-
Name
Boron trichloride
- EINECS 233-658-4
- CAS No. 10294-34-5
- Article Data333
- CAS DataBase
- Density 1.384 g/cm3
- Solubility Decomposes in water
- Melting Point -107 °C(lit.)
- Formula BCl3
- Boiling Point 12.5 °C at 760 mmHg
- Molecular Weight 117.17
- Flash Point -17 °C
- Transport Information UN 3390 6.1/PG 1
- Appearance colourless gas
- Safety 9-26-28-36/37/39-45-8-61-38-28A-16-1-60-33-23-7/9-62
- Risk Codes 14-26/28-36/37/38-40-67-65-62-51/53-48/20-34-11-50/53-26/27/28-63-39/23/24/25-24-21-10
-
Molecular Structure
-
Hazard Symbols
 T+,
T+,  T,
T,  N,
N,  F
F
- Synonyms Boronchloride (BCl3) (8CI);Trichloroborane;Trichloroboron;
- PSA 0.00000
- LogP 1.68770
Synthetic route

-
-
10294-34-5
boron trichloride

| Conditions | Yield |
|---|---|
| In melt Electrolysis; electrolysis in a chloride melt (400-500°C); | 100% |
| With chlorine 400°C;; 6-8 times vac. distn. spect. purity product obtained; | |
| With silver(I) chloride |

| Conditions | Yield |
|---|---|
| byproducts: HCl, ClCOCl; at room temp.;; | A n/a B 96% |


-
A
-
10294-34-5
boron trichloride

-
B
-
64486-90-4
1,3-di-tert-butyl-2-chloro-2,3-dihydro-1H-1,3,2-diazaborole

| Conditions | Yield |
|---|---|
| With sodium amalgam In hexane Ar atmosphere, stiring (overnight in the dark); decantation, filtration, removement of volatiles (vacuum), sublimation (40 to 60°C, 0.01 Torr); | A n/a B 91% |

| Conditions | Yield |
|---|---|
| With hydrogen condensation of HBCl2/BCl3/B2H6 mixt. on molecular sieve, heating in H2 stream, BCl3 not adsorbed, B2H6 easily desorbed, strongly adsorbed HBCl2 slowly symmetrysied; | A n/a B 90% |
| With H2 condensation of HBCl2/BCl3/B2H6 mixt. on molecular sieve, heating in H2 stream, BCl3 not adsorbed, B2H6 easily desorbed, strongly adsorbed HBCl2 slowly symmetrysied; | A n/a B 90% |
| equil. const. Kx determined at 273 K; |
-
-
13701-67-2
B2Cl4

-
A
-
32915-80-3
octaboron octachloride

-
B
-
31304-34-4
B9Cl9

-
C
-
10294-34-5
boron trichloride

| Conditions | Yield |
|---|---|
| In tetrachloromethane 100°C, 2 weeks; NMR monitoring; not isolated, detected by NMR; | A 88% B n/a C n/a |

-
-
7446-70-0
aluminium trichloride

-
-
7637-07-2
boron trifluoride

-
A
-
7784-18-1
aluminum(III) fluoride

-
B
-
10294-34-5
boron trichloride

| Conditions | Yield |
|---|---|
| heating with excess of BF3; condensation of BCl3 in an U-tube cooled the mixt. of CO2 and alcohol; fractionated distn.; | A n/a B 80% |
| heating with excess of BF3; condensation of BCl3 in an U-tube cooled the mixt. of CO2 and alcohol; fractionated distn.; | A n/a B 80% |

-
-
89033-11-4
1,3-bis(dichloroboryl)propane

-
-
13517-10-7
boron triiodide

-
A
-
157426-91-0
1,3-bis(diiodoboryl)propane

-
B
-
10294-34-5
boron trichloride

| Conditions | Yield |
|---|---|
| removal of BCl3 in vacuo;; distillation in vacuo after 4 h;; | A 78% B n/a |


-
-
13701-67-2
diboron tetrachloride

-
A
-
17927-57-0
monochlorodiborane

-
C
-
10325-39-0
dichloroborane

-
D
-
10294-34-5
boron trichloride

-
E
-
19287-45-7
diborane

| Conditions | Yield |
|---|---|
| In neat (no solvent) reaction carried out in Pyrex tubes to which Teflon valves had been sealed or in a N2-filled glovebox; reaction of B2Cl4 with excess B5H9 in a 1:3 mole ratio; vessel allowed to warm from -196 ° C to ambient temp., 20 min at ambient temp.,; fractionation, yield of 1-BCl2B5H8 based upon amt. of B2Cl4 employed: 73%, based upon amt. of B5H9 consumed: 95%; small amts. of B2H6, BCl2H, B2H5Cl, also material insufficiently volatileto move into vac. line formed;; | A n/a B 73% C n/a D n/a E n/a |

-
-
3677-81-4
diphenylboronchloride

-
-
13517-10-7
boron triiodide

-
A
-
17933-05-0
(diphenyl)iodoborane

-
B
-
10294-34-5
boron trichloride

| Conditions | Yield |
|---|---|
| heating at 130-140°C for 4 h; distn.; | A 72% B n/a |


| Conditions | Yield |
|---|---|
| 800-1000°C; ratio B2O3:NaCl=3:1; | 60% |
| 800-1000°C; ratio B2O3:NaCl=1:1; | 30% |
| 0% |
-
-
830-48-8
dichloro(pentafluorophenyl)borane

-
A
-
2720-03-8
bis(pentafluorophenyl)boron chloride

-
B
-
10294-34-5
boron trichloride

| Conditions | Yield |
|---|---|
| In neat (no solvent) pyrolysis at 220°C within 25 hours;; | A 59% B n/a |
| heating; | |
| heating; |

-
A
-
212375-81-0
1,3-bis(2,6-dimethylphenyl)-2-chloro-2,3-dihydro-1H-1,3,2-diazaborole

-
B
-
10294-34-5
boron trichloride

| Conditions | Yield |
|---|---|
| With sodium amalgam In hexane Ar atmosphere, stiring (room temp., 5 d); decantation, removement volatiles (vacuum); | A 55% B n/a |
-
-
74686-73-0
Cl2BNCl(C2H5)

-
-
13701-67-2
diboron tetrachloride

-
A
-
74686-75-2
N-ethylbis(dichloroboryl)amine

-
B
-
10294-34-5
boron trichloride

| Conditions | Yield |
|---|---|
| under N2, with exclusion of moisture, reagents in 8/4.4 mmol ratio, condensed at -196°C, gently warmed to room temp. for 15 min; BCl3 sepd. by distn.; elem. anal.; | A 37.5% B n/a |

-
-
20693-69-0
2,4-dicarba-closo-heptaborane

-
-
23704-81-6
1,6-dicarba-closo-decaborane(10)

-
A
-
28347-92-4
5-Cl-closo-2,4-C2B5H6

-
C
-
10294-34-5
boron trichloride

| Conditions | Yield |
|---|---|
| With aluminium trichloride; chlorine In neat (no solvent) byproducts: CH3BCl2, CH2(BCl2)2; addn. of mixt. of B-compds. to NMR tube contg. freshly sublimed AlCl3, addn. of Cl2 gas, warmed from -190°C to room temp. for 3 h; not isolated; detected by NMR; | A 36% B 32% C 4% |

-
-
28347-92-4
5-Cl-closo-2,4-C2B5H6

-
C
-
79550-11-1
5-CH3-6-Cl-closo-2,4-C2B5H5

-
D
-
40710-68-7
1,1-bis(dichloroboryl)methane

-
E
-
10294-34-5
boron trichloride

| Conditions | Yield |
|---|---|
| With chloroform In chloroform heating of 5-Cl-2,4-C2B5H6 and CH3Cl in a react. vessel at 126°Cfor 21.0 h; further products; cold-column vac. fractionation at -104 to -29°C; identification by (11)B-NMR; | A n/a B n/a C 34% D n/a E n/a |

-
-
40710-69-8
tris(dichloroboryl)methane

-
A
-
10294-34-5
boron trichloride

-
B
-
54573-10-3
hexachloro hexaboraadamantane

| Conditions | Yield |
|---|---|
| 450°C, sealed tube; cooling to room temp. over 12 h yields crystals, evapn. of volatile products, boraadamantane is sublimated in vac., elem. anal.; | A n/a B 28% |

-
-
13701-67-2
diboron tetrachloride

-
B
-
10294-34-5
boron trichloride

| Conditions | Yield |
|---|---|
| In neat (no solvent) reaction carried out in pyrex tubes to which Teflon valves had been sealed or N2 filled glovebox; reaction of B2Cl4 with excess B5H9 in a 1.0:1.4 mole ratio; vessel allowed to warm from -196 ° C to ambient temp., 25 min at ambient temp.,; fractionation,; | A 26% B n/a |
| In neat (no solvent) reaction carried out in a Pyrex tubes to which Teflon valves had been sealed, or in a N2-filled glovebox; reaction of B2Cl4 with excess B5H9 in a 2.9:1.0 mole ratio; vessel allowed to warm from -196 ° C to ambient temp., 12 min at ambient temp.,; fractionation, nonvolatile material remained within the vessel, yield of BCl2B5H8 was 8% based upon amt. of B2Cl4, 34% based upon unrecovered b5H9; | A 8% B n/a |

-
-
40710-68-7
1,1-bis(dichloroboryl)methane

-
A
-
10294-34-5
boron trichloride

-
B
-
54573-10-3
hexachloro hexaboraadamantane

| Conditions | Yield |
|---|---|
| byproducts: HCl; 450°C, sealed tube; cooling to room temp. over 12 h yields crystals, evapn. of volatile products, boraadamantane is sublimated in vac,, elem. anal.; | A n/a B 24% |

| Conditions | Yield |
|---|---|
| byproducts: N2; 200°C, in bomb tube; | A n/a B 24% |
| byproducts: N2; 200°C, in bomb tube; | A n/a B 24% |
-
-
30624-33-0
1-I-B5H8

-
A
-
19469-13-7
1-chloro-pentaborane(9)

-
B
-
10294-34-5
boron trichloride

-
C
-
19287-45-7
diborane

| Conditions | Yield |
|---|---|
| With mercury dichloride byproducts: H2, HCl; heating in closed react. tube at 105°C, 70 min;; | A 7% B n/a C n/a |
| With mercury dichloride byproducts: H2, HCl; in bomb tube at temp. >= 100°C;; |


| Conditions | Yield |
|---|---|
| With tetrachloromethane Vac.-distn. of B2Br4 to CCl4, heating in an oil-bath (96°C, 5.75 h).; Monitored by (11)B-NMR.; | A 5.5% B 4.5% C 1.3% |

-
-
7318-78-7
dichloro-methyl-borane

-
A
-
10294-34-5
boron trichloride

-
B
-
54573-10-3
hexachloro hexaboraadamantane

| Conditions | Yield |
|---|---|
| byproducts: HCl; 450°C, sealed tube; cooling to room temp. over 12 h yields crystals, evapn. of volatile products, boraadamantane is sublimated in vac., elem. anal.; | A n/a B 5% |
-
-
13701-67-2
diboron tetrachloride

-
-
7719-12-2, 52843-90-0
phosphorus trichloride

-
A
-
244036-47-3
closo-1,7-P2B10Cl10

-
B
-
112897-10-6
closo-3,4,5,6-tetrachloro-1,2-diphosphahexaborane(4)

-
C
-
263152-14-3
closo-1,10-P2B8Cl8

-
D
-
263152-13-2
conjuncto-3,3'-(1,2-P2B4Cl3)2

-
E
-
10294-34-5
boron trichloride

| Conditions | Yield |
|---|---|
| In neat (no solvent) (vac. or inert gas); B2Cl4 and PCl3 condensed in vac., sealed, heated at 400°C for 1 h, allowed to cool over 20 h; vac.-evapd. at 0°C, extd. with BCl3, evapd. at 0°C, fractionated by sublimation in vac.; | A 5% B n/a C n/a D n/a E n/a |

-
-
535-15-9
ethyl 1,1-dichloroacetate


-
A
-
79-36-7
dichloroacethyl chloride

-
B
-
75-00-3
chloroethane

-
C
-
10294-34-5
boron trichloride

| Conditions | Yield |
|---|---|
| at 45℃; |

-
-
16339-30-3
butoxy-dichloro-borane

-
A
-
18379-65-2
dibutoxy-chloro-borane

-
B
-
10294-34-5
boron trichloride

| Conditions | Yield |
|---|---|
| at 20℃; under 14 Torr; Disproportionierung; |
-
-
34456-12-7
dichloro(neopentyloxy)borane

-
A
-
115001-74-6
bis(neopentyloxy)chloroborane

-
B
-
10294-34-5
boron trichloride

| Conditions | Yield |
|---|---|
| at 20℃; Zersetzung; |
-
-
16339-31-4
(2-chloroethoxy)dichloroborane

-
A
-
22238-19-3
tris(2-chloroethoxy)borane

-
B
-
10294-34-5
boron trichloride

| Conditions | Yield |
|---|---|
| at 70℃; under 14 Torr; |
-
-
93314-06-8
bis(n-octyloxy)chloroborane

-
A
-
2467-12-1
tri-n-octyl borate

-
B
-
10294-34-5
boron trichloride

| Conditions | Yield |
|---|---|
| under 0.01 Torr; |
-
-
98561-14-9
(n-octyloxy)dichloroborane

-
A
-
111-85-3
1-Chlorooctane

-
B
-
2467-12-1
tri-n-octyl borate

-
C
-
10294-34-5
boron trichloride

| Conditions | Yield |
|---|---|
| Erwaermen unter vermindertem Druck; |


| Conditions | Yield |
|---|---|
| Disproportionierung bei Normaldruck; |


| Conditions | Yield |
|---|---|
| in presence of fluid BCl3; 100 % conversion at 20°C in 1 week; more than 60 % B2H5Cl reacts at 30°C in 4 h; | 100% |
| 100°C; 20 min.; | >99 |
| 100°C; 20 min; BCl3 in excess; | >99 |

| Conditions | Yield |
|---|---|
| ambient temp.; | 100% |
| ambient temp.; | 100% |
-
-
1493-13-6
trifluorormethanesulfonic acid

-
-
10294-34-5
boron trichloride

-
-
64371-01-3
tris(trifluoromethanesulfonyloxy)boron

| Conditions | Yield |
|---|---|
| at 10 - 20℃; for 4h; | 100% |
| In 1,1,2-Trichloro-1,2,2-trifluoroethane soln. of the components stirred at -78 ° C for 4-6 h; SO2ClF may also be used as solvent instead of Freon-113; soln. warmed to room temp.; white solid obtained after removal of the solvent; prduct purified by distn. (68-73 ° C, 0.5 Torr); elem. anal.;; |

| Conditions | Yield |
|---|---|
| 100% | |
| 100% |
-
-
1923-70-2
tetrabutylammonium perchlorate

-
-
10294-34-5
boron trichloride

-
-
54775-46-1
tetrabutylammonium trichloroperchloratoborate

| Conditions | Yield |
|---|---|
| In chloroform introducing of gaseous BCl3 to soln. of <(C4H9)4N> in CHCl3 at 20°C; distn. and drying in vac.; | 100% |
| In neat (no solvent) Bu4NClO4 reacted slowly with excess of BCl3 at 20°C; elem. anal.; | |
| In chloroform Bu4NClO4 reacted slowly with BCl3 in CHCl3 at 20°C; elem. anal.; | |
| In chloroform |

| Conditions | Yield |
|---|---|
| In hexane; dichloromethane (N2); a soln. of BCl3 in hexane added to a soln. of Fe-complex at -78°C; stirred for 1 h at room temp.; evapd.; added CH2Cl2; filtered; evapd.; recrystd. from CH2Cl2/hexane; | 100% |

-
-
10294-34-5
boron trichloride

| Conditions | Yield |
|---|---|
| In hexane; toluene byproducts: B2H6; (N2); using Schlenk techniques; addn. dropwise of soln. BCl3 (2 equiv.) in hexane to soln. of (AlH3)6(AlNMe3)2(CCH2CH2SiMe3)6 (1 equiv.) in toluene at -78°C, stirring for 0.5 h at this temp., stirring for 0.5 h at 0°C and 2 h at room temp.; removal of volatiles in vac. at 30°C, recrystn. from toluene (50°C/4°C), drying in vac., elem. anal.; | 100% |
-
-
158789-52-7
nitrido(tetra-tert-butylphthalocyaninato)rhenium(V)

-
-
10294-34-5
boron trichloride

-
-
582295-87-2
((CH3)3CC8H3NN)4ReNBCl3

| Conditions | Yield |
|---|---|
| In dichloromethane Re compd. dissolved in CH2Cl2 and cooled to -5°C, electrophile added dropwise to this soln., stirred for 6 h at -5°C; solvent removed at -5°C, elem. anal.; | 100% |

| Conditions | Yield |
|---|---|
| In neat (no solvent) (Ar); Re2O7 put into glass ampoule; BCl3 condensed onto it at -196°C; warmed slowly to -40°C then to room temp.; slowly cooled to -60°C; | 100% |
-
-
955084-10-3
5,5-difluoro-10-(p-tolyl)-5H-dipyrrolo[1,2-c:2',1'-f][1,3,2]diazaborinin-4-ium-5-uide

-
-
10294-34-5
boron trichloride

| Conditions | Yield |
|---|---|
| In dichloromethane for 0.25h; Inert atmosphere; UV-irradiation; | 100% |

| Conditions | Yield |
|---|---|
| In n-heptane; dichloromethane at -78 - 25℃; for 20h; Inert atmosphere; | 100% |
-
-
10294-34-5
boron trichloride

-
-
81631-74-5
4-n-butyl-1-trimethylsilylbenzene

| Conditions | Yield |
|---|---|
| In dichloromethane at 20℃; Inert atmosphere; | 100% |

| Conditions | Yield |
|---|---|
| at 6 - 20℃; for 4h; | 100% |
-
-
10294-34-5
boron trichloride

| Conditions | Yield |
|---|---|
| In dichloromethane at 20℃; for 0.333333h; Glovebox; Inert atmosphere; Schlenk technique; | 100% |
-
-
1073151-42-4
N,N-difluoroboryl-[N-(3-phenyl-2H-isoindol-1-yl)-N-(3-phenyl-1H-isoindol-1-ylidene)amine]

-
-
10294-34-5
boron trichloride

| Conditions | Yield |
|---|---|
| In dichloromethane at 20℃; for 0.333333h; Glovebox; Inert atmosphere; Schlenk technique; | 100% |

| Conditions | Yield |
|---|---|
| In n-heptane; dichloromethane at 21℃; for 1h; Inert atmosphere; | 100% |

| Conditions | Yield |
|---|---|
| In diethyl ether byproducts: LiCl, AlCl3; distn. of BCl3 to water free etheric soln. of LiAlH4, cooled with liq. N2; heating up to room temp.; fractionated condensation; | 99.4% |
| In diethyl ether byproducts: LiCl, AlCl3; distn. of BCl3 to water free etheric soln. of LiAlH4, cooled with liq. N2; heating up to room temp.; fractionated condensation; | 99.4% |

| Conditions | Yield |
|---|---|
| In neat (no solvent) (N2); BCl3 was condensed above Hg compd. in an evacuated tube, mixt. was warmed up to room temp. with stirring, after 35 min was cooled to 0°C and all volatiles removed in vac.; fractionation at -160°C yielded BF3; identified by (19)F- and (11)B-NMR and mass spect.; | 99% |

| Conditions | Yield |
|---|---|
| With ethene; tetraethyldiborane(6) react. of two equiv of B(C2H5)3 with one equiv of BCl3 at room temp., decompn. of catalyst with ethylene; distn.; | 99% |
| With ethylene; tetraethyldiborane(6) react. of two equiv of B(C2H5)3 with one equiv of BCl3 at room temp., decompn. of catalyst with ethylene; distn.; | 99% |
| tetraethyldiborane(6) |
-
-
12081-54-8
tetraethyldiborane(6)

-
-
1116-61-6
tripropylborane

-
-
10294-34-5
boron trichloride

-
-
22086-53-9
dipropylchloroborane

| Conditions | Yield |
|---|---|
| With decene-1 introduction of BCl3 in mixt. of B(n-C3H7) and (C2H5)2B2H2 for 15 min, addn. of decene-1; distn.; | 99% |

-
-
12116-45-9
(benzyl alcohol)tricarbonylchromium(0)

-
-
10294-34-5
boron trichloride

-
-
12170-17-1
(η6-benzyl chloride)tricarbonylchromium(0)

| Conditions | Yield |
|---|---|
| In dichloromethane stirring at -78°C for 1 h, quenching (satd. aq. NaHCO3), warming to room temp.; addn. of H2O, extn. (Et2O), drying (MgSO4), filtration, concn.; | 99% |

| Conditions | Yield |
|---|---|
| In toluene under N2 atm. to soln. BCl3 in toluene was added C5H4(SiMe3)(SnMe3), react. mixt. was heated to reflux for 5 h; volatiles were removed at 0.1 mm Hg, 50°C; | 99% |

| Conditions | Yield |
|---|---|
| In hexane; toluene (under Ar, Schlenk); soln. of BCl3 in hexane added to soln. of ligand intoluene at ambient temp., stirred overnight; solvent removed under reduced pressure; | 99% |

-
-
10294-34-5
boron trichloride

| Conditions | Yield |
|---|---|
| In hexane; dichloromethane BCl3 (4 equiv.) and ligand at 25°C for 12 h; | 99% |
-
-
1189139-25-0
5,5-dimethyl-5H-dibenzo[b,f]stannepine

-
-
1237745-13-9
(2,4,6-triisopropylphenyl)tin(Bu)3

-
-
10294-34-5
boron trichloride

-
-
1237745-15-1
5-(2,4,6-tri-iso-propylphenyl)-5H-dibenzo[b,f]borepin

| Conditions | Yield |
|---|---|
| In toluene Schlenk air-free technique; react. of Sn(CH3)2(C2H2(C6H4)2), tripyl-Sn(Bu)3 and BCl3 at -78°C; | 99% |

-
-
10294-34-5
boron trichloride

-
-
1417726-65-8
4,4-dichloro-4-bora-3a,4a-diaza-s-indacene

| Conditions | Yield |
|---|---|
| In dichloromethane at 22℃; for 1h; Inert atmosphere; | 99% |

-
-
10294-34-5
boron trichloride

-
-
1355045-95-2
4,4-dichloro-2,6-diethyl-1,3,5,7-tetramethyl-8H-4-bora-3a,4a-diaza-s-indacene

| Conditions | Yield |
|---|---|
| In dichloromethane at 22℃; for 1h; | 99% |
| In hexane; dichloromethane for 1h; Inert atmosphere; |
-
-
183619-19-4
1',2',3',4',5'-pentamethylazaferrocene

-
-
10294-34-5
boron trichloride

-
-
1610052-27-1
C14H19BCl3FeN

| Conditions | Yield |
|---|---|
| In dichloromethane for 0.5h; Inert atmosphere; | 99% |
-
-
1237706-56-7
1,3-bis-(2,4,6-trimethylphenyl)-4,6-diketo-5,5-dimethylpyrimidin-2-ylidene

-
-
10294-34-5
boron trichloride

| Conditions | Yield |
|---|---|
| In hexane; benzene at 20℃; for 1h; Inert atmosphere; Glovebox; | 99% |
BORON TRICHLORIDE Chemical Properties
Molecular Structure:
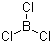
Molecular Formula:BCl3
Molecular Weight:117.16
EINECS:233-658-4
Density:0.738 g/cm3
Melting Point:-107 ℃
Boiling Point:12.5 ℃ at 760 mmHg
Flash Point:-17 ℃
Solubility:Decomposes in water
BORON TRICHLORIDE Uses
Boron trichloride(10294-34-5) is originally used for production of elemental boron.
BORON TRICHLORIDE Toxicity Data With Reference
| 1. | ihl-rat LCLo:20 ppm/7H | 14KTAK Boron, Metallo-Boron Compounds and Boranes R.M. Adams,New York, NY.: Wiley,1964,726. | ||
| 2. | ihl-mus LCLo:20 ppm/7H | 14KTAK Boron, Metallo-Boron Compounds and Boranes R.M. Adams,New York, NY.: Wiley,1964,726. |
BORON TRICHLORIDE Consensus Reports
BORON TRICHLORIDE Safety Profile
Hazard Codes:
 T+,T,
T+,T, N,
N, F
F Risk Statements: 14-26/28-36/37/38-40-67-65-62-51/53-48/20-34-11-50/53-26/27/28-63-39/23/24/25-24-21-10
14: Reacts violently with water
26/28: Very Toxic by inhalation and if swallowed
36/37/38: Irritating to eyes, respiratory system and skin
40: Limited evidence of a carcinogenic effect
67: Vapors may cause drowsiness and dizziness
65: Harmful: May cause lung damage if swallowed
62: Possible risk of impaired fertility
51/53: Toxic to aquatic organisms, may cause long-term adverse effects in the aquatic environment
48/20: Harmful: danger of serious damage to health by prolonged exposure through inhalation
34: Causes burns
11: Highly Flammable
50/53: Very Toxic to aquatic organisms, may cause long-term adverse effects in the aquatic environment
26/27/28: Very Toxic by inhalation, in contact with skin and if swallowed
63: Possible risk of harm to the unborn child
39/23/24/25: Toxic: danger of very serious irreversible effects through inhalation, in contact with skin and if swallowed
24: Toxic in contact with skin
21: Harmful in contact with skin
10: Flammable
Safety Statements: 9-26-28-36/37/39-45-8-61-38-28A-16-1-60-33-23-7/9-62
9: Keep container in a well-ventilated place
26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice
28: After contact with skin, wash immediately with plenty of ... (to be specified by the manufacturer)
36/37/39: Wear suitable protective clothing, gloves and eye/face protection
45: In case of accident or if you feel unwell, seek medical advice immediately (show label where possible)
8: Keep container dry
61: Avoid release to the environment. Refer to special instructions safety data sheet
38: In case of insufficient ventilation, wear suitable respiratory equipment
28: After contact with skin, wash immediately with plenty of ... (to be specified by the manufacturer)
16: Keep away from sources of ignition - No smoking
1: Keep locked up
60: This material and/or its container must be disposed of as hazardous waste
33: Take precautionary measures against static discharges
23: Do not breathe gas/fumes/vapor/spray (appropriate wording to be specified by the manufacturer)
7/9: Keep container tightly closed and in a well-ventilated place
62: If swallowed, do not induce vomiting: seek medical advice immediately and show this container or label
RIDADR: UN 3390 6.1/PG 1
WGK Germany: 3
RTECS: ED1925000
F: 10-21
F 10 Keep under argon
F 12 Shake before use
Hazard Note: Very toxic
HazardClass: 2.3
BORON TRICHLORIDE Standards and Recommendations
Related Products
- Boron
- BORON AZIDE DIIODIDE
- Boron bromide diiodide
- Boron carbide
- Boron nitride
- Boron oxide
- Boron phosphate
- Boron silicide (B<sub>4</sub>Si)
- Boron Sodium Oxide Hydrate
- Boron tribromide
- 10294-38-9
- 10294-40-3
- 10294-41-4
- 10294-42-5
- 10294-50-5
- 10294-54-9
- 10294-56-1
- 10294-68-5
- 10294-70-9
- 102947-88-6
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View