-
Name
1-Naphthyl acetonitrile
- EINECS 205-078-1
- CAS No. 132-75-2
- Article Data65
- CAS DataBase
- Density 1.116 g/cm3
- Solubility
- Melting Point 33-35 °C(lit.)
- Formula C12H9N
- Boiling Point 323.9 °C at 760 mmHg
- Molecular Weight 167.21
- Flash Point 180.3 °C
- Transport Information
- Appearance White crystalline powder
- Safety 26-37/39-24/25-37-36
- Risk Codes 20/21/22-36/37/38-22
-
Molecular Structure
-
Hazard Symbols
 Xn
Xn
- Synonyms 1-Cyanomethylnaphthalene;1-Naphthylacetonitrile;2-(1-Naphthyl)acetonitrile;NSC 9844;a-Naphthylacetonitrile;1-Naphthaleneacetonitrile;
- PSA 23.79000
- LogP 2.90588
Synthetic route

| Conditions | Yield |
|---|---|
| With 2,2'-azobis(isobutyronitrile); tri-n-butyl-tin hydride In benzene Heating; | A 98% B 95% |
-
-
139109-57-2
2-[(Z)-Hydroxyimino]-3-naphthalen-1-yl-propionic acid

-
-
132-75-2
naphthalen-1-ylacetonitrile

| Conditions | Yield |
|---|---|
| With 1,1'-carbonyldiimidazole In benzene 1.) r.t., 15 min, 2.) 68-70 deg C, 1 h; | 97% |

| Conditions | Yield |
|---|---|
| With trichloromethyl chloroformate In various solvent(s) 0-5 deg C then heated to 60 deg C, 5 min; | 96% |
| With (dimethoxy)methylsilane; copper diacetate; 1,2-bis-(dicyclohexylphosphino)ethane In tetrahydrofuran at 20℃; for 12h; Sealed tube; | 96% |
| With phosgene In tetrahydrofuran; toluene at 25℃; for 4h; Inert atmosphere; | 91% |

| Conditions | Yield |
|---|---|
| In acetonitrile at 60℃; for 3h; 18-crown-6 polyether; | 96% |
-
-
37630-26-5, 4735-49-3
β-(1-naphthyl)nitroethylene

-
-
132-75-2
naphthalen-1-ylacetonitrile

| Conditions | Yield |
|---|---|
| With titanium tetrachloride; tetraethylammonium tosylate In N,N-dimethyl-formamide electroreduction - 4 mA/cm2; | 95% |

| Conditions | Yield |
|---|---|
| With dicyclohexyl-(2',6'-dimethoxybiphenyl-2-yl)-phosphane; bis(η3-allyl-μ-chloropalladium(II)) In 1,3,5-trimethyl-benzene at 20 - 140℃; for 5.16667h; Inert atmosphere; Sealed tube; chemoselective reaction; | 95% |

| Conditions | Yield |
|---|---|
| With dicyclohexyl-(2',6'-dimethoxybiphenyl-2-yl)-phosphane; palladium diacetate; sodium carbonate In 1,4-dioxane; water at 60℃; for 12h; Inert atmosphere; Sealed tube; | 93% |
-
-
133712-83-1
2-[(Z)-Hydroxyimino]-3-naphthalen-1-yl-propionic acid

-
-
132-75-2
naphthalen-1-ylacetonitrile

| Conditions | Yield |
|---|---|
| With 1,1'-oxalyldiimidazole In benzene 1.) r.t., 15 min, 2.) 68-70 deg C, 1 h; | 90% |

-
-
132-75-2
naphthalen-1-ylacetonitrile

| Conditions | Yield |
|---|---|
| With 2,3-dicyano-5,6-dichloro-p-benzoquinone In 1,2-dichloro-ethane at 100℃; for 24h; | 90% |
| With 2,3-dicyano-5,6-dichloro-p-benzoquinone In 1,2-dichloro-ethane at 100℃; for 24h; | 90% |
-
-
74607-42-4
Methylsulfanyl-naphthalen-1-yl-acetonitrile

-
-
132-75-2
naphthalen-1-ylacetonitrile

| Conditions | Yield |
|---|---|
| With acetic acid; zinc at 100℃; for 1h; | 89% |
-
-
43017-75-0
1-naphthylacetaldehyde

-
-
132-75-2
naphthalen-1-ylacetonitrile

| Conditions | Yield |
|---|---|
| With HCl·DMPU; hydroxylamine hydrochloride In acetonitrile at 60℃; | 88% |
| With ammonia; iodine at 0℃; for 1h; | 74% |
-
-
557-21-1
zinc(II) cyanide

-
-
72681-59-5
1-naphthylmethyl 2,2-dimethylpropanoate

-
-
132-75-2
naphthalen-1-ylacetonitrile

| Conditions | Yield |
|---|---|
| Stage #1: zinc(II) cyanide; 1-naphthylmethyl 2,2-dimethylpropanoate With potassium phosphate; [1,1'-bis(diphenylphosphino)ferrocene]nickel(II) chloride In N,N-dimethyl-formamide for 0.0166667h; Inert atmosphere; Sealed tube; Stage #2: With diethylzinc In hexane; N,N-dimethyl-formamide at 110℃; for 16h; Inert atmosphere; Sealed tube; | 77% |

| Conditions | Yield |
|---|---|
| Stage #1: bromocyane; 1-naphthalene methanol With triphenylphosphine In dichloromethane at 20℃; Inert atmosphere; Stage #2: With 1,8-diazabicyclo[5.4.0]undec-7-ene In dichloromethane at 20℃; Cooling with ice; Inert atmosphere; | 69% |

| Conditions | Yield |
|---|---|
| With dicyclohexyl-(2',6'-dimethoxybiphenyl-2-yl)-phosphane; bis(η3-allyl-μ-chloropalladium(II)) In 1,3,5-trimethyl-benzene at 20 - 140℃; for 10.1667h; Inert atmosphere; Sealed tube; chemoselective reaction; | 68% |
-
-
773837-37-9
sodium cyanide

-
-
3163-27-7
2-(bromomethyl)naphthalene

-
-
132-75-2
naphthalen-1-ylacetonitrile

| Conditions | Yield |
|---|---|
| With tetra-(n-butyl)ammonium iodide In acetonitrile Inert atmosphere; Reflux; | 67% |
-
-
13922-41-3
1-Naphthylboronic acid

-
-
6011-14-9
2-aminoacetonitrile hydrochloride

-
-
132-75-2
naphthalen-1-ylacetonitrile

| Conditions | Yield |
|---|---|
| With sodium nitrite In water; toluene at 50℃; for 24h; Schlenk technique; | 65% |
-
-
38622-91-2, 36635-61-7
[(p-methylphenyl)sulfonylmethyl]isonitrile

-
-
66-77-3
1-naphthaldehyde

-
-
132-75-2
naphthalen-1-ylacetonitrile

| Conditions | Yield |
|---|---|
| With potassium tert-butylate In 1,2-dimethoxyethane at -55℃; for 1h; | 62% |

| Conditions | Yield |
|---|---|
| With potassium hexamethylsilazane In tetrahydrofuran at 20 - 80℃; for 1.5h; Inert atmosphere; Schlenk technique; Sealed tube; | 53% |

| Conditions | Yield |
|---|---|
| Stage #1: ethyl 2-cyanoacetate; 1-Chloronaphthalene With N-(2-methylnaphthalen-1-yl)-N’-(pyridin-2-ylmethyl)oxalamide; copper(I) bromide; sodium t-butanolate In isopropyl alcohol at 105℃; for 24h; Schlenk technique; Inert atmosphere; Stage #2: With water In isopropyl alcohol at 105℃; for 12h; Schlenk technique; Inert atmosphere; Cooling; | 51% |
-
-
90-14-2
1-Iodonaphthalene

-
-
105-56-6
ethyl 2-cyanoacetate

-
A
-
132-75-2
naphthalen-1-ylacetonitrile

-
B
-
13234-71-4
ethyl 2-cyano-2-(naphthalen-1-yl)acetate

| Conditions | Yield |
|---|---|
| With potassium carbonate; copper(l) iodide In dimethyl sulfoxide at 120℃; for 9h; | A 27% B 47% |
| With potassium carbonate; copper(l) iodide In dimethyl sulfoxide at 120℃; for 9h; | A 27% B 27% |

-
-
556-64-9
methyl thiocyanate

-
-
66-77-3
1-naphthaldehyde

-
A
-
132-75-2
naphthalen-1-ylacetonitrile

-
B
-
4906-34-7
[1]thionaphthoic acid S-methyl ester

| Conditions | Yield |
|---|---|
| With tributylphosphine 1.) 1h, 0 deg C, 2.) 2h, ambient temperature; | A 32% B 15% |
| With tributylphosphine 1.) 0 deg C, 1 h, 2.) room temperature, 2 h; | A 32% B 15% |

-
-
97550-35-1
(+)-(1R)-1-exo-Cyano-1a,7b-dihydro-1H-cyclopropanaphthalene

-
A
-
132-75-2
naphthalen-1-ylacetonitrile

-
B
-
54276-84-5
6-Cyano-2,3-benzonorcaradien

-
C
-
84518-19-4
(1S)-1-endo-Cyano-1a,7b-dihydro-1H-cyclopropanaphthalene

-
D
-
84424-58-8, 84518-19-4, 97550-33-9, 97550-34-0, 97550-35-1
(1R)-1-endo-Cyano-1a,7b-dihydro-1H-cyclopropanaphthalene

-
E
-
84424-58-8
(1S)-1-exo-Cyano-1a,7b-dihydro-1H-cyclopropanaphthalene

| Conditions | Yield |
|---|---|
| In benzene for 0.5h; Product distribution; Irradiation; | A 19% B 14% C n/a D n/a E n/a |


| Conditions | Yield |
|---|---|
| With iron(III) chloride at 175℃; | |
| With iron(III) oxide; potassium bromide |

| Conditions | Yield |
|---|---|
| With cyanogen chloride at 650℃; | |
| Multi-step reaction with 2 steps 1: N-bromosuccinimide / CCl4 / 4 h / 80 °C / Irradiation 2: 18-crown-6 / dimethylsulfoxide / 20 h View Scheme | |
| Multi-step reaction with 2 steps 1: bromine / 190 - 230 °C / UV-Licht 2: sodium cyanide; potassium cyanide; aqueous methanol View Scheme | |
| Multi-step reaction with 2 steps 1: N-Bromosuccinimide; dibenzoyl peroxide / tetrachloromethane / Inert atmosphere; Reflux 2: tetra-(n-butyl)ammonium iodide / acetonitrile / Inert atmosphere; Reflux View Scheme | |
| With cyanogen chloride at 650℃; |

| Conditions | Yield |
|---|---|
| With methanol; potassium cyanide; sodium cyanide | |
| With potassium cyanide; sodium cyanide; ethanol | |
| With sodium cyanide; water |

| Conditions | Yield |
|---|---|
| With ethanol |

| Conditions | Yield |
|---|---|
| With potassium cyanide; sodium cyanide; ethanol | |
| With methanol; potassium cyanide; sodium cyanide |

| Conditions | Yield |
|---|---|
| In ethanol |
-
-
91-20-3
naphthalene

-
-
107-14-2
chloroacetonitrile

-
A
-
132-75-2
naphthalen-1-ylacetonitrile

-
B
-
7498-57-9
2-naphthaleneacetonitrile

| Conditions | Yield |
|---|---|
| In acetonitrile for 22h; Kinetics; Irradiation; |

-
-
151-50-8
potassium cyanide

-
-
3163-27-7
2-(bromomethyl)naphthalene

-
-
132-75-2
naphthalen-1-ylacetonitrile

| Conditions | Yield |
|---|---|
| 18-crown-6 ether In dimethyl sulfoxide for 20h; |

| Conditions | Yield |
|---|---|
| With lithium aluminium tetrahydride; aluminium trichloride | 100% |
| With nickel dichloride; diborane In tetrahydrofuran; methanol for 1h; Ambient temperature; | 80% |
| With hydrogenchloride; hydrogen; palladium on activated charcoal In methanol at 20℃; under 1810.02 Torr; for 2.5h; | 60% |
-
-
132-75-2
naphthalen-1-ylacetonitrile

-
-
2082-66-8
2-chlorobenzylthiocyanate

-
-
5518-09-2
2-(naphthalen-1-yl)malononitrile

| Conditions | Yield |
|---|---|
| With lithium diisopropyl amide In benzene at 5℃; for 1h; | 100% |
| With hydrogenchloride; lithium diisopropyl amide In benzene | 32% |
-
-
132-75-2
naphthalen-1-ylacetonitrile

-
-
117070-77-6
5-(1-Naphthylmethyl)-1H-tetrazole

| Conditions | Yield |
|---|---|
| With sodium azide; triethylamine hydrochloride In toluene at 110℃; for 16h; Inert atmosphere; | 97% |
| With sodium azide; cerium(III) chloride heptahydrate In water; isopropyl alcohol at 160℃; for 4h; Microwave irradiation; | 94% |
| With sodium azide; acetic acid; isopropyl alcohol |
-
-
132-75-2
naphthalen-1-ylacetonitrile

-
-
74-88-4
methyl iodide

-
-
55615-31-1
2-methyl-2-(naphthalen-1-yl)propanenitrile

| Conditions | Yield |
|---|---|
| Stage #1: naphthalen-1-ylacetonitrile With lithium diisopropyl amide In tetrahydrofuran at -78℃; for 0.666667h; Stage #2: methyl iodide In tetrahydrofuran at -78 - 20℃; for 10h; | 96% |
| With lithium diisopropyl amide In tetrahydrofuran; N,N,N,N,N,N-hexamethylphosphoric triamide at -78℃; | 83% |
| Stage #1: naphthalen-1-ylacetonitrile With sodium hydride In N,N-dimethyl-formamide at 20℃; for 0.5h; Metallation; Stage #2: methyl iodide In N,N-dimethyl-formamide for 1h; Methylation; | 78% |
-
-
132-75-2
naphthalen-1-ylacetonitrile

-
-
60-23-1
Cysteamine

-
-
18732-66-6
2-(1-naphthylmethyl)-4,5-dihydrothiazole

| Conditions | Yield |
|---|---|
| In ethanol Heating; | 95% |
-
-
132-75-2
naphthalen-1-ylacetonitrile

-
-
106-95-6
allyl bromide

-
-
56477-54-4
2-allyl-2-(1-naphthyl)pent-4-enenitrile

| Conditions | Yield |
|---|---|
| Stage #1: naphthalen-1-ylacetonitrile With lithium diisopropyl amide In tetrahydrofuran at -78℃; for 0.666667h; Stage #2: allyl bromide In tetrahydrofuran at -78 - 20℃; for 10h; | 95% |
-
-
132-75-2
naphthalen-1-ylacetonitrile

-
-
3376-23-6, 7372-59-0, 59862-60-1
C-phenyl-N-methylnitrone

-
-
79271-56-0
Triethylsilyl trifluoromethanesulfonate

| Conditions | Yield |
|---|---|
| With triethylamine In 1,2-dichloro-ethane at -30℃; for 0.5h; Inert atmosphere; | 95% |


| Conditions | Yield |
|---|---|
| Stage #1: naphthalen-1-ylacetonitrile; 1-methyl-3,4,dihydroisoquinoline With manganese; chloro-trimethyl-silane; bis(cyclopentadienyl)titanium(IV) diphenoxide; triethylamine hydrochloride In tetrahydrofuran at 60℃; for 24h; Schlenk technique; Inert atmosphere; Stage #2: With hydrogenchloride; water In tetrahydrofuran; diethyl ether; dichloromethane Cooling; regioselective reaction; | 95% |

| Conditions | Yield |
|---|---|
| With water; 1,8-diazabicyclo[5.4.0]undec-7-ene at 20℃; for 40h; Green chemistry; diastereoselective reaction; | 95% |

| Conditions | Yield |
|---|---|
| With water; potassium carbonate In isopropyl alcohol at 150℃; for 0.25h; Microwave irradiation; | 94% |
| With hydrogenchloride | |
| With sulfuric acid; water at 120 - 125℃; |
-
-
132-75-2
naphthalen-1-ylacetonitrile

-
-
100-53-8
phenylmethanethiol

-
-
27869-65-4
(Z)-2-(naphthalen-1-yl)-3-phenylacrylonitrile

| Conditions | Yield |
|---|---|
| With 1,10-Phenanthroline; potassium tert-butylate In toluene at 120℃; for 24h; | 94% |
-
-
132-75-2
naphthalen-1-ylacetonitrile

-
-
144223-33-6
2-furoyl-1H-1,2,3-benzotriazole

-
-
63753-52-6
3-(2-furyl)-2-(1-naphthyl)-3-oxopropanenitrile

| Conditions | Yield |
|---|---|
| Stage #1: naphthalen-1-ylacetonitrile With potassium tert-butylate In dimethyl sulfoxide at 10℃; for 0.166667h; Stage #2: 2-furoyl-1H-1,2,3-benzotriazole In dimethyl sulfoxide at 25℃; for 12h; | 92% |
-
-
109-65-9
1-bromo-butane

-
-
132-75-2
naphthalen-1-ylacetonitrile

-
-
94910-78-8
α,α-Dibutyl-(1-naphthyl)-acetonitril

| Conditions | Yield |
|---|---|
| Stage #1: naphthalen-1-ylacetonitrile With lithium diisopropyl amide In tetrahydrofuran at -78℃; for 0.666667h; Stage #2: 1-bromo-butane In tetrahydrofuran at -78 - 20℃; for 10h; | 92% |

| Conditions | Yield |
|---|---|
| With potassium phosphate buffer at 30℃; for 96h; Rhodococcus sp. AJ270 cells; | 91.3% |
| Stage #1: naphthalen-1-ylacetonitrile With sodium hydroxide In water for 3h; Reflux; Stage #2: With hydrogenchloride In water pH=2; Time; | 74.8% |
| With sulfuric acid; acetic acid |

| Conditions | Yield |
|---|---|
| With sodium hydroxide; 5,11,17,23,29,35-hexakis-37,38,39,40,41,42-hexamethoxycalix<6>arene In di-isopropyl ether at 70℃; for 5h; Product distribution; Further Variations:; Catalysts; | 91% |
| With sodium hydroxide; calix[8]arene with trimethylammoniomethyl groups In various solvent(s) at 70℃; for 2h; Alkylation; | 78% |
-
-
132-75-2
naphthalen-1-ylacetonitrile

-
-
75-36-5
acetyl chloride

-
-
39739-56-5
methyl-2-(1-naphthalenyl)acetimidate hydrochloride

| Conditions | Yield |
|---|---|
| In methanol | 91% |

| Conditions | Yield |
|---|---|
| With lithium hexamethyldisilazane In tetrahydrofuran at 60℃; under 760.051 Torr; for 6h; Inert atmosphere; Sealed tube; | 90% |

| Conditions | Yield |
|---|---|
| With ruthenium-grafted hydrotalcite at 180℃; for 20h; | 89% |
| ruthenium-grafted hydrotalcite at 180℃; for 20h; | 89% |
-
-
132-75-2
naphthalen-1-ylacetonitrile

-
-
17649-87-5
3-methyl-4,4-bis(methylthio)but-3-en-2-one

| Conditions | Yield |
|---|---|
| With sodium hydride In N,N-dimethyl-formamide at 0 - 20℃; | 89% |

| Conditions | Yield |
|---|---|
| With lithium tert-butoxide In N,N-dimethyl-formamide at 20℃; for 0.5h; Inert atmosphere; regioselective reaction; | 89% |
1-Naphthyl acetonitrile Chemical Properties
Product Name: 1-Naphthyl acetonitrile (CAS NO.132-75-2)
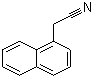
Molecular Formula: C12H9N
Molecular Weight: 167.21g/mol
Mol File: 132-75-2.mol
EINECS: 205-078-1
Appearance: White to yellow solid or melt
Melting Point: 33-35 °C(lit.)
Boiling point: 323.9 °C at 760 mmHg
Flash Point: 180.3 °C
Density: 1.116 g/cm3
Refractive index: n20/D 1.6192(lit.)
Index of Refraction: 1.634
Molar Refractivity: 53.55 cm3
Molar Volume: 149.7 cm3
Surface Tension: 48.8 dyne/cm
Enthalpy of Vaporization: 58 kJ/mol
Vapour Pressure: 0.000255 mmHg at 25°C
XLogP3-AA: 2.9
H-Bond Donor: 0
H-Bond Acceptor: 1
Structure Descriptors of 1-Naphthyl acetonitrile (CAS NO.132-75-2):
IUPAC Name: 2-naphthalen-1-ylacetonitrile
Canonical SMILES: C1=CC=C2C(=C1)C=CC=C2CC#N
InChI: InChI=1S/C12H9N/c13-9-8-11-6-3-5-10-4-1-2-7-12(10)11/h1-7H,8H2
InChIKey: OQRMWUNUKVUHQO-UHFFFAOYSA-N
Product Categories: Intermediates of Dyes and Pigments; Aromatic Nitriles
1-Naphthyl acetonitrile Uses
1-Naphthyl acetonitrile (CAS NO.132-75-2) is used in organic synthesis.
1-Naphthyl acetonitrile Toxicity Data With Reference
| 1. | ivn-mus LD50:180 mg/kg | CSLNX* U.S. Army Armament Research & Development Command, Chemical Systems Laboratory, NIOSH Exchange Chemicals. (Aberdeen Proving Ground, MD 21010) NX#03835 . |
1-Naphthyl acetonitrile Consensus Reports
Cyanide and its compounds are on the Community Right-To-Know List. Reported in EPA TSCA Inventory.
1-Naphthyl acetonitrile Safety Profile
Poison by intravenous route. When heated to decomposition it emits toxic fumes of NOx and CN−. See also NITRILES.
Safety Information of 1-Naphthyl acetonitrile (CAS NO.132-75-2):
Hazard Codes: Xn
Risk Statements: 20/21/22-36/37/38-22
R20/21/22:Harmful by inhalation, in contact with skin and if swallowed.
R36/37/38:Irritating to eyes, respiratory system and skin.
R22:Harmful if swallowed.
Safety Statements: 26-37/39-24/25-37-36
S26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice.
S37/39:Wear suitable gloves and eye/face protection.
S24/25:Avoid contact with skin and eyes.
S37:Wear suitable gloves.
S36:Wear suitable protective clothing.
RIDADR: 3276
WGK Germany: 3
RTECS: AM1240000
HazardClass: 6.1
PackingGroup: III
1-Naphthyl acetonitrile Specification
1-Naphthyl acetonitrile , its CAS NO. is 132-75-2, the synonyms are Acetonitrile, (1-naphthyl)- ; alpha-(1-Naphthyl)acetonitrile ; alpha-Naphthyl acetonitrile ; alpha-Naphthylacetonitrile ; 1-Naphthaleneacetonitrile ; 1-Naphthylacetonitrile .
Related Products
- 1-Naphthyl 2-acetamido-3,4,6-tri-O-acetyl-2-deoxy-beta-D-glucopyranoside
- 1-Naphthyl acetate
- 1-Naphthyl acetonitrile
- 1-NAPHTHYL CHLOROFORMATE
- 1-Naphthyl isocyanate
- 1-Naphthyl isothiocyanate
- 1-NAPHTHYL METHYLNITROSOCARBAMATE
- 1-Naphthyl phosphate monosodium salt monohydrate
- 1-NAPHTHYL THIOACETAMIDE
- 1-Naphthylacetamide
- 1327-52-2
- 13275-25-7
- 1327-53-3
- 13275-68-8
- 1327-57-7
- 13276-52-3
- 132767-55-6
- 1327-69-1
- 132769-75-6
- 132769-77-8
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View