphenyl)-1,1′:2′,1″-terphenyl

| Conditions | Yield |
|---|---|
| With potassium phosphate; palladium bis(dibenzylideneacetone)palladium(0); catacxium A In toluene at 110℃; for 24h; Suzuki-Miyaura Coupling; Inert atmosphere; Sealed tube; | 98% |
-
-
75-69-4
trichlorofluoromethane

-
-
120-82-1
1,2,4-Trichlorobenzene

-
-
56148-83-5
2,4,5-trichloro-1-(trifluoromethyl)benzene

| Conditions | Yield |
|---|---|
| With aluminium trichloride 1a) 8 h, r.t., 1b) 16 h, -10 deg C; | 97% |

| Conditions | Yield |
|---|---|
| Stage #1: 1,2,4-Trichlorobenzene With sulfuric acid; potassium nitrate at 0 - 50℃; for 3h; Stage #2: With sodium hydroxide In water at 0℃; | 96% |
| With sodium nitrate; sulfuric acid at 40℃; for 3h; | 94% |
| With nitric acid In water at 0 - 20℃; for 3h; | 79% |
| With nitric acid | |
| durch Nitrierung; |

| Conditions | Yield |
|---|---|
| In various solvent(s) at 180℃; for 20h; | 96% |
| In various solvent(s) at 180℃; for 20h; Product distribution; Mechanism; var. temp.; var. solvents; other subst. chlorobenzenes; other thiolates; |
-
-
5720-06-9
2-Methoxyphenylboronic acid

-
-
120-82-1
1,2,4-Trichlorobenzene

-
-
136612-97-0
2,2″-dimethoxy-4'-(2-methoxyphenyl)-1,1':2′,1″-terphenyl

| Conditions | Yield |
|---|---|
| With potassium phosphate; palladium bis(dibenzylideneacetone)palladium(0); catacxium A In toluene at 110℃; for 24h; Suzuki-Miyaura Coupling; Inert atmosphere; Sealed tube; | 95% |

| Conditions | Yield |
|---|---|
| With boric acid | A 94.1% B n/a |


| Conditions | Yield |
|---|---|
| With hydrogen; palladium on activated charcoal; Aliquat 336 In water at 50℃; for 0.5h; | 94% |
| With palladium on activated charcoal; hydrogen at 24.84℃; for 10h; | 70% |
| With Ni[1,10-phenanthroline]2(PF6)2; water; zinc at 70℃; for 7h; Ionic liquid; | 42.2% |
-
-
123324-71-0
4-tert-butylphenylboronic acid

-
-
120-82-1
1,2,4-Trichlorobenzene

-
-
1392481-12-7
4,4″-di-tert-butyl-4′-(4-(tert-butyl)phenyl)-1,1′:2′,1″-terphenyl

| Conditions | Yield |
|---|---|
| With potassium phosphate; palladium bis(dibenzylideneacetone)palladium(0); catacxium A In toluene at 110℃; for 24h; Suzuki-Miyaura Coupling; Inert atmosphere; Sealed tube; | 94% |
-
-
10365-98-7
3-methoxyphenylboronic acid

-
-
120-82-1
1,2,4-Trichlorobenzene

-
-
639475-66-4
3,3''-dimethoxy-4'-(3-methoxyphenyl)-1,1':2',1''-terphenyl

| Conditions | Yield |
|---|---|
| With potassium phosphate; palladium bis(dibenzylideneacetone)palladium(0); catacxium A In toluene at 110℃; for 24h; Suzuki-Miyaura Coupling; Inert atmosphere; Sealed tube; | 93% |

| Conditions | Yield |
|---|---|
| With sodium ethanolate; sodium phenylsulfonate at 190℃; under 21752.2 Torr; for 10h; Reagent/catalyst; Temperature; Pressure; Autoclave; Inert atmosphere; | 92.6% |
| With sodium hydroxide In ethylene glycol at 180℃; for 3h; Temperature; | 70.4% |
| With sodium hydroxide at 180 - 190℃; |
-
-
1639-09-4
heptanethiol

-
-
120-82-1
1,2,4-Trichlorobenzene

-
A
-
10496-16-9
diheptyldisulfide

-
B
-
89165-37-7
Heptyl 2,5-dichlorophenyl sulfide

| Conditions | Yield |
|---|---|
| With potassium hydroxide; (Tricyclohexyl-n-dodecyl)phosphonium bromide In toluene at 50℃; for 3h; | A n/a B 92% |

-
-
68-12-2, 33513-42-7
N,N-dimethyl-formamide

-
-
120-82-1
1,2,4-Trichlorobenzene

-
A
-
35696-87-8
2,4,5-trichlorobenzaldehyde

-
B
-
56961-75-2
2,3,5-trichlorobenzaldehyde

| Conditions | Yield |
|---|---|
| Stage #1: 1,2,4-Trichlorobenzene With n-butyllithium In tetrahydrofuran Stage #2: N,N-dimethyl-formamide In tetrahydrofuran at -78 - 20℃; for 20h; | A 7% B 92% |


| Conditions | Yield |
|---|---|
| Stage #1: carbon dioxide With AuOH(1,3-bis(2,6-diisopropylphenyl)imidazol-2-ylidene); potassium hydroxide In tetrahydrofuran at 20℃; under 1125.11 Torr; for 0.25h; Stage #2: 1,2,4-Trichlorobenzene In tetrahydrofuran at 20℃; under 1125.11 Torr; for 12h; Stage #3: With hydrogenchloride In tetrahydrofuran; water regioselective reaction; | 92% |

| Conditions | Yield |
|---|---|
| With potassium fluoride; 5,11,17,23,29,35-hexakis(tert-butyl)-37,38,39,40,41-hexakis(hydroxy)calix[6]arene In sulfolane at 185℃; for 5h; Reagent/catalyst; Temperature; Inert atmosphere; Sealed tube; | 92% |
-
-
7732-18-5
water

-
-
86129-53-5
N-methyl-N-[2-(2'-chlorophenyl)ethyl]-2-chloroethylamine hydrochloride

-
-
86129-52-4
2-[[2-(2-chlorophenyl)ethyl](methyl)amino]ethanol

-
-
120-82-1
1,2,4-Trichlorobenzene

-
-
73943-10-9
SKF 86466

| Conditions | Yield |
|---|---|
| With hydrogenchloride; sodium hydroxide; aluminium trichloride In toluene | 91% |


| Conditions | Yield |
|---|---|
| With 1-[2-(diphenylphosphino)phenyl]ethanol; bis(acetylacetonate)nickel(II) In diethyl ether at 45℃; for 48h; | 89% |

| Conditions | Yield |
|---|---|
| Stage #1: phenylmagnesium bromide; 1,2,4-Trichlorobenzene; 1-[2-(diphenylphosphino)phenyl]ethanol; bis(acetylacetonate)nickel(II) In diethyl ether at 45℃; for 48h; Stage #2: With methanol In diethyl ether Product distribution / selectivity; | 89% |

| Conditions | Yield |
|---|---|
| In N,N,N,N,N,N-hexamethylphosphoric triamide at 120℃; for 1h; | 88% |
| at 180℃; | |
| With methanol at 180℃; |
1,2,4-TRICHLOROBENZENE Chemical Properties
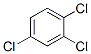
Formula Weight: 181.45
mp : 16 °C(lit.)
bp : 214 °C(lit.)
density : 1.454 g/mL at 25 °C(lit.)
Fp : >230 °F
Flash point : 110 °C
vapor density : >6 (vs air)
vapor pressure : 1 mm Hg ( 40 °C)
refractive index : n20/D 1.571(lit.)
storage temp. : 0-6°C
Water Solubility : INSOLUBLE
Appearance of 1,2,4-TRICHLOROBENZENE (120-82-1) : Colorless liquid
1,2,4-TRICHLOROBENZENE Uses
1,2,4-TRICHLOROBENZENE Toxicity Data With Reference
| 1. | skn-rbt 1950 mg/13W-I MOD | AEHLAU Archives of Environmental Health. 30 (1975),165. | ||
| 2. | mnt-mus-ipr 210 mg/kg/24H | MUTAEX Mutagenesis. 2 (1987),111. | ||
| 3. | orl-rat LD50:756 mg/kg | AOHYA3 Annals of Occupational Hygiene. 12 (1969),209. | ||
| 4. | orl-mus LD50:300 mg/kg | NAIZAM Nara Igaku Zasshi. Journal of the Nara Medical Association. 29 (1978),569. | ||
| 5. | ipr-mus LD50:1223 mg/kg | MUTAEX Mutagenesis. 2 (1987),111. |
1,2,4-TRICHLOROBENZENE Consensus Reports
1,2,4-TRICHLOROBENZENE Safety Profile
Risk Statements : 22-38-50/53-52/53-39/23/24/25-23/24/25-11
Safety Statements : 23-37/39-60-61-45-36/37-16
RIDADR : UN 2321 6.1/PG 3
WGK Germany : 3
RTECS : DC2100000
Hazard Note : Irritant
HazardClass : 6.1
PackingGroup : III
HS Code : 29036990
Poison by ingestion. Moderately toxic by intraperitoneal route. An experimental teratogen. Experimental reproductive effects. Mutation data reported. A skin irritant. Combustible when exposed to heat or flame. Can react vigorously with oxidizing materials. To fight fire, use water, foam, CO2, dry chemical. When heated to decomposition it emits toxic fumes of Cl−. See also CHLORINATED HYDROCARBONS, AROMATIC.
1,2,4-TRICHLOROBENZENE Standards and Recommendations
ACGIH TLV: CL 5 ppm
DFG MAK: Confirmed Animal Carcinogen with Unknown Relevance to Humans
1,2,4-TRICHLOROBENZENE Analytical Methods
Related Products
- 10,10'-Bis([1,1'-biphenyl]-4-yl)-9,9'-bianthracene
- 10,10'-Dibromo-9,9'-bianthryl
- 10,10-Dimethylanthrone
- 10,10-Oxybisphenoxarsine
- 10-[1,1'-Biphenyl]-4-yl-2-(1-methylethyl)-9-oxo-9H-thioxanthenium hexafluorophosphate
- 10,11-Dehydroimipramine
- 10,11-Dihydro-11-oxodibenzo[b,f][1,4]thiazepine
- 10,11-Dihydro-2-(4-methyl-1-piperazinyl)-11-(2-athiazolyl)-pyridazino(3,4-b)(1,4)benzoxazepine
- 10,11-DIHYDRO-2-(4-METHYL-1-PIPERAZINYL)-11-(3,4-XYLYL)PYRIDAZINO(3,4b)(1,4)-BENZOXAZEPINE MALEATE
- 10,11-Dihydro-5-(3-dimethylamino-2-methylpropyl)-5h-dibenz (b,f)azepine
- 12082-47-2
- 120-83-2
- 12083-92-0
- 12084-29-6
- 120849-36-7
- 120851-71-0
- 1208-52-2
- 12086-40-7
- 1208-64-6
- 1208-65-7
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View