-
Name
4-Chlorobenzotrichloride
- EINECS 226-009-1
- CAS No. 5216-25-1
- Article Data26
- CAS DataBase
- Density 1.505 g/cm3
- Solubility decomposes in water
- Melting Point 5.8 °C
- Formula C7H4Cl4
- Boiling Point 245 °C at 760 mmHg
- Molecular Weight 229.921
- Flash Point 109.9 °C
- Transport Information UN 1760 8/PG 2
- Appearance Clear colorless liquid
- Safety 53-45
- Risk Codes 45-21/22-37/38-48/23-62
-
Molecular Structure
-
Hazard Symbols
 T
T
- Synonyms Toluene, p,a,a,a-tetrachloro- (6CI,7CI,8CI);1-Chloro-4-(trichloromethyl)benzene;4,a,a,a-Tetrachlorotoluene;p,a,a,a-Tetrachlorotoluene;p-(Trichloromethyl)chlorobenzene;p-Chloro-a,a,a-trichlorotoluene;p-Chlorophenyltrichloromethane;a,a,a,4-Tetrachlorotoluene;a,a,a-Trichloro-4-chlorotoluene;
- PSA 0.00000
- LogP 4.16670
Synthetic route

-
A
-
5216-25-1
4-chlorotrichloromethylbenzene

-
B
-
98555-85-2
(4-trichloromethylphenyl)phosphonic dichloride

| Conditions | Yield |
|---|---|
| With sulfur dioxide for 1h; | A n/a B 87% |

| Conditions | Yield |
|---|---|
| In tetrachloromethane | 83.9% |

| Conditions | Yield |
|---|---|
| With P,P-dichlorophenylphosphine oxide; phosphorus pentachloride at 160℃; for 5h; | 64.9% |
| With phosphorus pentachloride |

| Conditions | Yield |
|---|---|
| With P,P-dichlorophenylphosphine oxide; phosphorus pentachloride at 160℃; for 5h; | 54.6% |

| Conditions | Yield |
|---|---|
| With phosphorus pentachloride at 108 - 112℃; |
-
-
100-44-7
benzyl chloride

-
A
-
5216-25-1
4-chlorotrichloromethylbenzene

-
B
-
855753-65-0
1,2,3,4,5,6-hexachloro-1-dichloromethyl-cyclohexane

-
C
-
98-07-7
Benzotrichlorid

| Conditions | Yield |
|---|---|
| Chlorierung im Sonnenlicht; |


| Conditions | Yield |
|---|---|
| With iodine durch Chlorieren; |

| Conditions | Yield |
|---|---|
| With chlorine at 150 - 200℃; |
-
-
13940-94-8
(4-chlorophenyl)dichloromethane

-
-
5216-25-1
4-chlorotrichloromethylbenzene

| Conditions | Yield |
|---|---|
| With hypochlorous anhydride In tetrachloromethane at 25℃; Rate constant; |
-
-
7647-18-9
antimonypentachloride

-
-
98-07-7
Benzotrichlorid

-
A
-
5216-25-1
4-chlorotrichloromethylbenzene

-
B
-
2136-81-4
3-chlorobenzotrichloride

-
C
-
2136-89-2
2-chlorobenzotrichloride

| Conditions | Yield |
|---|---|
| at 0 - 50℃; Einleiten von Chlor; |

-
-
7553-56-2
iodine

-
-
98-07-7
Benzotrichlorid

-
A
-
5216-25-1
4-chlorotrichloromethylbenzene

-
B
-
2136-81-4
3-chlorobenzotrichloride

-
C
-
20020-72-8
2,3,4-trichloro-1-(trichloromethyl)benzene

| Conditions | Yield |
|---|---|
| at 25 - 30℃; Einleiten von Chlor; |


-
-
5216-25-1
4-chlorotrichloromethylbenzene

| Conditions | Yield |
|---|---|
| With phosphorus pentachloride at 290 - 300℃; |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: Cl2 / CCl4 / 8 h / Heating; Irradiation 2: SO2 / 1 h View Scheme |
-
-
5216-25-1
4-chlorotrichloromethylbenzene

-
-
108-90-7
chlorobenzene

-
-
74-11-3
para-chlorobenzoic acid

-
-
90-98-2
4,4'-Dichlorobenzophenone

| Conditions | Yield |
|---|---|
| With iron(III) oxide at 145℃; under 4500.45 Torr; for 7h; | 95.7% |

-
-
1121-24-0
ortho-mercaptophenol

-
-
5216-25-1
4-chlorotrichloromethylbenzene

-
-
58948-38-2
2-(4-Chlorophenyl)-1,3-benzoxathiolium tetrafluoroborate

| Conditions | Yield |
|---|---|
| With tetrafluoroboric acid diethyl ether for 2h; Ambient temperature; | 95% |
-
-
117-21-5
3-chlorophthalic anhydride

-
-
5216-25-1
4-chlorotrichloromethylbenzene

-
A
-
113770-84-6
3-chloro-phthaloyl dichloride

-
B
-
122-01-0
4-chloro-benzoyl chloride

| Conditions | Yield |
|---|---|
| zirconium(IV) chloride at 160℃; for 16h; | A 61% B 95% |

-
-
5216-25-1
4-chlorotrichloromethylbenzene

-
-
74-11-3
para-chlorobenzoic acid

-
-
71-43-2
benzene

-
-
134-85-0
4-chlorobenzophenone

| Conditions | Yield |
|---|---|
| With iron(III) oxide at 120℃; under 1500.15 Torr; for 8h; | 94.1% |

-
-
5216-25-1
4-chlorotrichloromethylbenzene

-
-
121-44-8
triethylamine

-
-
7461-38-3
4-chloro-N,N-diethyl-benzamide

| Conditions | Yield |
|---|---|
| With B12 complex immobilized on rhodium(III) modified anatase TiO2; air In acetonitrile at 20℃; for 3h; Irradiation; | 94% |
| With heptamethyl Coβ-perchlorato-cob(II)yrinate; tetrabutylammonium perchlorate In acetonitrile at 20℃; for 3h; Electrolysis; | 89% |
-
-
85-44-9
phthalic anhydride

-
-
5216-25-1
4-chlorotrichloromethylbenzene

-
A
-
122-01-0
4-chloro-benzoyl chloride

-
B
-
88-95-9
Phthaloyl dichloride

| Conditions | Yield |
|---|---|
| zirconium(IV) chloride at 160℃; for 6h; | A 93% B 70% |


| Conditions | Yield |
|---|---|
| at 20℃; for 3h; UV-irradiation; | 93% |
-
-
5216-25-1
4-chlorotrichloromethylbenzene

| Conditions | Yield |
|---|---|
| With sodium carbonate In toluene for 12h; Reflux; | 92.3% |
-
-
5216-25-1
4-chlorotrichloromethylbenzene

| Conditions | Yield |
|---|---|
| With sodium carbonate In toluene for 12h; Reflux; | 92.3% |
-
-
5216-25-1
4-chlorotrichloromethylbenzene

| Conditions | Yield |
|---|---|
| With sodium carbonate In toluene for 12h; Reflux; | 91.3% |
-
-
5216-25-1
4-chlorotrichloromethylbenzene

| Conditions | Yield |
|---|---|
| With sodium carbonate In toluene for 12h; Reflux; | 91.3% |

| Conditions | Yield |
|---|---|
| With antimonypentachloride In dichloromethane -30 deg C 15 min, reflux 30 min; | 88% |
-
-
5216-25-1
4-chlorotrichloromethylbenzene

| Conditions | Yield |
|---|---|
| With sodium carbonate In toluene for 12h; Reflux; | 84.1% |

| Conditions | Yield |
|---|---|
| With hydrazine hydrate In ethanol for 0.666667h; Heating; | 81% |

| Conditions | Yield |
|---|---|
| With hydrogen fluoride; bromine | 81% |
-
-
5216-25-1
4-chlorotrichloromethylbenzene

| Conditions | Yield |
|---|---|
| With sodium carbonate In toluene for 12h; Reflux; | 80.1% |

| Conditions | Yield |
|---|---|
| With iron(II) chloride In acetonitrile at 25℃; for 7h; | 80% |
| With copper(l) chloride; triethyl phosphite |
-
-
91-16-7
1,2-dimethoxybenzene

-
-
5216-25-1
4-chlorotrichloromethylbenzene

-
-
79-34-5
1,1,2,2-tetrachloroethane

-
-
116412-83-0
4-chloro-3',4'-dimethoxy benzophenone

| Conditions | Yield |
|---|---|
| graphite In water | 79.5% |

-
-
5216-25-1
4-chlorotrichloromethylbenzene

-
-
756-79-6
dimethyl methane phosphonate

-
A
-
1126-46-1
Methyl 4-chlorobenzoate

-
B
-
74-87-3
methylene chloride

| Conditions | Yield |
|---|---|
| at 180℃; for 53h; | A 79% B n/a C n/a |

-
-
5216-25-1
4-chlorotrichloromethylbenzene

-
-
95-55-6
2-amino-phenol

-
-
1141-35-1
2-(4-chlorophenyl)benzoxazole

| Conditions | Yield |
|---|---|
| With Sodium borate In water at 100℃; for 10h; | 78% |
| With PPA at 100 - 120℃; for 12h; Addition; | 60% |
-
-
5216-25-1
4-chlorotrichloromethylbenzene

-
-
12775-96-1, 15158-11-9, 15721-63-8, 16941-75-6, 17493-86-6, 19498-52-3, 20499-83-6, 20499-84-7, 20499-85-8, 20499-86-9, 20573-10-8, 20573-11-9, 21595-51-7, 21595-52-8, 22206-52-6, 26445-28-3, 28959-95-7, 37362-93-9, 39417-05-5, 54603-16-6, 54603-23-5, 54603-32-6, 54603-40-6, 54603-48-4, 54603-81-5, 54603-89-3, 56316-56-4, 95985-91-4, 122297-32-9, 7440-50-8
copper

-
-
4137-78-4
4,α,α,4',α',α'-hexachloro-bibenzyl

| Conditions | Yield |
|---|---|
| With pyridine; hydrogenchloride In (2S)-N-methyl-1-phenylpropan-2-amine hydrate | 75% |

| Conditions | Yield |
|---|---|
| With antimonypentachloride In dichloromethane 1.) -30 deg C, 10 min, 2.) 23 deg C, 4 h; | 73% |

| Conditions | Yield |
|---|---|
| With antimonypentachloride In dichloromethane 1.) -30 deg C, 2.) 23 deg C, 3 h; | 71% |
-
-
5216-25-1
4-chlorotrichloromethylbenzene

-
-
1076689-72-9
4-(5-amino-1,3,4-thiadiazol-2-ylsulfanyl)benzenesulfonyl chloride

-
-
1076689-67-2
4-[5-(4-trichloromethylphenylamino)-[1,3,4]thiadiazol-2-ylsulfanyl]-benzenesulfonylchloride

| Conditions | Yield |
|---|---|
| In diethyl ether at 0 - 5℃; for 0.5h; | 71% |

-
-
5216-25-1
4-chlorotrichloromethylbenzene

| Conditions | Yield |
|---|---|
| In methanol (N2); standard Schlenk technique; MeOH and soln. of ClC6H4CCl3 in methanol were added to Ir complex; | 70% |
-
-
5216-25-1
4-chlorotrichloromethylbenzene

-
-
28144-70-9
anthranilic acid amide

-
-
7455-77-8
2-(4-chlorophenyl)-4(3H)-quinazolinone

| Conditions | Yield |
|---|---|
| With Sodium borate In water at 100℃; for 14h; | 69% |
-
-
5216-25-1
4-chlorotrichloromethylbenzene

-
-
22706-11-2
3-phenyl-4-amino-5-mercapto-1,2,4-triazole

-
-
1149378-80-2
C15H11Cl3N4S

| Conditions | Yield |
|---|---|
| Stage #1: 3-phenyl-4-amino-5-mercapto-1,2,4-triazole With potassium hydroxide In water at 20℃; Stage #2: 4-chlorotrichloromethylbenzene In water at 0℃; | 68% |

| Conditions | Yield |
|---|---|
| With oxygen; sodium hydroxide In water at 100℃; Sealed tube; Green chemistry; | 68% |
4-Chlorobenzotrichloride Chemical Properties
The Molecular Weight of p-TRICHLOROMETHYLCHLOROBENZENE(5216-25-1):229.92
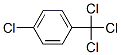
The Molecular Structure of p-TRICHLOROMETHYLCHLOROBENZENE(5216-25-1) is:
Density:1.505 g/cm3
Melting point:5.8 °C
Boiling point:245 °C at 760 mmHg
Flash point:109.9 °C
Solubility in water:decomposes
Sensitive:Moisture Sensitive
Index of Refraction:1.571
Molar Refractivity:50.18 cm3
Molar Volume:152.7 cm3
Polarizability:19.89 10-24cm3
Surface Tension:41.3 dyne/cm
Enthalpy of Vaporization:46.25 kJ/mol
Vapour Pressure:0.046 mmHg at 25°C
Appearance:clear colorless liquid.
IUPAC name:1-chloro-4-(trichloromethyl)benzene
Synonyms:4-CHLOROBENZOTRICHLORIDE;A,A,A,P-TETRACHLOROTOLUENE;1-chloro-4-(trichloromethyl)-benzen;ALPHA,ALPHA,ALPHA,4-TETRACHLOROTOLUENE;P-CHLORO-A,A,A-TRICHLOROTOLUENE;P-CHLOROBENZOTRICHLORIDE;PCTC;P-CHLORO(TRICHLOROMETHYL)BENZENE
Stability: Stable. Combustible. Incompatible with strong oxidizing agents.
4-Chlorobenzotrichloride Uses
4-Chlorobenzotrichloride Toxicity Data With Reference
| 1. | orl-rat LD50:820 mg/kg | EPASR* United States Environmental Protection Agency, Office of Pesticides and Toxic Substances. (U.S. Environmental Protection Agency, 401M St., S.W., Washington, DC 20460) History Unknown 8EHQ-0281-0360 . | ||
| 2. | ihl-rat LC50:125 mg/m3 | GTPZAB Gigiena Truda i Professionalnye Zabolevaniia. Labor Hygiene and Occupational Diseases. 28 (6)(1984),41. | ||
| 3. | orl-mus LD50:700 mg/kg | GTPZAB Gigiena Truda i Professionalnye Zabolevaniia. Labor Hygiene and Occupational Diseases. 28 (6)(1984),41. | ||
| 4. | ihl-mus LC50:125 mg/m3 | GTPZAB Gigiena Truda i Professionalnye Zabolevaniia. Labor Hygiene and Occupational Diseases. 28 (6)(1984),41. | ||
| 5. | skn-rbt LD50:>2 g/kg | EPASR* United States Environmental Protection Agency, Office of Pesticides and Toxic Substances. (U.S. Environmental Protection Agency, 401M St., S.W., Washington, DC 20460) History Unknown 8EHQ-0281-0360 . |
4-Chlorobenzotrichloride Consensus Reports
4-Chlorobenzotrichloride Safety Profile
Suspected carcinogen. A poison by inhalation. Moderately toxic by ingestion and skin contact. When heated to decomposition it emits toxic vapors of Cl−.
Health Hazard ACUTE/CHRONIC HAZARDS: Corrosive; Lachrymator.
Hazard Codes:T
Risk Statements
R45:May cause cancer.
R21/22:Harmful in contact with skin and if swallowed .
R37/38:Irritating to respiratory system and skin .
R48/23:Toxic: danger of serious damage to health by prolonged exposure through inhalation .
R62:Possible risk of impaired fertility.
Safety Statements
S53:Avoid exposure - obtain special instruction before use .
S45:In case of accident or if you feel unwell, seek medical advice immediately (show label where possible) .
RIDADR:UN 1760 8/PG 2
WGK Germany:3
RTECS:XT8580000
HazardClass:8
PackingGroup:II
4-Chlorobenzotrichloride Standards and Recommendations
4-Chlorobenzotrichloride Specification
Reactivity Profile: Simple aromatic halogenated organic compounds, such as 4-Chlorobenzotrichloride , are very unreactive. Reactivity generally decreases with increased degree of substitution of halogen for hydrogen atoms. Materials in this group may be incompatible with strong oxidizing and reducing agents. Also, they may be incompatible with many amines, nitrides, azo/diazo compounds, alkali metals, and epoxides.
Conditions to Avoid: Incompatible materials, exposure to moist air or water.
Chemical Stability: Moisture sensitive.
Incompatibilities with Other Materials: Strong oxidizing agents.
Hazardous Decomposition Products: Hydrogen chloride, carbon monoxide, carbon dioxide.
Hazardous Polymerization: Has not been reported.
Related Products
- 4-Chlorobenzotrichloride
- 5216-36-4
- 52168-48-6
- 52173-35-0
- 521-74-4
- 52175-10-7
- 52176-13-3
- 52178-50-4
- 52179-26-7
- 52179-72-3
- 52179-74-5
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View