-
Name
5-Chloro-1,3-xylene
- EINECS
- CAS No. 556-97-8
- Article Data10
- CAS DataBase
- Density 1,04 g/cm3
- Solubility
- Melting Point -41.95°C (estimate)
- Formula C8H9 Cl
- Boiling Point 188.1 ºC at 760 mmHg
- Molecular Weight 140.612
- Flash Point 63.1 ºC
- Transport Information
- Appearance liquid
- Safety Experimental reproductive effects. When heated to decomposition it emits toxic fumes of Cl−.
- Risk Codes 36/37/38
-
Molecular Structure
- Hazard Symbols Xi
- Synonyms m-Xylene,5-chloro- (8CI); 1-Chloro-3,5-dimethylbenzene; 3,5-Dimethylchlorobenzene;3,5-Dimethylphenyl chloride; 3-Methyl-5-chlorotoluene;5-Chloro-1,3-dimethylbenzene; 5-Chloro-m-xylene; 5-Chloro-m-xylene
- PSA 0.00000
- LogP 2.95680
Synthetic route

-
-
556-97-8
1-chloro-3,5-dimethylbenzene

| Conditions | Yield |
|---|---|
| With hydrogenchloride; copper(l) chloride | 54% |
| With hydrogenchloride; copper(l) chloride Product distribution; triazene moiety as protecting group for aromatic amines; var. triazenes; |

| Conditions | Yield |
|---|---|
| With hydrogenchloride Diazotization.Behandlung der Diazoniumsalz-Loesung mit CuCl; |

-
-
556-97-8
1-chloro-3,5-dimethylbenzene

| Conditions | Yield |
|---|---|
| With quinoline |
-
-
95-47-6
o-xylene

-
A
-
95-66-9
2,4-dimethylchlorobenzene

-
B
-
556-97-8
1-chloro-3,5-dimethylbenzene

-
C
-
95-72-7
2-chloro-1,4-dimethyl-benzene

-
D
-
6781-98-2
2,6-dimethyl-1-chlorobenzene

-
E
-
615-60-1
4-chloro-1,2-dimethylbenzene

-
F
-
608-23-1
3-chloro-o-xylene

-
G
-
20824-80-0
1,2-dichloro-4,5-dimethyl-benzene

-
H
-
68266-67-1
1,2-dichloro-3,4-dimethyl-benzene

-
I
-
34060-72-5
2-methyl-5-chlorobenzyl chloride

-
J
-
52331-02-9
1,4-dichloro-2,3-dimethyl-benzene

-
K
-
70172-92-8
3,5-dichloro-o-xylene

-
L
-
552-45-4
1-chloromethyl-2-methylbenzene

-
M
-
55676-90-9
alpha,2-dichloro-6-methyltoluene

| Conditions | Yield |
|---|---|
| With chlorine; the fluorine-containing K-L-type zeolite In 1,2-dichloro-ethane at 80℃; for 1.75 - 3.66667h; Product distribution / selectivity; | |
| With chlorine; a K-L-type zeolite In 1,4-dioxane; 1,2-dichloro-ethane at 80℃; for 6.75h; |

-
-
7758-89-6
copper(I) chloride

-
-
108-69-0
3,5-dimethylaminoaniline

-
-
556-97-8
1-chloro-3,5-dimethylbenzene

| Conditions | Yield |
|---|---|
| With hydrogenchloride; sodium nitrite In water |
-
-
556-97-8
1-chloro-3,5-dimethylbenzene

-
A
-
17057-88-4
3,5-dimethylbiphenyl

-
B
-
98-80-6
phenylboronic acid

| Conditions | Yield |
|---|---|
| With cesium fluoride; Pd(dba)2 In toluene | A n/a B 99% |

| Conditions | Yield |
|---|---|
| With ammonium hydroxide; potassium phosphate In dimethyl sulfoxide at 80℃; UV-irradiation; | 99% |
| Multi-step reaction with 2 steps 1.1: sulfuric acid; nitric acid / 30 °C / Cooling with ice 2.1: palladium on activated charcoal / ethanol / 0.5 h / 50 °C / Autoclave 2.2: 0.83 h / 65 °C / 2250.23 Torr / Autoclave View Scheme |

| Conditions | Yield |
|---|---|
| With potassium phosphate In dimethyl sulfoxide at 80℃; UV-irradiation; | 99% |

| Conditions | Yield |
|---|---|
| With cesium fluoride; Pd(dba)2 In toluene | 98% |
| With potassium phosphate; C75H58Cl2N4Pd In water; isopropyl alcohol at 25℃; for 4.5h; Reagent/catalyst; Suzuki-Miyaura Coupling; Schlenk technique; | 98% |
| With potassium phosphate; C20H28Br2N2Pd In water; isopropyl alcohol at 25℃; for 7.5h; Reagent/catalyst; Suzuki-Miyaura Coupling; | 95% |
-
-
556-97-8
1-chloro-3,5-dimethylbenzene

-
-
2835-99-6
4-amino-3-methylphenol

-
-
1198117-98-4
4-(3,5-dimethylphenylamino)-3-methyl-phenol

| Conditions | Yield |
|---|---|
| With chloro[2-(dicyclohexylphosphino)-3,6-dimethoxy-2',4',6'-triisopropyl-1,1'-biphenyl][2-(2-aminoethyl)phenyl]palladium(ll); sodium t-butanolate In 1,4-dioxane at 110℃; for 3h; Inert atmosphere; | 97% |
-
-
617-89-0
furan-2-ylmethanamine

-
-
556-97-8
1-chloro-3,5-dimethylbenzene

-
-
416887-98-4
N-(furan-2-yl-methyl)-3,5-dimethylaniline

| Conditions | Yield |
|---|---|
| With potassium phosphate In dimethyl sulfoxide at 80℃; UV-irradiation; | 97% |
-
-
556-97-8
1-chloro-3,5-dimethylbenzene

-
-
36343-05-2
N-methylheptylamine

| Conditions | Yield |
|---|---|
| With sodium t-butanolate; 3-dioxolane; Pd(dba)2*2-(2'-dicyclohexylphosphinophenyl)-2-methyl-1 In toluene at 105℃; for 1h; | 95% |
-
-
556-97-8
1-chloro-3,5-dimethylbenzene

-
-
2835-96-3
4-amino-2-methylphenol

-
-
1198117-78-0
4-[(3,5-dimethylphenyl)amino]-2-methylphenol

| Conditions | Yield |
|---|---|
| With chloro[2-(dicyclohexylphosphino)-3,6-dimethoxy-2',4',6'-triisopropyl-1,1'-biphenyl][2-(2-aminoethyl)phenyl]palladium(ll); sodium t-butanolate In 1,4-dioxane at 110℃; for 1.33333h; Inert atmosphere; | 94% |

| Conditions | Yield |
|---|---|
| With 2-(2,6-dimethoxyphenyl)-1-methyl-3-(diphenylphosphino)-1H-indole; palladium diacetate; lithium tert-butoxide In 1,4-dioxane at 100℃; for 1h; Schlenk technique; Inert atmosphere; | 94% |
-
-
110-91-8
morpholine

-
-
556-97-8
1-chloro-3,5-dimethylbenzene

-
-
16800-77-4
N (dimethyl-3,5-phenyl-1) morpholine

| Conditions | Yield |
|---|---|
| With sodium t-butanolate; 3-dioxolane; Pd(dba)2*2-(2'-dicyclohexylphosphinophenyl)-2-methyl-1 In toluene at 105℃; for 1h; | 92% |
-
-
556-97-8
1-chloro-3,5-dimethylbenzene

-
-
960-16-7
tributylphenylstannane

-
-
17057-88-4
3,5-dimethylbiphenyl

| Conditions | Yield |
|---|---|
| With potassium fluoride; tetrabutylammomium bromide; tris-(o-tolyl)phosphine; copper(I) oxide at 125 - 130℃; for 72h; Stille cross-coupling; | 92% |
| With palladium diacetate; orotic acid; cesium fluoride In 1,4-dioxane at 100℃; for 22h; Stille Cross Coupling; Sealed tube; Inert atmosphere; Schlenk technique; | 44% |
-
-
556-97-8
1-chloro-3,5-dimethylbenzene

-
-
13105-53-8
butylcarbamic acid benzyl ester

-
-
1198294-06-2
benzyl butyl(3,5-dimethylphenyl)carbamate

| Conditions | Yield |
|---|---|
| With bis(η3-allyl-μ-chloropalladium(II)); bis(3,5-bis(trifluoromethyl)phenyl)(2’,4’,6’-triisopropyl-3,6-dimethoxy-[1,1’-biphenyl]-2-yl)phosphine; caesium carbonate In toluene at 130℃; for 17h; Inert atmosphere; Molecular sieve; | 92% |
-
-
556-97-8
1-chloro-3,5-dimethylbenzene

-
-
100-61-8
N-methylaniline

-
-
474352-69-7
N,3,5-trimethyl-N-phenylaniline

| Conditions | Yield |
|---|---|
| Stage #1: 1-chloro-3,5-dimethylbenzene; N-methylaniline With palladium diacetate; sodium sulfate; sodium t-butanolate; XPhos for 1h; Buchwald-Hartwig Coupling; Milling; Stage #2: In water; ethyl acetate for 0.0333333h; Buchwald-Hartwig Coupling; Milling; | 90% |
-
-
556-97-8
1-chloro-3,5-dimethylbenzene

| Conditions | Yield |
|---|---|
| Stage #1: 1-chloro-3,5-dimethylbenzene; (Ra)-2,2'-bis(methoxy-methyloxy)-1,1'-binaphthalene With [1,3-bis(2,6-diisopropylphenyl)imidazol-2-ylidene](3-chloropyridyl)palladium(ll) dichloride; tert.-butyl lithium In n-heptane at 20℃; for 0.166667h; Schlenk technique; Stage #2: In n-heptane Schlenk technique; Acidic conditions; | 88% |
-
-
556-97-8
1-chloro-3,5-dimethylbenzene

-
-
73183-34-3
bis(pinacol)diborane

-
-
325142-93-6
2-(3,5-dimethylphenyl)-4,4,5,5-tetramethyl-1,3,2-dioxaborolane

| Conditions | Yield |
|---|---|
| With p-phenylpyridine; sodium methylate In acetonitrile at 20℃; for 12h; Sealed tube; Inert atmosphere; Irradiation; | 88% |
| With dicyclohexyl-(2',6'-dimethoxybiphenyl-2-yl)-phosphane; bis(dibenzylideneacetone)-palladium(0) In toluene at 105℃; for 18h; Sealed tube; Inert atmosphere; | 80% |
| With [1,3-bis(cyclohexyl)imidazol-2-ylidene]copper(I) chloride; potassium tert-butylate In methyl cyclohexane at 90℃; for 42h; Inert atmosphere; Glovebox; | 68% |
-
-
556-97-8
1-chloro-3,5-dimethylbenzene

-
-
18412-55-0
triethoxy(o-tolyl)silane

-
-
76708-77-5
2,3′,5′-trimethyl-1,1′-biphenyl

| Conditions | Yield |
|---|---|
| With Pd((CH3)2C6H3NC(CH3)CHC(CH3)NC6H3(CH3)2)(CH3)(P(CH2CH3)3); tetrabutylammomium bromide; sodium hydroxide In water at 60℃; for 10h; Hiyama coupling; Inert atmosphere; | 87% |
-
-
556-97-8
1-chloro-3,5-dimethylbenzene

-
-
68971-87-9
(2-tolyl)tributyltin

-
-
76708-77-5
2,3′,5′-trimethyl-1,1′-biphenyl

| Conditions | Yield |
|---|---|
| With Pd((CH3)2C6H3NC(CH3)CHC(CH3)NC6H3(CH3)2)(CH3)(P(CH2CH3)3); cesium fluoride In tetrahydrofuran at 50℃; for 8h; Stille Cross Coupling; | 87% |

| Conditions | Yield |
|---|---|
| With copper(l) iodide; C14H16N2O; caesium carbonate In N,N-dimethyl-formamide at 130℃; for 24h; Inert atmosphere; Sealed tube; | 87% |
| With copper(l) iodide; C14H16N2O; caesium carbonate In N,N-dimethyl-formamide at 130℃; for 24h; Sealed tube; Inert atmosphere; | 87% |

| Conditions | Yield |
|---|---|
| With C40H30Cl2N2NiO2S2; sodium hydroxide In N,N-dimethyl-formamide at 70℃; for 3h; | 86% |

| Conditions | Yield |
|---|---|
| With manganese; 2,9-dibutyl-4,7-dimethyl-1,10-phenanthroline; tetraethylammonium iodide; lithium acetate; cobalt(II) bromide In N,N-dimethyl acetamide at 100℃; under 760.051 Torr; for 12h; Inert atmosphere; Schlenk technique; | 84% |
-
-
556-97-8
1-chloro-3,5-dimethylbenzene

-
-
66-25-1
hexanal

-
-
959706-71-9
(2Z)-2-butyl-4-(3,5-dimethylphenyl)-2-octenal

| Conditions | Yield |
|---|---|
| With caesium carbonate; palladium diacetate In N,N-dimethyl-formamide at 80℃; for 12h; | 82% |

| Conditions | Yield |
|---|---|
| With 1,1'-bis-(diphenylphosphino)ferrocene; nickel diacetate; lithium hexamethyldisilazane In neat (no solvent) at 80℃; for 16h; Sealed tube; Glovebox; Inert atmosphere; regioselective reaction; | 82% |
-
-
556-97-8
1-chloro-3,5-dimethylbenzene

-
-
62358-81-0
1-(bromomethyl)-3-chloro-5-methylbenzene

| Conditions | Yield |
|---|---|
| With N-Bromosuccinimide; azobisisobutyronitrile In tetrachloromethane | 80% |
| With N-Bromosuccinimide In tetrachloromethane for 3h; Heating / reflux; UV-irradiation; | 68% |
| With N-Bromosuccinimide; dibenzoyl peroxide In tetrachloromethane for 0.333333h; Heating / reflux; | 60% |

| Conditions | Yield |
|---|---|
| With potassium phosphate; palladium diacetate In 1-methyl-pyrrolidin-2-one at 125℃; for 24h; | 77% |

| Conditions | Yield |
|---|---|
| With potassium hydroxide In water for 8h; Reflux; | 77% |
| With C40H30Cl2N2NiO2S2; sodium hydroxide In N,N-dimethyl-formamide at 70℃; for 3h; | 77% |
-
-
556-97-8
1-chloro-3,5-dimethylbenzene

| Conditions | Yield |
|---|---|
| With palladium diacetate; potassium carbonate; XPhos; Trimethylacetic acid In o-xylene at 190℃; for 21h; Schlenk technique; Inert atmosphere; | 77% |
-
-
556-97-8
1-chloro-3,5-dimethylbenzene

-
-
150-76-5
4-methoxy-phenol

-
-
120957-65-5
1-(4-methoxyphenoxy)-3,5-dimethylbenzene

| Conditions | Yield |
|---|---|
| With 2-acetonylpyridine; caesium carbonate; copper(I) bromide In dimethyl sulfoxide at 150℃; for 24h; Inert atmosphere; chemoselective reaction; | 70% |
-
-
556-97-8
1-chloro-3,5-dimethylbenzene

-
-
5123-13-7
5,5-dimethyl-2-phenyl-1,3,2-dioxaborinane

-
-
17057-88-4
3,5-dimethylbiphenyl

| Conditions | Yield |
|---|---|
| With potassium tert-butylate; cobalt(II) chloride; 1,3-bis[2,6-diisopropylphenyl]imidazolium chloride In tetrahydrofuran at 60℃; for 24h; Suzuki Coupling; | 69% |
-
-
556-97-8
1-chloro-3,5-dimethylbenzene

-
-
103-84-4
Acetanilid

-
-
51786-48-2
N-(3,5-dimethylphenyl)-N-phenylacetamide

| Conditions | Yield |
|---|---|
| With bis(η3-allyl-μ-chloropalladium(II)); bis(3,5-bis(trifluoromethyl)phenyl)(2’,4’,6’-triisopropyl-3,6-dimethoxy-[1,1’-biphenyl]-2-yl)phosphine; caesium carbonate In toluene at 130℃; for 17h; Inert atmosphere; Molecular sieve; | 68% |

| Conditions | Yield |
|---|---|
| With 3 A molecular sieve; caesium carbonate; palladium diacetate In N,N-dimethyl-formamide at 20 - 145℃; for 26h; | 67% |
5-Chloro-1,3-xylene Chemical Properties
Chemical Name: 5-Chloro-1,3-xylene
IUPAC NAME: 1-Chloro-3,5-dimethylbenzene
CAS No.: 556-97-8
Molecular Formula: C8H9Cl
Molecular Weight: 140.61 g/mol
Density: 1.049 g/cm3
Flash Point: 63.1 °C
Boiling Point: 188.1 °C at 760 mmHg
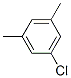
Following is the structure of 1-Chloro-3,5-xylene (CAS No.556-97-8):
Product Categories about 1-Chloro-3,5-xylene (CAS No.556-97-8) are Halogen toluene ; Chlorine Compounds
The chemical synonymous of 1-Chloro-3,5-xylene (CAS No.556-97-8) are 5-Chloro-M-Xylene ; 1-Chloro-3,5-Dimethylbenzene ; 1-Chloro-3,5-Xylene ; 3,5-Dimethylchlorobenzene ; 3,5-Dimethylphenylchloride ; 5-Chloro-1,3-Xylene ; 5-Chloro-M-Xylen ; 1,3-Dimethyl-5-Chlorobenzene
5-Chloro-1,3-xylene Safety Profile
Experimental reproductive effects. When heated to decomposition it emits toxic fumes of Cl−.
Hazard Codes:  Xi
Xi
Risk Statements about 1-Chloro-3,5-xylene (CAS No.556-97-8):
R36/37/38:Irritating to eyes, respiratory system and skin.
Safety Statements about 1-Chloro-3,5-xylene (CAS No.556-97-8):
S26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice.
S36:Wear suitable protective clothing.
S36/37/39:Wear suitable protective clothing, gloves and eye/face protection.
Related Products
- 5-Chloro-1-(2-chlorophenyl)-1H-pyrazole-4-carbonitrile
- 5-Chloro-1-(2-pyridinyl)-1H-pyrazole-4-carbonitrile
- 5-Chloro-1-(2-pyrimidinyl)-1H-pyrazole-4-carboxylic acid ethyl ester
- 5-Chloro-1-(3-chlorophenyl)-1H-pyrazole-4-carbonitrile
- 5-Chloro-1-(3-chlorophenyl)-1H-pyrazole-4-carboxylic acid ethyl ester
- 5-Chloro-1-(4-chlorophenyl)-1H-pyrazole-4-carbonitrile
- 5-Chloro-1-(4-fluorophenyl)-1H-indole
- 5-Chloro-1-(4-fluorophenyl)-1H-pyrazole-4-carbonitrile
- 5-Chloro-1-(4-fluorophenyl)-1H-pyrazole-4-carboxylic acid ethyl ester
- 5-Chloro-1-(4-methylphenyl)-1H-pyrazole-3-carboxylic acid ethyl ester
- 55699-10-0
- 55699-85-9
- 55700-14-6
- 557-00-6
- 55701-03-6
- 55701-05-8
- 55701-06-9
- 557-01-7
- 5570-18-3
- 5570-19-4
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View