-
Name
4-Methylbenzenesulfonhydrazide
- EINECS 216-407-3
- CAS No. 1576-35-8
- Article Data120
- CAS DataBase
- Density 1.298 g/cm3
- Solubility water: 5 g/L (15 °C)
- Melting Point 103-108 °C(lit.)
- Formula C7H10N2O2S
- Boiling Point 340.5 °C at 760 mmHg
- Molecular Weight 186.235
- Flash Point 159.7 °C
- Transport Information UN 3226 4.1
- Appearance white to off-white powder
- Safety 16
- Risk Codes 11-22-44
-
Molecular Structure
-
Hazard Symbols
 F,
F, Xn
Xn
- Synonyms p-Toluenesulfonylhydrazine;p-Tosyl hydrazide;Unifor H;p-Methylbenzenesulfonic acid hydrazide;p-Methylphenylsulfonylhydrazine;p-Toluenesulfonyl hydrazide;(4-Tolylsulfonyl)hydrazide;4-Methylbenzenesulfonicacid hydrazide;Cellmic H;
- PSA 80.57000
- LogP 2.31900
Synthetic route

| Conditions | Yield |
|---|---|
| With carbazic acid In neat (no solvent) at 100℃; for 5h; Green chemistry; | 94% |
| Multi-step reaction with 2 steps 1: triethylamine / acetonitrile / 0.01 h / 20 °C / Sonication 2: hydrazine hydrate / 0.02 h / Sonication View Scheme |

| Conditions | Yield |
|---|---|
| With hydrazine hydrate In tetrahydrofuran; water at 0 - 20℃; | 92% |
| With hydrazine hydrate In tetrahydrofuran; water at 5℃; | 92% |
| With hydrazine hydrate In dichloromethane for 0.25h; | 92% |
-
-
260788-12-3
tert-butyl 2-tosylhydrazine-1-carboxylate

-
-
1576-35-8
toluene-4-sulfonic acid hydrazide

| Conditions | Yield |
|---|---|
| With trifluoroacetic acid for 0.5h; | 78% |
-
-
151416-59-0
dimethyl 4-amino-1-(p-toluenesulfonyl)pyrazolo<3,4-b>pyridine-5,6-dicarboxylate

-
A
-
1576-35-8
toluene-4-sulfonic acid hydrazide

| Conditions | Yield |
|---|---|
| With hydrazine hydrate In ethanol 1.) reflux, 2 h, 2.) from 150 deg C to 200 deg C, 20 min.; | A n/a B 76% |

| Conditions | Yield |
|---|---|
| With hydrazine at 21℃; Rate constant; |
-
-
7803-57-8
hydrazine hydrate

-
-
98-59-9
p-toluenesulfonyl chloride

-
-
1576-35-8
toluene-4-sulfonic acid hydrazide

| Conditions | Yield |
|---|---|
| With water; benzene |
-
-
1666-17-7
(E)-N′-benzylidene-4-methylbenzenesulfonohydrazide

-
-
1576-35-8
toluene-4-sulfonic acid hydrazide

| Conditions | Yield |
|---|---|
| With water In phosphate buffer; ethanol at 25℃; pH=2.47; Kinetics; Further Variations:; pH-values; |
-
-
1666-17-7
(E)-N′-benzylidene-4-methylbenzenesulfonohydrazide

-
A
-
100-52-7
benzaldehyde

-
B
-
1576-35-8
toluene-4-sulfonic acid hydrazide

| Conditions | Yield |
|---|---|
| With water In ethanol for 3.8h; pH=2.47; Rate constant; Phosphoric acid buffer; |
-
-
62460-99-5
(E)-4-methyl-N′-(1-phenylethylidene)benzenesulfonohydrazide

-
A
-
98-86-2
acetophenone

-
B
-
1576-35-8
toluene-4-sulfonic acid hydrazide

| Conditions | Yield |
|---|---|
| With water In ethanol pH=2.47; Rate constant; Phosphoric acid buffer; | |
| With water In ethanol pH=4.48; Rate constant; Citric acid buffer; |

| Conditions | Yield |
|---|---|
| With hydrazine hydrate for 0.0166667h; Sonication; |

| Conditions | Yield |
|---|---|
| With hydrazine hydrate In methanol Reflux; Inert atmosphere; |
-
-
108-94-1
cyclohexanone

-
-
1576-35-8
toluene-4-sulfonic acid hydrazide

-
-
4545-18-0
N'-cyclohexylidene-4-methylbenzene-1-sulfonohydrazide

| Conditions | Yield |
|---|---|
| In methanol at 20℃; for 3h; | 100% |
| In methanol at 20℃; Inert atmosphere; | 99.5% |
| With hydrogenchloride In methanol for 24h; Heating; | 97% |
-
-
66-77-3
1-naphthaldehyde

-
-
1576-35-8
toluene-4-sulfonic acid hydrazide

-
-
19350-73-3
(Z)-4-methyl-N'-(naphthalen-1-ylmethylene) benzenesulfonohydrazide

| Conditions | Yield |
|---|---|
| In diethyl ether for 1h; Ambient temperature; | 100% |
| In methanol | 85% |
| In methanol at 60℃; |
-
-
120-92-3
cyclopentanone

-
-
1576-35-8
toluene-4-sulfonic acid hydrazide

-
-
17529-98-5
cyclopentanone p-tolylsulfonylhydrazone

| Conditions | Yield |
|---|---|
| In ethanol at 100℃; for 1h; | 100% |
| In ethanol at 100℃; for 1.66667h; Inert atmosphere; | 100% |
| In methanol at 20℃; Inert atmosphere; | 99.5% |

| Conditions | Yield |
|---|---|
| for 3h; | 100% |
-
-
529-34-0
3,4-dihydronaphthalene-1(2H)-one

-
-
1576-35-8
toluene-4-sulfonic acid hydrazide

-
-
17336-59-3
1-tetralone tosylhydrazone

| Conditions | Yield |
|---|---|
| In methanol at 20℃; Schlenk technique; | 100% |
| In methanol at 20℃; | 88% |
| Stage #1: 3,4-dihydronaphthalene-1(2H)-one With methanol for 0.25h; Heating / reflux; Stage #2: toluene-4-sulfonic acid hydrazide With toluene-4-sulfonic acid In methanol for 4.5h; Heating / reflux; | 87% |
-
-
502-49-8
cycloactanone

-
-
1576-35-8
toluene-4-sulfonic acid hydrazide

-
-
2567-85-3
cyclooctanone p-tolylsulfonylhydrazone

| Conditions | Yield |
|---|---|
| In ethanol at 100℃; for 1h; | 100% |
| In ethanol at 100℃; for 1.66667h; Inert atmosphere; | 100% |
| In methanol at 20℃; Schlenk technique; | 100% |
-
-
5164-64-7
bicyclo[2.1.1]hexan-2-one

-
-
1576-35-8
toluene-4-sulfonic acid hydrazide

-
-
62708-51-4
bicyclo<2.1.1>hexan-2-one (p-tolylsulfonyl)hydrazone

| Conditions | Yield |
|---|---|
| In methanol for 8h; Heating; | 100% |
-
-
10265-39-1
7,7-dimethoxynorbornan-2-one

-
-
1576-35-8
toluene-4-sulfonic acid hydrazide

-
-
53329-08-1, 74035-49-7
7,7-dimethoxybicyclo<2.2.1>heptan-2-one p-toluenesulfonylhydrazone

| Conditions | Yield |
|---|---|
| In methanol for 24h; Ambient temperature; | 100% |
-
-
6102-41-6, 6102-44-9, 14116-78-0, 70749-10-9, 70749-11-0, 78341-47-6
rel-(4aS,8aS)-1,2,3,4,4a,5,8,8a-octahydronaphthalen-2-one

-
-
1576-35-8
toluene-4-sulfonic acid hydrazide

-
-
111219-19-3
C17H22N2O2S

| Conditions | Yield |
|---|---|
| In ethanol for 1h; Heating; | 100% |
-
-
2009-81-6
2-dimethylaminomethylene-1,3-bis(dimethylimonio)propane diperchlorate

-
-
1576-35-8
toluene-4-sulfonic acid hydrazide

-
-
137305-40-9
1-(4'-Methylphenylsulfonyl)-4-pyrazolecarboxaldehyde 4'-methylphenylsulfonylhydrazone

| Conditions | Yield |
|---|---|
| With sodium carbonate In ethanol Ambient temperature; | 100% |
-
-
132272-85-6
dimethyl cis-1,2-dimethyl-5-oxocyclohex-3-ene-1,2-dicarboxylate

-
-
1576-35-8
toluene-4-sulfonic acid hydrazide

| Conditions | Yield |
|---|---|
| In methanol 1/ 21 h, RT, 2/ 20 min, reflux; | 100% |
-
-
129001-03-2, 129098-99-3
(1S,5R)-9-(tert-butoxycarbonyl)-5-(methoxycarbonyl)-9-azabicyclo<4.2.1>-nonan-2-one

-
-
1576-35-8
toluene-4-sulfonic acid hydrazide

-
-
129001-08-7, 129099-07-6, 129099-08-7, 129099-09-8
(1S,5S)-9-(tert-butoxycarbonyl)-5-(methoxycarbonyl)-9-azabicyclo<4.2.1>nonan-2-one (4-tolylsulfonyl)hydrazone

| Conditions | Yield |
|---|---|
| With pyridine; succinic acid anhydride; dmap; 3 A molecular sieve; pyridinium p-toluenesulfonate 1) CH2Cl2, RT, 3 h; 2) overnight; | 100% |
-
-
132235-18-8
pentaspiro[2.0.3.0.3.0.3.0.3.0]nonadecane-1-carbaldehyde

-
-
1576-35-8
toluene-4-sulfonic acid hydrazide

-
-
132235-19-9
N-pentaspiro[2.0.3.0.3.0.3.0.3.0]nonadec-1-ylidene-N'-(p-tosyl)hydrazine

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane | 100% |
-
-
132235-24-6
hexaspiro[2.0.3.0.3.0.3.0.3.0.3.0]tricosane-1-carbaldehyde

-
-
1576-35-8
toluene-4-sulfonic acid hydrazide

-
-
132235-25-7
N-hexaspiro[2.0.3.0.3.0.3.0.3.0.3.0]tricosan-1-ylidene-N'-(p-tosyl)hydrazine

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane | 100% |
-
-
132235-32-6
[2-13C]hexaspiro[2.0.3.0.3.0.3.0.3.0.3.0]tricosane-1-carbaldehyde

-
-
1576-35-8
toluene-4-sulfonic acid hydrazide

-
-
132235-33-7
[2-13C]-N-hexaspiro[2.0.3.0.3.0.3.0.3.0.3.0]tricosan-1-ylidene-N'-(p-tosyl)hydrazine

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane | 100% |
-
-
78167-14-3
9-Chloro-2,3-dihydro-1-thiaphenalen-3-one

-
-
1576-35-8
toluene-4-sulfonic acid hydrazide

-
-
78167-15-4
9-Chloro-2,3-dihydro-1-thiaphenalene-3-tosylhydrazone

| Conditions | Yield |
|---|---|
| In ethanol for 6h; Heating; | 100% |
-
-
111476-69-8
methyl 4-O-acetyl-2-O-benzoyl-6-deoxy-α-L-arabinohexopyranosid-3-ulose

-
-
1576-35-8
toluene-4-sulfonic acid hydrazide

-
-
113668-65-8
methyl 4-O-acetyl-2-O-benzoyl-6-deoxy-α-L-arabinohexapyranosid-3-ulose (p-tolylsulfonyl)hydrazone

| Conditions | Yield |
|---|---|
| In ethanol for 0.5h; Ambient temperature; | 100% |
-
-
104788-46-7
(1R,2S,7S,8S)-10-Benzoyl-4-oxo-9-oxa-10-aza-tricyclo[6.2.2.02,7]dodec-5-ene-7-carboxylic acid methyl ester

-
-
1576-35-8
toluene-4-sulfonic acid hydrazide

| Conditions | Yield |
|---|---|
| In methanol | 100% |
-
-
10265-39-1, 53585-72-1, 74035-53-3, 83829-13-4
(1R,4S)-7,7-Dimethoxy-bicyclo[2.2.1]heptan-2-one

-
-
1576-35-8
toluene-4-sulfonic acid hydrazide

-
-
53329-08-1, 73997-66-7, 74035-49-7
C16H22N2O4S

| Conditions | Yield |
|---|---|
| In methanol for 24h; Ambient temperature; | 100% |
-
-
124549-68-4, 124600-41-5
1-hydroxy-7-azatricyclo<6.3.1.02.7>dodecan-9-one

-
-
1576-35-8
toluene-4-sulfonic acid hydrazide

-
-
124549-76-4, 124600-43-7
tosylhydrazone of 1-hydroxy-7-azatricyclo<6.3.1.02.7>dodecan-9-one

| Conditions | Yield |
|---|---|
| With trimethyl orthoformate In methanol at 50℃; for 16h; | 100% |
-
-
124549-68-4, 124600-41-5
1-hydroxy-7-azatricyclo<6.3.1.02.7>dodecan-9-one

-
-
1576-35-8
toluene-4-sulfonic acid hydrazide

-
-
124549-76-4, 124600-43-7
tosylhydrazone of 1-hydroxy-7-azatricyclo<6.3.1.02.7>dodecan-9-one

| Conditions | Yield |
|---|---|
| With trimethyl orthoformate In methanol at 50℃; for 16h; | 100% |
-
-
118451-46-0
heptacyclo<7.7.0.02,6.03,15.04,12.05,10.011,16>hexadecane-7,13-dione

-
-
1576-35-8
toluene-4-sulfonic acid hydrazide

| Conditions | Yield |
|---|---|
| In ethanol for 16h; Heating; | 100% |
-
-
73586-29-5
4-methylene-2-adamantanone

-
-
1576-35-8
toluene-4-sulfonic acid hydrazide

-
-
87433-44-1, 87433-45-2, 102780-96-1
4-methylene-2-adamantanone tosylhydrazone

| Conditions | Yield |
|---|---|
| In ethanol | 100% |
-
-
1576-35-8
toluene-4-sulfonic acid hydrazide

-
-
502-42-1
cycloheptanone

-
-
56382-69-5
cycloheptanone p-tolylsulfonylhydrazone

| Conditions | Yield |
|---|---|
| In methanol at 20℃; Schlenk technique; | 100% |
| In ethanol at 100℃; for 0.25h; Inert atmosphere; | 99% |
| In ethanol for 2h; Reflux; Inert atmosphere; | 90% |
-
-
591-31-1
3-methoxy-benzaldehyde

-
-
1576-35-8
toluene-4-sulfonic acid hydrazide

-
-
19350-71-1
N'-(3-methoxybenzylidene)-4-methylbenzen sulfonohydrazide

| Conditions | Yield |
|---|---|
| In ethanol for 8h; Reflux; | 100% |
| With polystyrene sulfonic acid In water at 100℃; for 0.0833333h; Wavelength; Reagent/catalyst; Solvent; Temperature; Microwave irradiation; Green chemistry; | 92% |
| In methanol at 20℃; for 3h; | 90% |
-
-
1576-35-8
toluene-4-sulfonic acid hydrazide

-
-
173044-65-0
Benzoic acid (1R,2R,6S,7S,9R)-2-hydroxy-2,6,10,10-tetramethyl-3-oxo-11-oxa-tricyclo[7.2.1.01,6]dodec-7-yl ester

-
-
173044-66-1
3-(4-Toluenesulfonylhydrazono)-4β-hydroxy-9β-benzoyloxy-β-dihydroagarofuran

| Conditions | Yield |
|---|---|
| In ethanol for 10h; Ambient temperature; | 100% |

| Conditions | Yield |
|---|---|
| 100% |
-
-
69649-19-0
pentacyclo[5.4.0.02,6.03,10.05,8]undecane-8-one

-
-
1576-35-8
toluene-4-sulfonic acid hydrazide

| Conditions | Yield |
|---|---|
| In ethanol at 80℃; | 100% |
-
-
1576-35-8
toluene-4-sulfonic acid hydrazide

| Conditions | Yield |
|---|---|
| In toluene Heating; | 100% |
-
-
1576-35-8
toluene-4-sulfonic acid hydrazide

| Conditions | Yield |
|---|---|
| In toluene Heating; | 100% |
4-Methylbenzenesulfonhydrazide Consensus Reports
4-Methylbenzenesulfonhydrazide Specification
The Benzenesulfonic acid,4-methyl-, hydrazide, with the CAS registry number 1576-35-8, is also known as p-Toluenesulfonylhydrazine. It belongs to the product category of Aromatic Hydrazides, Hydrazines, Hydrazones and Oximes. Its EINECS number is 216-407-3. This chemical's molecular formula is C7H10N2O2S and molecular weight is 186.23. What's more, its systematic name is 4-methylbenzenesulfonohydrazide. Its classification code is Drug / Therapeutic Agent. It is stable at common pressure and temperature, and it should be sealed and stored in a cool, ventilated and dry place. Moreover, it should be protected from oxides and heat. It is used in organic synthesis. It is also used as foaming agent.
Physical properties of Benzenesulfonic acid,4-methyl-, hydrazide are: (1)ACD/LogP: 0.32; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): 0.32; (4)ACD/LogD (pH 7.4): 0.32; (5)ACD/BCF (pH 5.5): 1.03; (6)ACD/BCF (pH 7.4): 1.02; (7)ACD/KOC (pH 5.5): 35.57; (8)ACD/KOC (pH 7.4): 35.22; (9)#H bond acceptors: 4; (10)#H bond donors: 3; (11)#Freely Rotating Bonds: 2; (12)Polar Surface Area: 49 Å2; (13)Index of Refraction: 1.575; (14)Molar Refractivity: 47.46 cm3; (15)Molar Volume: 143.4 cm3; (16)Polarizability: 18.81×10-24cm3; (17)Surface Tension: 48.9 dyne/cm; (18)Density: 1.298 g/cm3; (19)Flash Point: 159.7 °C; (20)Enthalpy of Vaporization: 58.41 kJ/mol; (21)Boiling Point: 340.5 °C at 760 mmHg; (22)Vapour Pressure: 8.55E-05 mmHg at 25°C.
Preparation: this chemical can be prepared by hydrazine hydrate and ptoluenesulfonyl chloride at the ambient temperature. This reaction will need reagent sulfuric acid.
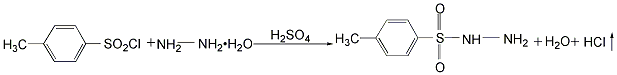
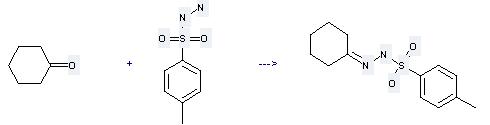
Uses of Benzenesulfonic acid,4-methyl-, hydrazide: it can be used to produce cyclohexanone (toluene-4-sulfonyl)-hydrazone at the ambient temperature. It will need solvent hexane with the reaction time of 2 hours. The yield is about 91%.
When you are using this chemical, please be cautious about it as the following:
This chemical is highly flammable, so you should keep it away from sources of ignition - No smoking. It is harmful if swallowed. Moreover, it has a risk of explosion if heated under confinement.
You can still convert the following datas into molecular structure:
(1)SMILES: O=S(=O)(c1ccc(cc1)C)NN
(2)Std. InChI: InChI=1S/C7H10N2O2S/c1-6-2-4-7(5-3-6)12(10,11)9-8/h2-5,9H,8H2,1H3
(3)Std. InChIKey: ICGLPKIVTVWCFT-UHFFFAOYSA-N
The toxicity data is as follows:
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| mouse | LD50 | intraperitoneal | 1200mg/kg (1200mg/kg) | Russian Pharmacology and Toxicology Vol. 36, Pg. 27, 1973. | |
| rat | LD50 | oral | 283mg/kg (283mg/kg) | BEHAVIORAL: SOMNOLENCE (GENERAL DEPRESSED ACTIVITY) BEHAVIORAL: CONVULSIONS OR EFFECT ON SEIZURE THRESHOLD | International Journal of Toxicology. Vol. 16. |
Related Products
- 4-Methylbenzenesulfonhydrazide
- 157636-45-8
- 157636-81-2
- 1576-37-0
- 15764-16-6
- 1576-43-8
- 1576-46-1
- 157664-47-6
- 157665-53-7
- 157666-05-2
- 157666-07-4
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View