-
Name
1-Cyclopropyl-6,7-difluoro-1,4-dihydro-8-methoxy-4-oxo-3-quinolinecarboxylic acid
- EINECS 214-439-2
- CAS No. 112811-72-0
- Article Data12
- CAS DataBase
- Density 1.174 g/cm3
- Solubility
- Melting Point 192-194 ºC
- Formula C14H11F2NO4
- Boiling Point 245.2 ºC at 760 mmHg
- Molecular Weight 295.242
- Flash Point 102.1 ºC
- Transport Information
- Appearance off-white to light-yellow powder
- Safety 24/25
- Risk Codes 22
-
Molecular Structure
- Hazard Symbols Xn
- Synonyms 1-Cyclopropyl-1,4-dihydro-6,7-difluoro-8-methoxy-4-oxoquinoline-3-carboxylicacid;1-Cyclopropyl-6,7-difluoro-1,4-dihydro-8-methoxy-4-oxo-3-quinolinecarboxylicacid;1-Cyclopropyl-6,7-difluoro-8-methoxy-1,4-dihydro-4-oxoquinoline-3-carboxylicacid;1-Cyclopropyl-6,7-difluoro-8-methoxy-4-oxo-1,4-dihydro-3-quinolinecarboxylic acid;1-Cyclopropyl-6,7-difluoro-8-methoxy-4-oxo-1,4-dihydroquinolin-3-carboxylicacid;1-Cyclopropyl-6,7-difluoro-8-methoxy-4-quinolone-3-carboxylic acid;3-Quinolinecarboxylicacid, 1-cyclopropyl-6,7-difluoro-1,4-dihydro-8-methoxy-4-oxo-;
- PSA 68.53000
- LogP 2.32140
Synthetic route
-
-
112811-71-9
ethyl 8-methoxy-1-cyclopropyl-6,7-difluoro-1,4-dihydro-4-oxo-3-quinolinecarboxylate

-
-
112811-72-0
1-cyclopropyl-6,7-difluoro-1,4-dihydro-8-methoxy-4-oxo-3-quinolinecarboxylic acid

| Conditions | Yield |
|---|---|
| With hydrogenchloride In methanol; water at 65℃; for 15h; Solvent; Temperature; | 96.83% |
| With sulfuric acid; acetic acid In water for 2h; Reflux; | 95.92% |
| With hydrogenchloride for 3h; Heating; | 90% |
-
-
154093-72-8
1-cyclopropyl-6,7-difluoro-1,4-dihydro-8-hydroxy-4-oxo-3-quinoline carboxylic acid

-
-
77-78-1
dimethyl sulfate

-
A
-
141290-02-0
1-cyclopropyl-6,7-difluoro-1,4-dihydro-8-methoxy-4-oxo-3-quinoline carboxylic acid methyl ester

-
B
-
112811-72-0
1-cyclopropyl-6,7-difluoro-1,4-dihydro-8-methoxy-4-oxo-3-quinolinecarboxylic acid

| Conditions | Yield |
|---|---|
| With potassium chloride In acetone Product distribution / selectivity; Heating / reflux; |

-
-
141290-02-0
1-cyclopropyl-6,7-difluoro-1,4-dihydro-8-methoxy-4-oxo-3-quinoline carboxylic acid methyl ester

-
-
112811-72-0
1-cyclopropyl-6,7-difluoro-1,4-dihydro-8-methoxy-4-oxo-3-quinolinecarboxylic acid

| Conditions | Yield |
|---|---|
| With water; potassium carbonate for 0.5h; Product distribution / selectivity; Heating / reflux; |
-
-
112811-70-8
ethyl 3-(cyclopropylamino)-2-(2,4,5-trifluoro-3-methoxybenzoyl)acrylate

-
-
112811-72-0
1-cyclopropyl-6,7-difluoro-1,4-dihydro-8-methoxy-4-oxo-3-quinolinecarboxylic acid

| Conditions | Yield |
|---|---|
| With sodium fluoride In N,N-dimethyl-formamide | |
| Multi-step reaction with 2 steps 1: sodiumfluoride / N,N-dimethyl-formamide / 5 h 2: sulfuric acid; water / acetic acid / 1 h View Scheme |
-
-
112811-64-0
2,4,5-trifluoro-3-methoxy-benzamide

-
-
112811-72-0
1-cyclopropyl-6,7-difluoro-1,4-dihydro-8-methoxy-4-oxo-3-quinolinecarboxylic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 8 steps 1: sulfuric acid / 3.5 h / 100 °C 2: thionyl chloride / 3 h 3: magnesium ethylate / toluene 4: toluene-4-sulfonic acid / water / 9 h 5: acetic anhydride / 11 h 6: ethanol / 2 h / 20 °C 7: sodiumfluoride / N,N-dimethyl-formamide / 5 h 8: sulfuric acid; water / acetic acid / 1 h View Scheme |
-
-
112811-63-9
3-methoxy-2,4,5-trifluorobenzonitrile

-
-
112811-72-0
1-cyclopropyl-6,7-difluoro-1,4-dihydro-8-methoxy-4-oxo-3-quinolinecarboxylic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 9 steps 1: sulfuric acid; water / 1 h / 110 °C 2: sulfuric acid / 3.5 h / 100 °C 3: thionyl chloride / 3 h 4: magnesium ethylate / toluene 5: toluene-4-sulfonic acid / water / 9 h 6: acetic anhydride / 11 h 7: ethanol / 2 h / 20 °C 8: sodiumfluoride / N,N-dimethyl-formamide / 5 h 9: sulfuric acid; water / acetic acid / 1 h View Scheme |
-
-
112811-67-3
diethyl 3-methoxy-2,4,5-trifluorobenzoylmalonate

-
-
112811-72-0
1-cyclopropyl-6,7-difluoro-1,4-dihydro-8-methoxy-4-oxo-3-quinolinecarboxylic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1: toluene-4-sulfonic acid / water / 9 h 2: acetic anhydride / 11 h 3: ethanol / 2 h / 20 °C 4: sodiumfluoride / N,N-dimethyl-formamide / 5 h 5: sulfuric acid; water / acetic acid / 1 h View Scheme |
-
-
13332-24-6
1-bromo-2,4,5-trifluoro-3-methoxybenzene

-
-
112811-72-0
1-cyclopropyl-6,7-difluoro-1,4-dihydro-8-methoxy-4-oxo-3-quinolinecarboxylic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 10 steps 1: 1-methyl-pyrrolidin-2-one / 4.5 h / 140 - 150 °C 2: sulfuric acid; water / 1 h / 110 °C 3: sulfuric acid / 3.5 h / 100 °C 4: thionyl chloride / 3 h 5: magnesium ethylate / toluene 6: toluene-4-sulfonic acid / water / 9 h 7: acetic anhydride / 11 h 8: ethanol / 2 h / 20 °C 9: sodiumfluoride / N,N-dimethyl-formamide / 5 h 10: sulfuric acid; water / acetic acid / 1 h View Scheme |
-
-
112811-65-1
3-methoxy-2,4,5-trifluorobenzoic acid

-
-
112811-72-0
1-cyclopropyl-6,7-difluoro-1,4-dihydro-8-methoxy-4-oxo-3-quinolinecarboxylic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 7 steps 1: thionyl chloride / 3 h 2: magnesium ethylate / toluene 3: toluene-4-sulfonic acid / water / 9 h 4: acetic anhydride / 11 h 5: ethanol / 2 h / 20 °C 6: sodiumfluoride / N,N-dimethyl-formamide / 5 h 7: sulfuric acid; water / acetic acid / 1 h View Scheme |
-
-
112811-66-2
2,4,5-trifluoro-3-methoxybenzoyl chloride

-
-
112811-72-0
1-cyclopropyl-6,7-difluoro-1,4-dihydro-8-methoxy-4-oxo-3-quinolinecarboxylic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 6 steps 1: magnesium ethylate / toluene 2: toluene-4-sulfonic acid / water / 9 h 3: acetic anhydride / 11 h 4: ethanol / 2 h / 20 °C 5: sodiumfluoride / N,N-dimethyl-formamide / 5 h 6: sulfuric acid; water / acetic acid / 1 h View Scheme |
-
-
112811-68-4
ethyl 3-methoxy-2,4,5-trifluorobenzoylacetate

-
-
112811-72-0
1-cyclopropyl-6,7-difluoro-1,4-dihydro-8-methoxy-4-oxo-3-quinolinecarboxylic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: acetic anhydride / 11 h 2: ethanol / 2 h / 20 °C 3: sodiumfluoride / N,N-dimethyl-formamide / 5 h 4: sulfuric acid; water / acetic acid / 1 h View Scheme |
-
-
122375-85-3
ethyl 3-ethoxy-2-(2,4,5-trifluoro-3-methoxybenzoyl)acrylate

-
-
112811-72-0
1-cyclopropyl-6,7-difluoro-1,4-dihydro-8-methoxy-4-oxo-3-quinolinecarboxylic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: ethanol / 2 h / 20 °C 2: sodiumfluoride / N,N-dimethyl-formamide / 5 h 3: sulfuric acid; water / acetic acid / 1 h View Scheme |
-
-
530-62-1
1,1'-carbonyldiimidazole

-
-
112811-72-0
1-cyclopropyl-6,7-difluoro-1,4-dihydro-8-methoxy-4-oxo-3-quinolinecarboxylic acid

-
-
869310-07-6
1-cyclopropyl-6,7-difluoro-3-(imidazole-1-carbonyl)-8-methoxy-1H-quinolin-4-one

| Conditions | Yield |
|---|---|
| In tetrahydrofuran Heating / reflux; | 100% |
| In chloroform Heating / reflux; | 100% |
| In tetrahydrofuran Heating / reflux; | 100% |
| In tetrahydrofuran Heating / reflux; | 100% |
-
-
11113-50-1
boric acid

-
-
108-24-7
acetic anhydride

-
-
112811-72-0
1-cyclopropyl-6,7-difluoro-1,4-dihydro-8-methoxy-4-oxo-3-quinolinecarboxylic acid

-
-
139693-52-0
1-cyclopropyl-6,7-difluoro-1,4-dihydro-8-methoxy-4-oxo-3-quinoline carboxylic acid-O3,O4(bis(acyloxy-O)) borate

| Conditions | Yield |
|---|---|
| Stage #1: boric acid; acetic anhydride With potassium hydroxide; zinc(II) chloride at 100℃; for 12h; Stage #2: 1-cyclopropyl-6,7-difluoro-1,4-dihydro-8-methoxy-4-oxo-3-quinolinecarboxylic acid at 50℃; for 1h; Concentration; Temperature; Reagent/catalyst; | 97.4% |

-
-
112811-72-0
1-cyclopropyl-6,7-difluoro-1,4-dihydro-8-methoxy-4-oxo-3-quinolinecarboxylic acid

-
-
847233-56-1
1-cyclopropyl-1,4-dihydro-6,7-difluoro-8-methoxy-4-oxo-quinoline-3-carboxylic acid difluoroborate ester

| Conditions | Yield |
|---|---|
| Stage #1: 1-cyclopropyl-6,7-difluoro-1,4-dihydro-8-methoxy-4-oxo-3-quinolinecarboxylic acid With potassium carbonate In tetrahydrofuran at 20℃; for 0.0833333h; Stage #2: With boron trifluoride diethyl etherate In tetrahydrofuran at 20 - 66℃; for 6.08333h; | 97% |
| With boron trifluoride diethyl etherate; potassium carbonate In tetrahydrofuran at 20 - 66℃; Inert atmosphere; | 97% |
| With BF3OEt In various solvent(s) Cyclization; Heating; | 83% |
| With boron trifluoride diethyl etherate In tetrahydrofuran for 36h; Heating / reflux; | |
| Multi-step reaction with 2 steps 1: acetonitrile / 1 h / 70 - 80 °C / Inert atmosphere; Large scale 2: 1 h / 0 - 15 °C / Large scale View Scheme |
-
-
109-63-7
boron trifluoride diethyl etherate

-
-
112811-72-0
1-cyclopropyl-6,7-difluoro-1,4-dihydro-8-methoxy-4-oxo-3-quinolinecarboxylic acid

| Conditions | Yield |
|---|---|
| Stage #1: 1-cyclopropyl-6,7-difluoro-1,4-dihydro-8-methoxy-4-oxo-3-quinolinecarboxylic acid With potassium carbonate In tetrahydrofuran at 20℃; for 0.0833333h; Inert atmosphere; Stage #2: boron trifluoride diethyl etherate In tetrahydrofuran at 60℃; for 6h; Temperature; Reagent/catalyst; | 97% |
-
-
124-68-5
2-Amino-2-methyl-1-propanol

-
-
112811-72-0
1-cyclopropyl-6,7-difluoro-1,4-dihydro-8-methoxy-4-oxo-3-quinolinecarboxylic acid

| Conditions | Yield |
|---|---|
| In toluene at 105 - 115℃; Temperature; Solvent; Inert atmosphere; Industrial scale; | 96.6% |
-
-
112811-72-0
1-cyclopropyl-6,7-difluoro-1,4-dihydro-8-methoxy-4-oxo-3-quinolinecarboxylic acid

-
-
151213-40-0
(1S,6S)-2,8-diazabicyclo[4.3.0]nonane

-
-
151096-09-2
moxifloxacin

| Conditions | Yield |
|---|---|
| With [BCl2(4-picoline)][AlCl4]; triethylamine In methanol at 85℃; for 6h; Catalytic behavior; Reagent/catalyst; Solvent; Temperature; | 96% |
| In dimethyl sulfoxide at 65 - 70℃; for 6 - 8h; | 89.7% |
| With triethylamine In N,N-dimethyl-formamide; acetonitrile at 0 - 78℃; for 11h; Reflux; | 66% |
-
-
151213-42-2
[R,R]-2,8-diazabicyclo[4.3.0]nonane

-
-
112811-72-0
1-cyclopropyl-6,7-difluoro-1,4-dihydro-8-methoxy-4-oxo-3-quinolinecarboxylic acid

| Conditions | Yield |
|---|---|
| With nano iron oxide on ZrO2 coated sulfonic acid In water for 0.416667h; Reflux; | 96% |
| at 150℃; Microwave irradiation; | 83% |
-
-
4887-24-5
triacetoxyborane

-
-
112811-72-0
1-cyclopropyl-6,7-difluoro-1,4-dihydro-8-methoxy-4-oxo-3-quinolinecarboxylic acid

| Conditions | Yield |
|---|---|
| In water at 0 - 90℃; for 4h; | 95.3% |

-
-
112811-72-0
1-cyclopropyl-6,7-difluoro-1,4-dihydro-8-methoxy-4-oxo-3-quinolinecarboxylic acid

| Conditions | Yield |
|---|---|
| Stage #1: 3-methylaminopiperidine dihydrochloride; 1-cyclopropyl-6,7-difluoro-1,4-dihydro-8-methoxy-4-oxo-3-quinolinecarboxylic acid With boron trifluoride-tetrahydrofuran complex; triethylamine In acetonitrile at 20℃; for 26h; Stage #2: With methanol for 6h; Reflux; Stage #3: With sodium hydroxide In methanol; water at 20℃; for 16h; pH=8; | 92% |
| With triethylamine In acetonitrile |
-
-
13264-88-5, 66918-24-9, 75336-85-5, 75336-89-9, 93334-99-7
(+/-)-2-methylpiperazine bis(hydrochloride)

-
-
112811-72-0
1-cyclopropyl-6,7-difluoro-1,4-dihydro-8-methoxy-4-oxo-3-quinolinecarboxylic acid

-
-
160738-57-8, 112811-59-3
gatifloxacin

| Conditions | Yield |
|---|---|
| Stage #1: (+/-)-2-methylpiperazine bis(hydrochloride); 1-cyclopropyl-6,7-difluoro-1,4-dihydro-8-methoxy-4-oxo-3-quinolinecarboxylic acid With boron trifluoride-tetrahydrofuran complex; triethylamine In acetonitrile at 20℃; for 25h; Stage #2: With methanol for 5h; Reflux; Stage #3: With sodium hydroxide In methanol; water at 20℃; for 16h; pH=7; | 91% |
-
-
87-69-4
L-Tartaric acid

-
-
112811-72-0
1-cyclopropyl-6,7-difluoro-1,4-dihydro-8-methoxy-4-oxo-3-quinolinecarboxylic acid

-
-
151213-40-0
(1S,6S)-2,8-diazabicyclo[4.3.0]nonane

-
-
1082245-30-4
L(+)-moxifloxacin tartrate

| Conditions | Yield |
|---|---|
| Stage #1: 1-cyclopropyl-6,7-difluoro-1,4-dihydro-8-methoxy-4-oxo-3-quinolinecarboxylic acid; (1S,6S)-2,8-diazabicyclo[4.3.0]nonane In N,N-dimethyl-formamide at 65 - 70℃; for 6 - 8h; Stage #2: L-Tartaric acid In N,N-dimethyl-formamide at 25 - 80℃; for 13 - 16h; Product distribution / selectivity; | 90.32% |

-
-
112811-72-0
1-cyclopropyl-6,7-difluoro-1,4-dihydro-8-methoxy-4-oxo-3-quinolinecarboxylic acid

-
-
154093-72-8
1-cyclopropyl-6,7-difluoro-1,4-dihydro-8-hydroxy-4-oxo-3-quinoline carboxylic acid

| Conditions | Yield |
|---|---|
| With hydrogenchloride; calcium chloride In water; dimethyl sulfoxide at -5 - 25℃; for 3.5h; pH=4; | 90% |
| With hydrogen bromide; acetic acid at 100℃; for 24h; | |
| Stage #1: 1-cyclopropyl-6,7-difluoro-1,4-dihydro-8-methoxy-4-oxo-3-quinolinecarboxylic acid With hydrogen bromide In acetic acid at 100℃; for 1h; Stage #2: With water In acetic acid at 0℃; | |
| With hydrogen bromide; acetic acid at 100℃; for 24h; |
-
-
112811-72-0
1-cyclopropyl-6,7-difluoro-1,4-dihydro-8-methoxy-4-oxo-3-quinolinecarboxylic acid

-
-
151213-40-0
(1S,6S)-2,8-diazabicyclo[4.3.0]nonane

-
-
186826-86-8
moxifloxacin hydrochloride

| Conditions | Yield |
|---|---|
| Stage #1: 1-cyclopropyl-6,7-difluoro-1,4-dihydro-8-methoxy-4-oxo-3-quinolinecarboxylic acid; (1S,6S)-2,8-diazabicyclo[4.3.0]nonane With titanium(IV) isopropylate; triethylamine In isopropyl alcohol at 100℃; Inert atmosphere; Stage #2: With hydrogenchloride In methanol at 20℃; for 1h; pH=1; Solvent; Reagent/catalyst; | 90% |
| Multi-step reaction with 2 steps 1: Alkaline conditions 2: hydrogenchloride View Scheme | |
| Stage #1: 1-cyclopropyl-6,7-difluoro-1,4-dihydro-8-methoxy-4-oxo-3-quinolinecarboxylic acid; (1S,6S)-2,8-diazabicyclo[4.3.0]nonane With 1,8-diazabicyclo[5.4.0]undec-7-ene In acetonitrile at 20 - 85℃; for 36h; Inert atmosphere; Stage #2: With hydrogenchloride In water at 15℃; for 1h; pH=1.4 - 1.8; Inert atmosphere; | 3.3 g |
-
-
21850-12-4
cycloperhydroisoindole

-
-
112811-72-0
1-cyclopropyl-6,7-difluoro-1,4-dihydro-8-methoxy-4-oxo-3-quinolinecarboxylic acid

| Conditions | Yield |
|---|---|
| With triethylamine In acetonitrile for 24h; Reflux; | 89% |
-
-
123-75-1
pyrrolidine

-
-
121-43-7
Trimethyl borate

-
-
112811-72-0
1-cyclopropyl-6,7-difluoro-1,4-dihydro-8-methoxy-4-oxo-3-quinolinecarboxylic acid

| Conditions | Yield |
|---|---|
| In ethanol; acetonitrile | 88.2% |

-
-
660-68-4
diethyl amine hydrochloride

-
-
112811-72-0
1-cyclopropyl-6,7-difluoro-1,4-dihydro-8-methoxy-4-oxo-3-quinolinecarboxylic acid

-
-
1028205-74-4
1-cyclopropyl-N,N-diethyl-6,7-difluoro-1,4-dihydro-8-methoxy-4-oxoquinoline-3-carboxamide

| Conditions | Yield |
|---|---|
| With benzotriazol-1-ol; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride; N-ethyl-N,N-diisopropylamine In N,N-dimethyl-formamide at 10 - 35℃; Inert atmosphere; | 87.3% |
-
-
882-33-7
diphenyldisulfane

-
-
112811-72-0
1-cyclopropyl-6,7-difluoro-1,4-dihydro-8-methoxy-4-oxo-3-quinolinecarboxylic acid

| Conditions | Yield |
|---|---|
| With palladium diacetate; triphenylphosphine; silver carbonate In dimethyl sulfoxide at 130℃; for 12h; Schlenk technique; | 87% |
-
-
121-43-7
Trimethyl borate

-
-
79286-87-6
(RS)-3-(acetyl-methyl-amino)-pyrrolidine

-
-
112811-72-0
1-cyclopropyl-6,7-difluoro-1,4-dihydro-8-methoxy-4-oxo-3-quinolinecarboxylic acid

| Conditions | Yield |
|---|---|
| In ethanol; acetonitrile | 83.9% |


-
-
112811-72-0
1-cyclopropyl-6,7-difluoro-1,4-dihydro-8-methoxy-4-oxo-3-quinolinecarboxylic acid

| Conditions | Yield |
|---|---|
| With potassium tert-butylate In N,N-dimethyl-formamide at 80℃; for 3h; | 82.3% |
-
-
109-07-9
(RS)-2-methylpiperazine

-
-
112811-72-0
1-cyclopropyl-6,7-difluoro-1,4-dihydro-8-methoxy-4-oxo-3-quinolinecarboxylic acid

-
-
160738-57-8, 112811-59-3
gatifloxacin

| Conditions | Yield |
|---|---|
| Stage #1: 1-cyclopropyl-6,7-difluoro-1,4-dihydro-8-methoxy-4-oxo-3-quinolinecarboxylic acid With 1,1,1,3,3,3-hexamethyl-disilazane In acetonitrile at 76 - 80℃; for 1h; Heating / reflux; Stage #2: With boron trifluoride diethyl etherate In acetonitrile at 0 - 25℃; for 2h; Stage #3: (RS)-2-methylpiperazine With triethylamine In acetonitrile at 15 - 25℃; for 3h; pH=~ 9; | 81.8% |
| In dimethyl sulfoxide at 55℃; for 24h; | 66% |
| In dimethyl sulfoxide at 60 - 65℃; for 29 - 45h; | 53.7% |
| In dimethyl sulfoxide at 20 - 73℃; for 13.9167 - 14.3333h; | 52% |
| In dimethyl sulfoxide at 70 - 95℃; for 2h; |

-
-
112811-72-0
1-cyclopropyl-6,7-difluoro-1,4-dihydro-8-methoxy-4-oxo-3-quinolinecarboxylic acid

| Conditions | Yield |
|---|---|
| With potassium tert-butylate In N,N-dimethyl-formamide at 80℃; for 3h; | 81.8% |

-
-
112811-72-0
1-cyclopropyl-6,7-difluoro-1,4-dihydro-8-methoxy-4-oxo-3-quinolinecarboxylic acid

| Conditions | Yield |
|---|---|
| With potassium tert-butylate In N,N-dimethyl-formamide at 80℃; for 3h; | 81.5% |

-
-
112811-72-0
1-cyclopropyl-6,7-difluoro-1,4-dihydro-8-methoxy-4-oxo-3-quinolinecarboxylic acid

-
-
1018938-14-1
C21H19FN4O6

| Conditions | Yield |
|---|---|
| With potassium carbonate In dimethyl sulfoxide for 0.0333333h; microwave irradiation; | 79% |
-
-
177-11-7
4,4-ethylenedioxy-piperidine

-
-
112811-72-0
1-cyclopropyl-6,7-difluoro-1,4-dihydro-8-methoxy-4-oxo-3-quinolinecarboxylic acid

-
-
848070-83-7
1-cylopropyl-7-(1,4-dioxa-8-aza-spiro[4,5]dec-8-yl)-6-fluoro-8-methoxy-4-oxo-1,4-dihydro-quinoline-3-carboxylic acid

| Conditions | Yield |
|---|---|
| With potassium carbonate In dimethyl sulfoxide for 0.0333333h; microwave irradiation; | 73% |
-
-
2744-08-3, 2744-09-4, 6329-61-9
decahydroisoquinoline

-
-
112811-72-0
1-cyclopropyl-6,7-difluoro-1,4-dihydro-8-methoxy-4-oxo-3-quinolinecarboxylic acid

| Conditions | Yield |
|---|---|
| With triethylamine In acetonitrile for 24h; Reflux; | 73% |
-
-
112811-72-0
1-cyclopropyl-6,7-difluoro-1,4-dihydro-8-methoxy-4-oxo-3-quinolinecarboxylic acid

-
-
151213-40-0
(1S,6S)-2,8-diazabicyclo[4.3.0]nonane

-
-
721970-36-1
1-cyclopropyl-6-fluoro-8-hydroxy-7-((4aS,7aS)-octahydro-6H-pyrrolo[3,4-b]pyridin-6-yl)-4-oxo-1,4-dihydroquinoline-3-carboxylic acid

| Conditions | Yield |
|---|---|
| Stage #1: 1-cyclopropyl-6,7-difluoro-1,4-dihydro-8-methoxy-4-oxo-3-quinolinecarboxylic acid With isobutylamine; copper dichloride In methanol; ethylene glycol at 50℃; for 4h; Inert atmosphere; Stage #2: (1S,6S)-2,8-diazabicyclo[4.3.0]nonane In methanol; ethylene glycol for 5h; Reflux; Stage #3: With sodium hydroxide In methanol; water; ethylene glycol for 1h; Reflux; | 72.1% |
-
-
123-90-0
Thiomorpholin

-
-
112811-72-0
1-cyclopropyl-6,7-difluoro-1,4-dihydro-8-methoxy-4-oxo-3-quinolinecarboxylic acid

-
-
114213-99-9
1-Cyclopropyl-6-fluoro-8-methoxy-7-thiomorpholino-1,4-dihydro-4-oxoquinoline-3-carboxylic acid

| Conditions | Yield |
|---|---|
| With potassium carbonate In dimethyl sulfoxide for 0.0333333h; microwave irradiation; | 70% |
-
-
3367-95-1
N,N-diethylpiperidine-3-carboxamide

-
-
112811-72-0
1-cyclopropyl-6,7-difluoro-1,4-dihydro-8-methoxy-4-oxo-3-quinolinecarboxylic acid

-
-
1018937-94-4
C24H30FN3O5

| Conditions | Yield |
|---|---|
| With potassium carbonate In dimethyl sulfoxide for 0.0333333h; microwave irradiation; | 70% |
1-Cyclopropyl-6,7-difluoro-1,4-dihydro-8-methoxy-4-oxo-3-quinolinecarboxylic acid Chemical Properties
Following is the structure of 1-Cyclopropyl-6,7-difluoro-1,4-dihydro-8-methoxy-4-oxo-3-quinolinecarboxylic acid (CAS NO.112811-72-0):
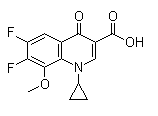
Empirical Formula: C14H11F2NO4
Molecular Weight: 295.2382 g/mol
Melting point: 192-194 °C
Molar Refractivity: 66.37 cm3
Molar Volume: 186.6 cm3
Density: 1.581 g/cm3
Flash Point: 240.7 °C
Index of Refraction: 1.629
Surface Tension: 66 dyne/cm
Enthalpy of Vaporization: 77.71 kJ/mol
Boiling Point: 474.4 °C at 760 mmHg
Vapour Pressure: 8.35E-10 mmHg at 25 °C
Appearance: Off-White to Light-Yellow Powder
Product Categories: Intermediates & Fine Chemicals; Pharmaceuticals; Gatifloxacin Hemihydrate
SMILES: Fc3c(F)c(OC)c1c(C(=O)\C(=C/N1C2CC2)C(=O)O)c3
InChI: InChI=1/C14H11F2NO4/c1-21-13-10(16)9(15)4-7-11(13)17(6-2-3-6)5-8(12(7)18)14(19)20/h4-6H,2-3H2,1H3,(H,19,20)
InChIKey: WQJZXSSAMGZVTM-UHFFFAOYAT
1-Cyclopropyl-6,7-difluoro-1,4-dihydro-8-methoxy-4-oxo-3-quinolinecarboxylic acid Uses
1-Cyclopropyl-6,7-difluoro-1,4-dihydro-8-methoxy-4-oxo-3-quinolinecarboxylic acid (CAS NO.112811-72-0) can be used as Moxifloxacin intermediate.
1-Cyclopropyl-6,7-difluoro-1,4-dihydro-8-methoxy-4-oxo-3-quinolinecarboxylic acid Specification
1-Cyclopropyl-6,7-difluoro-1,4-dihydro-8-methoxy-4-oxo-3-quinolinecarboxylic acid , its cas register number is 112811-72-0. It also can be called 3-Quinolinecarboxylic acid, 1-cyclopropyl-6,7-difluoro-1,4-dihydro-8-methoxy-4-oxo- .
Related Products
- 1-Cyclopropyl-6,7,8-trifluoro-4-oxo-1,4-dihydroquinoline-3-carboxylic acid
- 1-Cyclopropyl-6,7-difluoro-1,4-dihydro-8-methoxy-4-oxo-3-quinolinecarboxylic acid
- 1-Cyclopropyl-6,7-difluoro-1,4-dihydro-8-methoxy-4-oxo-3-quinolinecarboxylic acid ethyl ester
- 112811-90-2
- 11281-65-5
- 112822-85-2
- 112-82-3
- 1128-23-0
- 1128268-02-9
- 112828-00-9
- 112828-13-4
- 1128-28-5
- 112835-62-8
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View