-
Name
4-Vinylbenzoic acid
- EINECS 214-053-4
- CAS No. 1075-49-6
- Article Data57
- CAS DataBase
- Density 1.158 g/cm3
- Solubility
- Melting Point 142-144 °C(lit.)
- Formula C9H8O2
- Boiling Point 291.3 °C at 760 mmHg
- Molecular Weight 148.161
- Flash Point 132.8 °C
- Transport Information
- Appearance Off-white solid
- Safety 26-36-37/39
- Risk Codes 36/37/38
-
Molecular Structure
-
Hazard Symbols
 Xi
Xi
- Synonyms Benzoicacid, p-vinyl- (7CI,8CI);4-Carboxystyrene;4-Ethenylbenzoic acid;NSC 176003;p-Carboxystyrene;p-Vinylbenzoic acid;Benzoic acid,4-ethenyl-;
- PSA 37.30000
- LogP 2.02780
Synthetic route

| Conditions | Yield |
|---|---|
| With potassium hydroxide; disodium P-phenyl-3,3'-phosphinediyl-bis(benzenesulfonate); palladium dichloride at 90℃; for 3h; | 97% |

| Conditions | Yield |
|---|---|
| With sodium hydroxide; palladium diacetate In water at 140℃; for 3h; Hiyama reaction; | 97% |
| Stage #1: Triethoxyvinylsilane With sodium hydroxide In water at 20℃; for 0.0833333h; Sealed tube; Stage #2: 4-Bromobenzoic acid With palladium diacetate In water at 140℃; for 3h; Reagent/catalyst; Sealed tube; | 97% |

| Conditions | Yield |
|---|---|
| With 10H-phenothiazine; bis(μ-chloro)bis{(2-(1-(hydroxyimino)methyl)phenyl-C1,N)palladium(II)}; potassium acetate In N,N-dimethyl acetamide at 105℃; under 775.743 Torr; for 18h; Heck-Mizoroki reaction; Inert atmosphere; | 95% |

| Conditions | Yield |
|---|---|
| With iodine; magnesium In tetrahydrofuran Grignard reaction; | 93% |
| With diethylzinc; palladium diacetate; tert-butyl XPhos In hexanes; N,N-dimethyl acetamide at 40℃; under 7600.51 Torr; Automated synthesizer; | 62% |
| With [2,2]bipyridinyl; lithium chloride; cobalt(II) iodide; zinc In N,N-dimethyl-formamide; acetonitrile at 40℃; under 760.051 Torr; for 14h; Sealed tube; | 43% |
-
-
124-38-9
carbon dioxide

-
-
676593-23-0
2-(4-ethenylphenyl)-5,5-dimethyl-1,3,2-dioxaborinane

-
-
1075-49-6
4-ethenylbenzoic acid

| Conditions | Yield |
|---|---|
| With potassium tert-butylate; copper(l) chloride; 1,3-bis[2,6-diisopropylphenyl]imidazolium chloride In tetrahydrofuran at 70℃; under 760.051 Torr; for 24h; | 93% |
| With (1,3-bis(2,6-diisopropyl-4-(morpholinomethyl)phenyl)imidazolidin-2-ylidene)copper(I) bromide; potassium tert-butylate In tetrahydrofuran under 760.051 Torr; for 24h; Inert atmosphere; Schlenk technique; Reflux; Green chemistry; | 88% |
| Stage #1: carbon dioxide; 5,5-dimethyl-2-(4-ethenylphenyl)-1,3,2-dioxaborinane With potassium tert-butylate; silver(I) acetate; triphenylphosphine In tetrahydrofuran at 70℃; under 15201 Torr; for 16h; Inert atmosphere; Autoclave; Stage #2: With hydrogenchloride In tetrahydrofuran; water Inert atmosphere; | 82% |
| Stage #1: carbon dioxide; 5,5-dimethyl-2-(4-ethenylphenyl)-1,3,2-dioxaborinane With copper(l) iodide; 5,5-bis(4,5-dihydrooxazol-2-yl)nonane; cesium fluoride In N,N-dimethyl-formamide at 90℃; under 760.051 Torr; for 10h; Stage #2: With hydrogenchloride; water In N,N-dimethyl-formamide | 70% |

| Conditions | Yield |
|---|---|
| Stage #1: vinyl magnesium bromide With gallium(III) trichloride In tetrahydrofuran; hexane; dimethyl sulfoxide at 25℃; Stage #2: 4-iodobenzoic acid With tris-(o-tolyl)phosphine; tris(dibenzylideneacetone)dipalladium(0) chloroform complex In tetrahydrofuran; hexane; dimethyl sulfoxide Heating; | 89% |

| Conditions | Yield |
|---|---|
| Stage #1: formaldehyd; ((4-carboxyphenyl)methyl)triphenylphosphonium bromide With sodium hydroxide In water at 0 - 20℃; for 48h; Stage #2: With hydrogenchloride In water | 89% |
| With sodium hydroxide at 20℃; | 73% |

| Conditions | Yield |
|---|---|
| With 3,4-benzo-1,1,2,2-tetraethyl-1,2-disilacyclobut-3-ene; cesium fluoride In N,N-dimethyl-formamide at 0 - 20℃; under 760.051 Torr; for 2h; | 87% |
-
-
1609956-63-9
(benzo[a]acridin-12-yl)methyl 4-vinylbenzoate

-
A
-
1075-49-6
4-ethenylbenzoic acid

-
B
-
1609956-67-3
(benzo[a]acridin-12-yl)methanol

| Conditions | Yield |
|---|---|
| In water; acetonitrile for 7.16667h; Quantum yield; Photolysis; Inert atmosphere; | A 83% B n/a |

| Conditions | Yield |
|---|---|
| With oxygen at 120℃; for 16h; Green chemistry; | 83% |

| Conditions | Yield |
|---|---|
| Stage #1: 4-amino-benzoic acid Diazotization; Stage #2: potassium vinyltrifluoroborate; Pd2(μ-OAc)22 In methanol at 20℃; for 0.25h; Arylation; | 72% |
-
-
1779-49-3
Methyltriphenylphosphonium bromide

-
-
619-66-9
4-Carboxybenzaldehyde

-
-
1075-49-6
4-ethenylbenzoic acid

| Conditions | Yield |
|---|---|
| Stage #1: Methyltriphenylphosphonium bromide With n-butyllithium In tetrahydrofuran; hexane at 0℃; for 2h; Schlenk technique; Stage #2: 4-Carboxybenzaldehyde In tetrahydrofuran at 0 - 20℃; | 72% |
| Wittig Olefination; Alkaline conditions; |

| Conditions | Yield |
|---|---|
| Stage #1: 4-vinylbenzyl chloride With magnesium In tetrahydrofuran at 35℃; for 2h; Stage #2: carbon dioxide In tetrahydrofuran for 3h; | 62% |
| With iodine; magnesium 1.) tetrahydrofuran, reflux, 1 h; 2.) tetrahydrofuran, aq H2SO4; Yield given. Multistep reaction; |

| Conditions | Yield |
|---|---|
| With sodium hydroxide In methanol; water at 100℃; for 18h; Hiyama Coupling; Sealed tube; | 59% |
-
-
119072-55-8, 7188-38-7
tert-butylisonitrile

-
-
2156-04-9
4-Vinylphenylboronic acid

-
-
1075-49-6
4-ethenylbenzoic acid

| Conditions | Yield |
|---|---|
| Stage #1: tert-butylisonitrile; 4-Vinylphenylboronic acid With copper diacetate; palladium diacetate In N,N-dimethyl-formamide at 100℃; for 24h; Molecular sieve; Sealed tube; Stage #2: With water In N,N-dimethyl-formamide Molecular sieve; Sealed tube; | 49% |

| Conditions | Yield |
|---|---|
| With N,N,N',N'',N'''-pentamethyldiethylenetriamine; tetrabutylammomium bromide; palladium diacetate; DavePhos In 1-methyl-pyrrolidin-2-one at 130℃; for 12h; Inert atmosphere; chemoselective reaction; | 41% |

| Conditions | Yield |
|---|---|
| With potassium hydroxide; hydroquinone |

| Conditions | Yield |
|---|---|
| With sodium hydroxide; copper | |
| Stage #1: methyl 4-vinylbenzoate With sodium hydroxide In methanol; water at 20℃; for 2h; Stage #2: With hydrogenchloride In methanol; water pH=1; Inert atmosphere; |

| Conditions | Yield |
|---|---|
| With silver(l) oxide |
-
-
105-06-6
1,4-Divinylbenzene

-
A
-
100-21-0
terephthalic acid

-
B
-
1075-49-6
4-ethenylbenzoic acid

-
C
-
1791-26-0
4-vinyl-benzaldehyde

-
D
-
623-27-8
terephthalaldehyde,

| Conditions | Yield |
|---|---|
| With pyridine; dimethylsulfide; oxygen; ozone 1.) CH2Cl2, -78 deg C- -75 deg C, 5 hrs., 2.) CH2Cl2, r.t., 2 hrs.; Yield given. Multistep reaction; |


| Conditions | Yield |
|---|---|
| With lithium hydroxide; caesium carbonate; potassium iodide; Merrifield resin; tetrakis(triphenylphosphine) palladium(0) 1.) DMF, 80 deg C, 16 h; 2.) DMF, 60 degC, 24 h; 3.) THF, MeOH, H2O, reflux, 18 h; Yield given. Multistep reaction; |

| Conditions | Yield |
|---|---|
| With lithium hydroxide; caesium carbonate; potassium iodide; Merrifield resin; tetrakis(triphenylphosphine) palladium(0) 1.) DMF, 80 deg C, 16 h; 2.) DMF, 60 degC, 24 h; 3.) THF, MeOH, H2O, reflux, 18 h; Yield given. Multistep reaction; |

-
-
1075-49-6
4-ethenylbenzoic acid

| Conditions | Yield |
|---|---|
| With potassium hydroxide; hydroquinone |

-
-
1075-49-6
4-ethenylbenzoic acid

| Conditions | Yield |
|---|---|
| With carbon dioxide | |
| With carbon dioxide |

-
-
1075-49-6
4-ethenylbenzoic acid

| Conditions | Yield |
|---|---|
| With tetrahydrofuran; carbon dioxide |

| Conditions | Yield |
|---|---|
| With palladium diacetate; triphenylphosphine In tetrahydrofuran; methanol at 100℃; for 1h; Suzuki coupling; |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 82 percent / acetone / 6 h / Heating 2: 73 percent / aq. NaOH / 20 °C View Scheme | |
| Multi-step reaction with 2 steps 1: acetone / 3 h / Reflux 2: sodium hydroxide / water / 48 h / 0 - 20 °C View Scheme |
-
-
23359-08-2
4-formylcinnamic acid

-
-
1075-49-6
4-ethenylbenzoic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: copper (II)-acetate; quinoline; copper-powder / 300 - 310 °C 2: silver oxide View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: pyridine; ethanol 2: copper (II)-acetate; quinoline; copper-powder / 300 - 310 °C 3: silver oxide View Scheme |

| Conditions | Yield |
|---|---|
| With oxalyl dichloride In chloroform; N,N-dimethyl-formamide for 5h; Reflux; | 100% |
| With phosphorus trichloride In acetonitrile at 60℃; for 6h; Inert atmosphere; | 81% |
| With thionyl chloride In N,N-dimethyl-formamide for 2.5h; Heating; | 76% |

| Conditions | Yield |
|---|---|
| Stage #1: 4-ethenylbenzoic acid With palladium diacetate; 4,4,5,5-tetramethyl-[1,3,2]-dioxaboralane In dichloromethane at 25℃; for 12h; Schlenk technique; Inert atmosphere; Stage #2: With hydrogenchloride In water | 99% |
| With palladium diacetate; 4,4,5,5-tetramethyl-[1,3,2]-dioxaboralane In dichloromethane at 25℃; for 12h; Sealed tube; Inert atmosphere; chemoselective reaction; | 99% |
| With Decaborane; palladium on activated charcoal In methanol at 25℃; for 1h; Reduction; | 92% |
| With ammonium formate; PdMCM-41 In methanol at 69.84℃; for 5h; | 80% |

| Conditions | Yield |
|---|---|
| Stage #1: 4-ethenylbenzoic acid With thionyl chloride; N,N-dimethyl-formamide In dichloromethane at 20℃; for 2h; Stage #2: benzyl alcohol With dmap; triethylamine In dichloromethane at 0 - 20℃; for 2h; | 99% |
| With dmap; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride In dichloromethane for 43h; | 92% |
| With dmap; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride In dichloromethane for 43h; | 92% |
-
-
1075-49-6
4-ethenylbenzoic acid

-
-
98590-59-1
4-(1,2-dibromoethyl)benzoic acid

| Conditions | Yield |
|---|---|
| With bromine In chloroform at 0 - 20℃; for 4h; | 98% |
| With 1,1,1,3',3',3'-hexafluoro-propanol; N,N,N,N-tetraethylammonium tetrafluoroborate; ethylene dibromide In acetonitrile at 20℃; Inert atmosphere; Electrolysis; | 32% |
| With bromine In chloroform |
-
-
1075-49-6
4-ethenylbenzoic acid

| Conditions | Yield |
|---|---|
| With dmap; dicyclohexyl-carbodiimide In dichloromethane at 20℃; for 12h; | 98% |
-
-
1075-49-6
4-ethenylbenzoic acid

-
-
456-27-9
4-nitrobenzenediazonium tetrafluoroborate

-
-
75-05-8
acetonitrile

| Conditions | Yield |
|---|---|
| With tris(2,2'-bipyridyl)ruthenium dichloride; water at 20℃; for 4h; Meerwein Arylation; Irradiation; | 97% |

-
-
1075-49-6
4-ethenylbenzoic acid

-
-
335248-50-5
((1S,2R,1'R,2'R,1''R,2''R,1'''R,2'''R,1''''R,2''''S)-2''''-Vinyl-[1,2';1',1'';2'',1''';2''',1'''']quinquecyclopropan-2-yl)-methanol

-
-
335248-51-6
(1R,3S,4R,6S,7R,9S,10R,12S,13R,15S)-15-ethenyl-1-quinquecyclopropanemethyl 4-ethenylbenzoate

| Conditions | Yield |
|---|---|
| With dmap; dicyclohexyl-carbodiimide In dichloromethane at 20℃; Steglich esterification; | 96% |

| Conditions | Yield |
|---|---|
| With oxalyl dichloride; triethylamine In dichloromethane at -60 - 20℃; for 0.5h; Swern oxidation; | 96% |
-
-
1075-49-6
4-ethenylbenzoic acid

-
-
1325728-24-2
C22H33NO3

| Conditions | Yield |
|---|---|
| With dmap; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride; triethylamine In dichloromethane at 0 - 20℃; | 96% |

| Conditions | Yield |
|---|---|
| In chlorobenzene at 90℃; for 48h; | 96% |

| Conditions | Yield |
|---|---|
| With dmap; dicyclohexyl-carbodiimide In dichloromethane | 95% |
| With dmap; dicyclohexyl-carbodiimide In dichloromethane at 20℃; for 12h; Inert atmosphere; | 95% |
| Stage #1: 4-ethenylbenzoic acid With thionyl chloride; N,N-dimethyl-formamide In dichloromethane at 20℃; for 2h; Stage #2: ethanol With dmap; triethylamine In dichloromethane at 0 - 20℃; for 2h; | 89% |

| Conditions | Yield |
|---|---|
| With cesium fluoride In N,N-dimethyl-formamide at 20℃; for 2.5h; Inert atmosphere; | 95% |

| Conditions | Yield |
|---|---|
| Stage #1: 4-ethenylbenzoic acid With thionyl chloride; N,N-dimethyl-formamide In dichloromethane at 20℃; for 2h; Stage #2: diethylamine With dmap In dichloromethane at 0 - 20℃; for 2h; | 94% |
| With fluorosulfonyl fluoride; N-ethyl-N,N-diisopropylamine In acetonitrile at 20℃; for 5h; | 74% |

| Conditions | Yield |
|---|---|
| Stage #1: 4-ethenylbenzoic acid With dmap; dicyclohexyl-carbodiimide In dichloromethane for 0.25h; Sealed tube; Darkness; Inert atmosphere; Stage #2: C28H35FeOPRu In dichloromethane at 20℃; for 1.5h; Sealed tube; Darkness; Inert atmosphere; | 94% |

| Conditions | Yield |
|---|---|
| With iodosylbenzene; water In acetonitrile at 20℃; for 20h; diastereoselective reaction; | 93% |

| Conditions | Yield |
|---|---|
| With sulfuric acid at 20℃; for 7h; | 92% |
| With thionyl chloride | 90% |
| With bromobenzene at 55 - 60℃; under 3750.38 - 4500.45 Torr; for 6h; Autoclave; | 90% |

| Conditions | Yield |
|---|---|
| rhenium(VII) oxide In dichloromethane at 20℃; for 24h; | 92% |

-
-
1075-49-6
4-ethenylbenzoic acid

-
-
69221-14-3, 69257-04-1, 95189-65-4
(8S,9R)-(-)-N-benzylcinchonidinium chloride

-
-
1092983-06-6
N-9-benzylcinchonidinium p-vinylbenzoate

| Conditions | Yield |
|---|---|
| With sodium carbonate In dichloromethane; water | 92% |

| Conditions | Yield |
|---|---|
| With dmap; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride In N,N-dimethyl-formamide at 0 - 20℃; | 92% |
| Stage #1: 4-ethenylbenzoic acid; phenol With dmap In N,N-dimethyl-formamide at 0℃; for 0.166667h; Stage #2: With 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride In N,N-dimethyl-formamide at 0 - 20℃; | 53% |

| Conditions | Yield |
|---|---|
| In chlorobenzene at 90℃; for 48h; | 92% |
-
-
91-21-4
1,2,3,4-tetrahydroisoquinoline

-
-
931-53-3
Cyclohexyl isocyanide

-
-
1075-49-6
4-ethenylbenzoic acid

-
-
104-87-0
4-methyl-benzaldehyde

| Conditions | Yield |
|---|---|
| In toluene at 80℃; Ugi Condensation; Molecular sieve; Sealed tube; | 92% |
| In toluene at 60℃; for 17h; Molecular sieve; | 92% |


| Conditions | Yield |
|---|---|
| Stage #1: potassium cyanide With acetic acid In ethylene glycol at 60℃; Sealed tube; Stage #2: 4-ethenylbenzoic acid With bis(1,5-cyclooctadiene)nickel (0); 4,5-bis(diphenylphos4,5-bis(diphenylphosphino)-9,9-dimethylxanthenephino)-9,9-dimethylxanthene at 60℃; for 18h; Sealed tube; | 92% |
-
-
1075-49-6
4-ethenylbenzoic acid

-
-
20118-38-1
chalcone-4-carboxylic acid

| Conditions | Yield |
|---|---|
| With cadmium(II) selenide In tetrahydrofuran at 20℃; for 48h; Irradiation; diastereoselective reaction; | A n/a B 92% C n/a |

-
-
1075-49-6
4-ethenylbenzoic acid

-
-
20118-38-1
chalcone-4-carboxylic acid

| Conditions | Yield |
|---|---|
| With cadmium(II) selenide In tetrahydrofuran at 20℃; for 48h; Irradiation; diastereoselective reaction; | 92% |
-
-
1075-49-6
4-ethenylbenzoic acid

-
-
20118-38-1
4-[(1E)-3-oxo-3-phenylprop-1-en-1-yl]benzoic acid

| Conditions | Yield |
|---|---|
| With oleate-capped CdSe quantum dots In tetrahydrofuran at 20℃; for 48h; Inert atmosphere; Sealed tube; Irradiation; | 92% |
4-Vinylbenzoic acid Specification
4-Vinylbenzoic acid is an organic compound with the formula C9H8O2, and its systematic name is the same with the product name. With the CAS registry number 1075-49-6, it is also named as 4-Ethenylbenzoic acid. It belongs to the product categories of Carboxylicacid; Styrenes; Aromatics Compounds; Fluorenes, etc. (reagent for high-performance polymer research); Functional Materials; Reagent for High-Performance Polymer Research; Aromatics; Metabolites & Impurities. Its EINECS number is 214-053-4. In addition, the molecular weight is 148.16. This chemical is stable at common pressure and temperature, and it should be sealed and stored in a cool and dry place. Moreover, it should be protected from oxides. It is used as a metabolite of 1,4-diethenylbenzene .
Physical properties of 4-Vinylbenzoic are: (1)ACD/LogP: 2.18; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): 0.90; (4)ACD/LogD (pH 7.4): -0.67; (5)ACD/BCF (pH 5.5): 1.42; (6)ACD/BCF (pH 7.4): 1.00; (7)ACD/KOC (pH 5.5): 19.37; (8)ACD/KOC (pH 7.4): 1.00; (9)#H bond acceptors: 2; (10)#H bond donors: 1; (11)#Freely Rotating Bonds: 2; (12)Polar Surface Area: 37.3 Å2; (13)Index of Refraction: 1.606; (14)Molar Refractivity: 44.107 cm3; (15)Molar Volume: 127.911 cm3; (16)Polarizability: 17.485×10-24cm3; (17)Surface Tension: 46.69 dyne/cm; (18)Density: 1.158 g/cm3; (19)Flash Point: 132.833 °C; (20)Enthalpy of Vaporization: 56.052 kJ/mol; (21)Boiling Point: 291.315 °C at 760 mmHg; (22)Vapour Pressure: 0.001 mmHg at 25°C.
Preparation of 4-Vinylbenzoic: this chemical can be prepared by 5-amino-1-(3-chloro-phenyl)-1H-pyrazole-4-carboxylic acid ethyl ester at the temperature of 90 °C. This reaction will need reagents 10% aq. KOH, PdCl2, PhP(m-C6H4SO3Na)2 with the reaction time of 3 hours. The yield is about 97%.
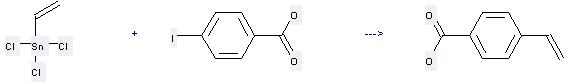
Uses of 4-Vinylbenzoic: it can be used to produce 4-ethenyl-benzoyl chloride by heating. It will need reagent SOCl2 and solvent dimethylformamide with the reaction time of 2.5 hours. The yield is about 76%.

When you are using this chemical, please be cautious about it as the following:
It is irritating to eyes, respiratory system and skin. In case of contact with eyes, you should rinse immediately with plenty of water and seek medical advice. When using it, you need to wear suitable protective clothing, gloves and eye/face protection.
You can still convert the following datas into molecular structure:
(1)SMILES: O=C(O)c1ccc(\C=C)cc1
(2)Std. InChI: InChI=1S/C9H8O2/c1-2-7-3-5-8(6-4-7)9(10)11/h2-6H,1H2,(H,10,11)
(3)Std. InChIKey: IRQWEODKXLDORP-UHFFFAOYSA-N
Related Products
- 4-Vinylbenzoic acid
- 107558-48-5
- 107559-02-4
- 1075-62-3
- 107565-44-6
- 1075-70-3
- 107572-07-6
- 107-57-3
- 1075-74-7
- 1075-76-9
- 107580-81-4
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View