-
Name
9-Bromoanthracene
- EINECS 216-359-3
- CAS No. 1564-64-3
- Article Data66
- CAS DataBase
- Density 1.479 g/cm3
- Solubility Insoluble in water.
- Melting Point 97-100 °C(lit.)
- Formula C14H9Br
- Boiling Point 389.7 °C at 760 mmHg
- Molecular Weight 257.129
- Flash Point 190.3 °C
- Transport Information
- Appearance white to light yellow crystal powder
- Safety 22-24/25
- Risk Codes
-
Molecular Structure
-
Hazard Symbols
 Xi
Xi
- Synonyms 10-Bromoanthracene;9-Anthracenyl bromide;9-Anthracyl bromide;Anthracene,9-bromo-;NSC 803;9-Bromo anthracene;
- PSA 0.00000
- LogP 4.75550
Synthetic route

| Conditions | Yield |
|---|---|
| With bis(trifluoroacetoxy)iodobencene; trimethylsilyl bromide In dichloromethane at 20℃; Inert atmosphere; | 100% |
| With N,N,N’,N’-tetrabromobenzene-1,3-disulfonamide In dichloromethane at 25℃; for 0.583333h; | 98% |
| With copper(ll) bromide In chloroform Reflux; | 94% |
-
-
641144-16-3
(10-bromo-9-anthracenyl)boronic acid

-
-
1564-64-3
9-Bromoanthracene

| Conditions | Yield |
|---|---|
| With [Pd(N-phenylbenzaldimine)(OAc)(PPh3)] In tetrahydrofuran at 20℃; for 24h; Schlenk technique; chemoselective reaction; | 99% |
-
-
24100-41-2
1-(anthracen-10-yl)-2-bromoethanone

-
A
-
784-04-3
9-acetylanthracene

-
B
-
1564-64-3
9-Bromoanthracene

| Conditions | Yield |
|---|---|
| With diethyl 2,6-dimethyl-1,4-dihydropyridine-3,5-dicarboxylate; acetic acid for 0.7h; Irradiation; Inert atmosphere; | A 94% B 2% |
-
-
22362-86-3
9-iodoanthracene

-
-
1564-64-3
9-Bromoanthracene

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 25℃; for 3h; Reagent/catalyst; Time; Temperature; | 92% |

| Conditions | Yield |
|---|---|
| With N-Bromosuccinimide; 2,4,6-trimethylaniline In dichloromethane at 0℃; for 8h; Inert atmosphere; regioselective reaction; | A 84% B 7% |
| With N-Bromosuccinimide; C5H13NO3S In n-heptane at 60℃; for 4.5h; Darkness; | A 81% B 8% |
| With N-Bromosuccinimide; iodine In tetrachloromethane Bromination; Heating; |
-
-
523-27-3
9,10-Dibromoanthracene

-
A
-
1564-64-3
9-Bromoanthracene

-
B
-
25882-75-1
9-<4-(N,N-dimethylamino)phenyl>-9,10-dihydroanthracene

-
C
-
101-61-1
4,4'-methylene-bis(N,N-dimethylaniline)

| Conditions | Yield |
|---|---|
| With N,N-dimethyl-aniline In acetonitrile Irradiation; Further byproducts given; | A 70% B 7.2% C 13.4% D 8.2% |

-
-
523-27-3
9,10-Dibromoanthracene

-
-
121-69-7
N,N-dimethyl-aniline

-
A
-
1564-64-3
9-Bromoanthracene

-
B
-
25882-75-1
9-<4-(N,N-dimethylamino)phenyl>-9,10-dihydroanthracene

-
C
-
3571-43-5
crystal violet bromide

-
D
-
101-61-1
4,4'-methylene-bis(N,N-dimethylaniline)

| Conditions | Yield |
|---|---|
| In acetonitrile Product distribution; Irradiation; photolysis; | A 70% B 7.2% C 1.2% D 13.4% E 8.2% |


| Conditions | Yield |
|---|---|
| With N-Bromosuccinimide In chloroform at 60℃; for 2h; Inert atmosphere; | 61.2% |
-
-
523-27-3
9,10-Dibromoanthracene

-
-
100-67-4
potassium phenolate

-
A
-
1564-64-3
9-Bromoanthracene

-
B
-
23674-00-2
bromo-9 phenoxy-10 anthracene

-
C
-
84-65-1
9,10-phenanthrenequinone

| Conditions | Yield |
|---|---|
| In N,N-dimethyl-formamide at 153℃; for 3h; | A 3% B 35% C 2% |


| Conditions | Yield |
|---|---|
| Kinetics; |
-
-
46491-50-3
9,19-Dibromo-9,10-dihydroanthracene

-
-
1564-64-3
9-Bromoanthracene

| Conditions | Yield |
|---|---|
| With toluene; copper(I) bromide |
-
-
120-12-7
anthracene

-
-
20244-61-5
2,4,4,6-Tetrabromo-2,5-cyclohexadien-1-one

-
-
1564-64-3
9-Bromoanthracene

| Conditions | Yield |
|---|---|
| Kinetics; |
-
-
46491-50-3
9,19-Dibromo-9,10-dihydroanthracene

-
-
67-66-3
chloroform

-
-
109-89-7
diethylamine

-
A
-
1564-64-3
9-Bromoanthracene

-
B
-
120-12-7
anthracene

-
-
75-77-4
chloro-trimethyl-silane

-
-
523-27-3
9,10-Dibromoanthracene

-
A
-
1564-64-3
9-Bromoanthracene

-
B
-
89811-60-9
9-Brom-10-(trimethylsilyl)anthracen

| Conditions | Yield |
|---|---|
| With n-butyllithium 1.) ether, pentane, 20 deg C, 45 min, 2.) ether, THF, 20 deg C, 20 h; Yield given. Multistep reaction. Yields of byproduct given. Title compound not separated from byproducts; |

-
-
120-12-7
anthracene

-
A
-
716-53-0
9-chloroanthracene

-
B
-
1564-64-3
9-Bromoanthracene

-
C
-
22362-86-3
9-iodoanthracene

| Conditions | Yield |
|---|---|
| With aluminium trichloride; cyanogen iodide; tetrabutylammomium bromide In diethyl ether; nitromethane at 25℃; for 20h; | A 6 % Chromat. B 88 % Chromat. C 1 % Chromat. |


-
-
46491-50-3
9,19-Dibromo-9,10-dihydroanthracene

-
-
7783-06-4
hydrogen sulfide

-
-
64-19-7
acetic acid

-
A
-
1564-64-3
9-Bromoanthracene

-
B
-
120-12-7
anthracene

| Conditions | Yield |
|---|---|
| at 50℃; |


| Conditions | Yield |
|---|---|
| With N-Bromosuccinimide; biphenyl In benzene at 20℃; for 2h; Irradiation; | A 88.9 % Chromat. B 11.1 % Chromat. |

| Conditions | Yield |
|---|---|
| With sodium hydrogencarbonate; N,N-dimethyl-formamide; palladium dichloride at 120℃; for 18h; |
-
-
77-48-5
1,3-dibromo-5,5-dimethylimidazolidine-2,4-dione

-
-
120-12-7
anthracene

-
A
-
1564-64-3
9-Bromoanthracene

-
B
-
523-27-3
9,10-Dibromoanthracene

| Conditions | Yield |
|---|---|
| Stage #1: 1,3-dibromo-5,5-dimethylimidazolidine-2,4-dione; anthracene In ethyl acetate for 2.5h; Refluxing; Stage #2: In tetrahydrofuran |


| Conditions | Yield |
|---|---|
| With dihydrogen peroxide; acetic acid; potassium bromide In water at 60℃; for 6h; regioselective reaction; | A 85 %Spectr. B 15 %Spectr. |
-
-
120-12-7
anthracene

-
A
-
1564-64-3
9-Bromoanthracene

-
B
-
523-27-3
9,10-Dibromoanthracene

-
C
-
84-65-1
9,10-phenanthrenequinone

| Conditions | Yield |
|---|---|
| With Oxone; quartz; sodium bromide In n-heptane for 0.333333h; Milling; chemoselective reaction; |

-
-
1564-64-3
9-Bromoanthracene

-
-
1066-54-2
trimethylsilylacetylene

-
-
104784-61-4
[2-(anthracen-9-yl)ethynyl]trimethylsilane

| Conditions | Yield |
|---|---|
| With bis-triphenylphosphine-palladium(II) chloride; copper(l) iodide; triethylamine In tetrahydrofuran for 15h; Heating; | 100% |
| With piperidine; bis-triphenylphosphine-palladium(II) chloride; copper(l) iodide In triethylamine at 110℃; Sonogashira Cross-Coupling; | 99% |
| Stage #1: 9-Bromoanthracene; trimethylsilylacetylene With triethylamine for 0.0833333h; Sonogashira coupling; Inert atmosphere; Stage #2: With copper(l) iodide at 50 - 65℃; Sonogashira coupling; Inert atmosphere; | 98% |
-
-
1564-64-3
9-Bromoanthracene

-
-
5720-06-9
2-Methoxyphenylboronic acid

-
-
1232-27-5
9-(2'-methoxyphenyl)anthracene

| Conditions | Yield |
|---|---|
| With potassium phosphate; tris-(dibenzylideneacetone)dipalladium(0); C31H27FeOP In toluene at 70℃; for 24h; Reagent/catalyst; Time; Suzuki-Miyaura Coupling; Schlenk technique; Inert atmosphere; Sealed tube; | 100% |
| With tris-(dibenzylideneacetone)dipalladium(0); potassium phosphate monohydrate; C30H25FeP In toluene at 100℃; for 24h; Suzuki-Miyaura Coupling; Sealed tube; | 95% |
| With tetrakis(triphenylphosphine) palladium(0); potassium carbonate In ethanol; toluene for 20h; Heating; | 90% |
-
-
685-83-6
bis(diethylamino)chlorophosphine

-
-
1564-64-3
9-Bromoanthracene

-
-
925428-12-2
9-anthryl-bis(diethylamino)phosphine

| Conditions | Yield |
|---|---|
| With n-butyllithium In diethyl ether; hexane at -78 - 20℃; for 16h; | 100% |
| Stage #1: 9-Bromoanthracene With n-butyllithium In diethyl ether; hexane at -78 - 0℃; for 1h; Inert atmosphere; Stage #2: bis(diethylamino)chlorophosphine In diethyl ether; hexane at -78 - 20℃; Inert atmosphere; |
-
-
1564-64-3
9-Bromoanthracene

-
-
4688-76-0
2-Biphenylboronic acid

-
-
849223-96-7
9‐([1,1'‐biphenyl]‐2‐yl)anthracene

| Conditions | Yield |
|---|---|
| With potassium phosphate monohydrate; R-Phos; bis(dibenzylideneacetone)-palladium(0) In 1,4-dioxane at 100℃; for 0.5h; Suzuki coupling; Inert atmosphere; | 100% |

| Conditions | Yield |
|---|---|
| With n-butyllithium In tetrahydrofuran; hexane at -78 - 20℃; for 1h; Inert atmosphere; | 99.4% |
| Stage #1: 9-Bromoanthracene With n-butyllithium In tetrahydrofuran at -78℃; for 1h; Stage #2: dimethylmonochlorosilane In tetrahydrofuran at -78 - 20℃; for 8h; | 59% |
| With n-butyllithium In diethyl ether; hexane at 25℃; for 72h; | 54% |
| With n-butyllithium In diethyl ether at -40 - 20℃; for 17.5h; Inert atmosphere; | 43% |

| Conditions | Yield |
|---|---|
| With 2-H-1,3-di-tert-butyl-1,3,2-diazaphosphorinane; 2,2'-azobis(isobutyronitrile) In toluene at 90℃; for 5h; | 99% |
| With 1,3-bis(2,6-diisopropylphenyl)-2,4-diphenylimidazole In N,N-dimethyl-formamide at 20℃; for 36h; Glovebox; | 98% |
| With N1,N1,N12,N12-tetramethyl-7,8-dihydro-6H-dipyrido[1,2-a:2,1'-c][1,4]diazepine-2,12-diamine In N,N-dimethyl-formamide at 20℃; Inert atmosphere; | 96% |

| Conditions | Yield |
|---|---|
| With potassium phosphate; d(4)-methanol; palladium diacetate; catacxium A In toluene at 80℃; for 16h; | 99% |
| With dimethylsulfoxide-d6; 9-N-methylamino-1-oxophenalene; potassium tert-butylate; potassium at 25 - 30℃; for 24h; Inert atmosphere; Glovebox; | 74% |
| With n-butyllithium; water-d2 In diethyl ether at 0℃; | 57% |

| Conditions | Yield |
|---|---|
| With potassium phosphate; rac-2,8-bis(diphenylphosphanyl)-4,10-dimethyl-6,12-dihydro-5,11-methanodibenzo[b,f][1,5]diazocine; palladium diacetate In toluene at 90℃; Suzuki Coupling; Schlenk technique; Inert atmosphere; | 99% |
| With potassium carbonate In (2)H8-toluene at 100℃; for 24h; Reagent/catalyst; Suzuki-Miyaura Coupling; | 99% |
| With {2,6-bis[(di-1-piperidinylphosphino)amino]phenyl}palladium(II) chloride; potassium carbonate In 1,4-dioxane; water; butan-1-ol at 100℃; for 0.5h; Suzuki-Miyaura reaction; | 98% |

| Conditions | Yield |
|---|---|
| With palladium diacetate; potassium carbonate; triphenylphosphine In 1,2-dimethoxyethane; water at 80℃; for 17h; Inert atmosphere; | 99% |
| With tetrakis(triphenylphosphine) palladium(0); potassium carbonate In ethanol; toluene for 24h; Inert atmosphere; Reflux; Sealed tube; | 90% |
| With tetrakis(triphenylphosphine) palladium(0); potassium carbonate In ethanol; water; toluene at 90℃; for 16h; Inert atmosphere; | 90% |
-
-
1564-64-3
9-Bromoanthracene

-
-
1158054-27-3
2-bromo-6-[2-(diphenylphosphinoyl)ethyl]pyridine

-
-
1158054-88-6
2-(9-anthracyl)-6-[2-(diphenylphosphinoyl)ethyl]pyridine

| Conditions | Yield |
|---|---|
| Stage #1: 9-Bromoanthracene With n-butyllithium In tetrahydrofuran; hexane at -78℃; for 0.5h; Stage #2: With zinc(II) chloride In tetrahydrofuran; hexane at 20℃; for 1h; Stage #3: 2-bromo-6-[2-(diphenylphosphinoyl)ethyl]pyridine With tetrakis(triphenylphosphine) palladium(0) In tetrahydrofuran; hexane at 60℃; for 24h; Negishi-Takahashi coupling; | 99% |
-
-
1564-64-3
9-Bromoanthracene

-
-
16419-60-6
2-Methylphenylboronic acid

-
-
23674-13-7
9-(2-methylphenyl)anthracene

| Conditions | Yield |
|---|---|
| With tris-(dibenzylideneacetone)dipalladium(0); potassium phosphate monohydrate; C30H25FeP In toluene at 100℃; for 24h; Suzuki-Miyaura Coupling; Sealed tube; | 99% |
| With potassium phosphate; C29H37O2P; palladium diacetate In toluene at 110℃; for 3h; Suzuki-Miyaura Coupling; Inert atmosphere; Schlenk technique; | 94% |
| With potassium phosphate; tris-(dibenzylideneacetone)dipalladium(0); 1′-triethylsilyl-1-(2-(E)-phenylvinyl)-2-(diphenylphosphino)ferrocene In toluene at 70℃; for 24h; Suzuki-Miyaura Coupling; Schlenk technique; Inert atmosphere; Sealed tube; | 89% |

| Conditions | Yield |
|---|---|
| With palladium; triphenylphosphine In tetrahydrofuran at 70℃; for 24h; Catalytic behavior; Negishi Coupling; Inert atmosphere; Green chemistry; | 99% |
-
-
1564-64-3
9-Bromoanthracene

-
-
195062-57-8
p-tolylboronic pinacol ester

-
-
23674-14-8
9-(4-methylphenyl)anthracene

| Conditions | Yield |
|---|---|
| With bis-triphenylphosphine-palladium(II) chloride; potassium hydroxide In neat (no solvent) at 110℃; for 24h; Suzuki-Miyaura Coupling; Schlenk technique; Inert atmosphere; | 99% |
-
-
1564-64-3
9-Bromoanthracene

-
-
31614-66-1
tributyl(p-tolyl)stannane

-
-
23674-14-8
9-(4-methylphenyl)anthracene

| Conditions | Yield |
|---|---|
| With palladium diacetate; cesium fluoride; tricyclohexylphosphine In neat (no solvent) at 110℃; under 760.051 Torr; for 24h; Stille Cross Coupling; Sealed tube; | 99% |

| Conditions | Yield |
|---|---|
| With manganese(II) chloride dihydrate; lithium In tetrahydrofuran at 25℃; for 0.5h; Birch reaction; Inert atmosphere; | 98% |
| With potassium hydroxide semihydrate; phosphorus; water In dimethyl sulfoxide at 100℃; for 3h; Inert atmosphere; | 90% |
| With Dicalcium Nitride Electride In tetrahydrofuran; isopropyl alcohol at 65℃; for 24h; Birch Reduction; Inert atmosphere; | 84% |
| With tetraethylammonium perchlorate; triethylamine In dimethyl sulfoxide at 20℃; for 4h; Electrolysis; Green chemistry; | 65% |
-
-
1564-64-3
9-Bromoanthracene

-
-
5720-07-0
4-methoxyphenylboronic acid

-
-
23674-15-9
9-(p-methoxyphenyl)anthracene

| Conditions | Yield |
|---|---|
| With tetrakis(triphenylphosphine) palladium(0); potassium carbonate In ethanol; water; toluene for 24h; Suzuki Coupling; Reflux; Inert atmosphere; | 98% |
| With tetrakis(triphenylphosphine) palladium(0); potassium carbonate In ethanol; water; toluene at 105℃; for 18h; Temperature; Inert atmosphere; | 90% |
| With tetrakis(triphenylphosphine) palladium(0); potassium carbonate In ethanol; toluene at 110℃; for 20h; Temperature; Inert atmosphere; | 90% |
-
-
1564-64-3
9-Bromoanthracene

-
-
13922-41-3
1-Naphthylboronic acid

-
-
7424-70-6
10-(naphthalen-1-yl)anthracene

| Conditions | Yield |
|---|---|
| With potassium phosphate; tris-(dibenzylideneacetone)dipalladium(0); C31H27FeOP In toluene at 70℃; for 24h; Reagent/catalyst; Time; Suzuki-Miyaura Coupling; Schlenk technique; Inert atmosphere; Sealed tube; | 98% |
| With C70H40Cl2F68N2O6Pd2; tetrabutylammomium bromide; potassium carbonate In water at 140℃; for 0.2h; Suzuki-Miyaura reaction; Microwave irradiation; | 97% |
| With tetrabutylammomium bromide; potassium carbonate In ethanol; water; toluene at 70 - 75℃; for 2h; Inert atmosphere; | 97.46% |
-
-
1564-64-3
9-Bromoanthracene

-
-
86853-97-6
7H-Dibenzofluorenone

| Conditions | Yield |
|---|---|
| Stage #1: 9-Bromoanthracene With n-butyllithium In tetrahydrofuran; hexane at -78℃; for 1h; Inert atmosphere; Stage #2: 7H-Dibenzofluorenone In tetrahydrofuran; hexane at -78 - 20℃; Inert atmosphere; | 98% |

| Conditions | Yield |
|---|---|
| With palladium diacetate; caesium carbonate; XPhos In tetrahydrofuran-d8 for 20h; Sonogashira Cross-Coupling; Glovebox; Schlenk technique; Inert atmosphere; Heating; | 98% |
-
-
1564-64-3
9-Bromoanthracene

-
-
32316-92-0
naphthalene-2-boronic acid

-
-
7424-72-8
9-(naphthyl-2-yl)anthracene

| Conditions | Yield |
|---|---|
| With tetrakis(triphenylphosphine) palladium(0); sodium hydroxide In tetrahydrofuran at 50 - 90℃; for 13h; Inert atmosphere; | 97.4% |
| With potassium carbonate; triphenylphosphine; palladium diacetate In 1,2-dimethoxyethane; water for 8h; Suzuki cross-coupling; Heating; | 92% |
| With potassium carbonate; palladium diacetate; tris-(o-tolyl)phosphine In 1,2-dimethoxyethane; water at 80℃; for 3h; Suzuki-Miyaura coupling; | 92% |

| Conditions | Yield |
|---|---|
| With C70H40Cl2F68N2O6Pd2; tetrabutylammomium bromide; potassium carbonate In water at 140℃; for 0.2h; Suzuki-Miyaura reaction; Microwave irradiation; | 97% |

| Conditions | Yield |
|---|---|
| With tris-(dibenzylideneacetone)dipalladium(0); monophosphine 1,2,3,4,5-pentaphenyl-1'-(di-tert-butylphosphino)ferrocene In toluene at 20℃; for 1h; | 97% |
| With tris-(dibenzylideneacetone)dipalladium(0); DavePhos In toluene at 20℃; for 1h; Reagent/catalyst; |
-
-
1564-64-3
9-Bromoanthracene


| Conditions | Yield |
|---|---|
| With palladium diacetate; potassium carbonate; XPhos In water; toluene at 80℃; for 18h; Inert atmosphere; | 97% |

| Conditions | Yield |
|---|---|
| With C30H43O2P*C13H12N(1-)*CH3O3S(1-)*Pd(2+); lithium hexamethyldisilazane In 1,4-dioxane at 100℃; for 16h; Inert atmosphere; | 97% |
-
-
1564-64-3
9-Bromoanthracene

| Conditions | Yield |
|---|---|
| Stage #1: N-tert-butoxycarbonyl-N,N-bis[4-(di-tert-butylphenylamino)phenyl]amine With trifluoroacetic acid at 20℃; for 0.166667h; Stage #2: 9-Bromoanthracene With tris-(dibenzylideneacetone)dipalladium(0); tri-tert-butyl phosphine; sodium t-butanolate In toluene Inert atmosphere; Reflux; | 97% |

| Conditions | Yield |
|---|---|
| Stage #1: 9-Bromoanthracene With bis(tri-t-butylphosphine)palladium(0); oxygen In toluene at 20℃; for 0.0166667h; Schlenk technique; Stage #2: methyllithium In toluene at 20℃; for 0.0333333h; Schlenk technique; | 97% |
-
-
1564-64-3
9-Bromoanthracene

-
-
1383803-71-1
2-(4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)phenyl)quinoline

| Conditions | Yield |
|---|---|
| Stage #1: 9-Bromoanthracene; 2-(4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)phenyl)quinoline With potassium carbonate In ethanol; toluene for 0.333333h; Inert atmosphere; Stage #2: With tetrakis(triphenylphosphine) palladium(0) In ethanol; toluene Reflux; | 97% |

| Conditions | Yield |
|---|---|
| With bis[chloro(1,2,3-trihapto-allylbenzene)palladium(II)]; C30H42N2; sodium t-butanolate In tetrahydrofuran at 80℃; for 1h; Kinetics; Buchwald-Hartwig Coupling; Glovebox; Inert atmosphere; Molecular sieve; Sealed tube; | 97% |
9-Bromoanthracene Chemical Properties
Molecule structure of 9-Bromoanthracene (CAS NO.1564-64-3):
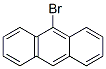
IUPAC Name: 9-Bromoanthracene
Molecular Weight: 257.12526 g/mol
Molecular Formula: C14H9Br
Density: 1.479 g/cm3
Melting Point: 97-100 °C(lit.)
Boiling Point: 389.7 °C at 760 mmHg
Flash Point: 190.3 °C
Index of Refraction: 1.733
Molar Refractivity: 69.62 cm3
Molar Volume: 173.8 cm3
Surface Tension: 51.3 dyne/cm
Enthalpy of Vaporization: 61.41 kJ/mol
Vapour Pressure: 6.28E-06 mmHg at 25 °C
Storage Temp.: 0-6 °C
XLogP3: 5.2
Exact Mass: 255.988763
MonoIsotopic Mass: 255.988763
Canonical SMILES: C1=CC=C2C(=C1)C=C3C=CC=CC3=C2Br
InChI: InChI=1S/C14H9Br/c15-14-12-7-3-1-5-10(12)9-11-6-2-4-8-13(11)14/h1-9H
InChIKey: ZIRVQSRSPDUEOJ-UHFFFAOYSA-N
EINECS: 216-359-3
Product Categories: Aromatic Compounds; blocks; Bromides; Aromatic Halides (substituted); Anthracenes; Anthracene series; Aryl; C13 to C37+; Halogenated Hydrocarbons
9-Bromoanthracene Safety Profile
Hazard Codes:  Xi
Xi
Safety Statements: 22-24/25
S22:Do not breathe dust.
S24/25:Avoid contact with skin and eyes.
WGK Germany: 3
Hazard Note: Irritant
May cause eye irritation. if absorbed through the skin,it will cause skin irritation.
Ingestion:if swallowed,it May cause the irritation of digestive tract.if inhaled.it May be harmful for your respiratory.
9-Bromoanthracene Specification
9-Bromoanthracene (CAS NO.1564-64-3) is also named as NSC 803 ; Anthracene, 9-bromo- ; 10-Bromoanthracene ; 9-Anthracenyl bromide ; 9-Bromo anthracene . 9-Bromoanthracene (CAS NO.1564-64-3) is white to light yellow crystal powder.
Related Products
- 9-Bromoanthracene
- 15646-46-5
- 156472-94-5
- 156478-71-6
- 15648-16-5
- 156481-74-2
- 156489-68-8
- 156491-84-8
- 15649-31-7
- 156496-70-7
- 156499-64-8
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View