-
Name
Linoleic acid
- EINECS 200-470-9
- CAS No. 60-33-3
- Article Data102
- CAS DataBase
- Density 0.912 g/cm3
- Solubility insoluble in water
- Melting Point -5 °C
- Formula C18H32O2
- Boiling Point 360.552 °C at 760 mmHg
- Molecular Weight 280.451
- Flash Point 272.976 °C
- Transport Information
- Appearance Colorless to straw colored liquid
- Safety 26-24/25-36
- Risk Codes 36
-
Molecular Structure
-
Hazard Symbols
 Xi
Xi
- Synonyms 9,12-Octadecadienoicacid (Z,Z)-;Linoleic acid (8CI);(9Z,12Z)-9,12-Octadecadienoic acid;(Z,Z)-9,12-Octadecadienoic acid;9,12-Octadecadienoic acid, (Z,Z)-;9-cis,12-cis-Linoleic acid;9Z,12Z-Linoleic acid;9Z,12Z-Octadecadienoic acid;Emersol 315;Extra Linoleic 90;all-cis-9,12-Octadecadienoic acid;cis,cis-Linoleic acid;cis-9,cis-12-Octadecadienoic acid;cis-D9,12-Octadecadienoic acid;
- PSA 37.30000
- LogP 5.88450
Synthetic route
-
-
19836-71-6
linoleic nitrile

-
-
60-33-3
linoleic acid

| Conditions | Yield |
|---|---|
| With sodium hydroxide In ethanol; water Heating; | 81% |
-
-
141-22-0
Ricinoleic acid

-
A
-
544-70-7, 544-71-8, 872-23-1, 1839-11-8, 2540-56-9
cis-9,trans-11-octadecadienoic acid

-
B
-
544-70-7, 544-71-8, 872-23-1, 1839-11-8, 2540-56-9
trans-9,trans-11-octadecadienoic acid

-
C
-
1072-36-2, 2420-44-2, 2420-56-6, 7307-45-1, 22880-03-1
trans-10,cis-12-octadecadienoic acid

-
D
-
60-33-3
linoleic acid

-
E
-
1072-36-2
10E,12E-octadecadienoic acid

-
F
-
59744-10-4, 62272-86-0, 68399-13-3, 69651-29-2, 127062-51-5, 127062-52-6, 127062-54-8
(R,Z)-13-hexyloxacyclotridec-10-en-2-one

| Conditions | Yield |
|---|---|
| With 3-butyl-1-methyl-1H-imidazol-3-ium hexafluorophosphate; potassium hydroxide In ethylene glycol at 180℃; for 0.5h; Microwave irradiation; | A n/a B n/a C n/a D 22% E n/a F 46% |
| With 1-n-butyl-3-methylimidazolim bromide; potassium hydroxide In ethylene glycol at 180℃; for 0.5h; Microwave irradiation; | A n/a B n/a C n/a D 12% E n/a F 9% |


-
B
-
112-80-1
cis-Octadecenoic acid

-
C
-
60-33-3
linoleic acid

-
D
-
463-40-1
(9Z,12Z,15Z)-octadeca-9-12,15-trienoic acid

-
E
-
57-11-4
stearic acid

| Conditions | Yield |
|---|---|
| With hydrogen; mineral malachite at 165℃; under 3102.97 Torr; for 4h; Conversion of starting material; | A 16.23% B 37.82% C 41.48% D 2.92% E 5.39% |

-
-
2462-85-3, 2566-97-4, 20221-26-5, 20221-27-6, 112-63-0
linoleic acid methyl ester

-
-
60-33-3
linoleic acid

| Conditions | Yield |
|---|---|
| With potassium hydroxide |

| Conditions | Yield |
|---|---|
| With quinoline; Lindlar's catalyst; Petroleum ether Hydrogenation; | |
| With pyridine; palladium on activated charcoal; Lindlar's catalyst Hydrogenation; |

-
-
60-33-3
linoleic acid

| Conditions | Yield |
|---|---|
| With potassium hydroxide In methanol | |
| With water; sodium hydroxide In methanol for 1h; Heating; |
-
-
1794-89-4, 54324-50-4, 56846-03-8, 56846-04-9
rac-(9R,10R,12S,13S)-9,10,12,13-Tetrabromostearic acid

-
-
60-33-3
linoleic acid

| Conditions | Yield |
|---|---|
| With tetrabutylammonium tetrafluoroborate In N,N-dimethyl-formamide -1.4 V (vs. SCE) at a mercury pool (divided cell); Yield given; |
-
-
119520-51-3
(9Z,12Z)-N-(pyridin-3-yl)octadeca-9,12-dienamide

-
-
60-33-3
linoleic acid

| Conditions | Yield |
|---|---|
| With carbon disulfide; n-butyllithium; benzeneseleninic acid; 1,1,1,3,3,3-hexamethyl-disilazane 1.) THF, hexane, -20 deg C, 30 min, 2.) THF, rt, 3.) THF, rt, 5 h; Yield given. Multistep reaction; |

-
A
-
60-33-3
linoleic acid

| Conditions | Yield |
|---|---|
| With boric acid-borax buffer; Lipase type XI; Triton X-100 at 38℃; for 1h; |

-
-
60-33-3
linoleic acid

| Conditions | Yield |
|---|---|
| With diethyl ether; magnesium anschliessend mit Kohlendioxid; |
-
-
2258-92-6, 2277-28-3, 67968-46-1, 107380-08-5, 126455-47-8, 126455-48-9
1-monolinoleoyl-rac-glycerol

-
-
60-33-3
linoleic acid

-
-
60-33-3
linoleic acid
-
-
7647-01-0
hydrogenchloride

-
-
64-17-5
ethanol

-
-
1794-89-4
9,10,12,13-tetrabromooctadecanoic acid

-
-
60-33-3
linoleic acid

-
-
7647-01-0
hydrogenchloride

-
-
64-17-5
ethanol

-
-
1794-89-4
9,10,12,13-tetrabromooctadecanoic acid

-
A
-
60-33-3
linoleic acid

| Conditions | Yield |
|---|---|
| #threo,threo!-isomer(ic); |

-
-
119520-47-7
(9Z,12Z)-N-Pyridin-3-yl-octadeca-9,12-dienimidothioic acid pyridin-2-yl ester

-
-
60-33-3
linoleic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 89 percent / water / dioxane / 40 h / Ambient temperature 2: 1.) hexamethyldisilazane, 1.6 M n-BuLi, 2.) CS2, 3.) PhSeO2H / 1.) THF, hexane, -20 deg C, 30 min, 2.) THF, rt, 3.) THF, rt, 5 h View Scheme |
-
-
119520-41-1
(9Z,12Z)-Octadeca-9,12-dienoic acid 2-thioxo-2H-pyridin-1-yl ester

-
-
60-33-3
linoleic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 97 percent / CH2Cl2 / 0 °C / Irradiation 2: 81 percent / NaOH / ethanol; H2O / Heating View Scheme | |
| Multi-step reaction with 2 steps 1: 83 percent Spectr. / CH2Cl2 / 0 °C / Irradiation 2: 81 percent / NaOH / ethanol; H2O / Heating View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: tetrahydrofuran; ethyl magnesium bromide / anschliessenden Erwaermen des Reaktionsgemisches mit 1-Brom-oct-2-in und Kupfer(I)-cyanid 2: Lindlar-catalyst; quinoline; petroleum ether / Hydrogenation View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1: H2SO4 / benzene 2: NaI / acetone 3: Mg, I2, HgCl2 / diethyl ether 4: H2 / Pd / methanol 5: KOH / methanol View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: H2 / Pd / methanol 2: KOH / methanol View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: NaI / acetone 2: Mg, I2, HgCl2 / diethyl ether 3: H2 / Pd / methanol 4: KOH / methanol View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: Mg, I2, HgCl2 / diethyl ether 2: H2 / Pd / methanol 3: KOH / methanol View Scheme |

-
B
-
13296-76-9
9,11,13-octadecatrienoic acid

-
C
-
104096-79-9
10,12,14-C18:3

-
D
-
25574-96-3
10E,12Z,14E-C18:3

-
E
-
112-80-1
cis-Octadecenoic acid

-
F
-
544-70-7, 544-71-8, 872-23-1, 1839-11-8, 2540-56-9
cis-9,trans-11-octadecadienoic acid

-
G
-
1072-36-2, 2420-44-2, 2420-56-6, 7307-45-1, 22880-03-1
trans-10,cis-12-octadecadienoic acid

-
H
-
506-17-2
cis-vaccenic acid

-
I
-
60-33-3
linoleic acid

-
J
-
463-40-1
(9Z,12Z,15Z)-octadeca-9-12,15-trienoic acid

-
K
-
25491-26-3
9-(6-propyl-cyclohexa-2,4-dienyl)-nonanoic acid

-
L
-
15909-18-9
rumelenic acid

-
M
-
57-10-3
1-hexadecylcarboxylic acid

-
N
-
57-11-4
stearic acid

| Conditions | Yield |
|---|---|
| With sodium hydroxide In propylene glycol at 160℃; for 2h; |


-
A
-
657403-47-9
6Z,8E,12Z-C18:3

-
C
-
25692-11-9
cis-Δ9-docosenoic acid

-
D
-
112-80-1
cis-Octadecenoic acid

-
E
-
544-70-7, 544-71-8, 872-23-1, 1839-11-8, 2540-56-9
cis-9,trans-11-octadecadienoic acid

-
F
-
1072-36-2, 2420-44-2, 2420-56-6, 7307-45-1, 22880-03-1
trans-10,cis-12-octadecadienoic acid

-
G
-
506-17-2
cis-vaccenic acid

-
H
-
60-33-3
linoleic acid

-
I
-
29204-02-2
gadoleic acid

-
J
-
463-40-1
(9Z,12Z,15Z)-octadeca-9-12,15-trienoic acid

-
K
-
57-10-3
1-hexadecylcarboxylic acid

-
L
-
57-11-4
stearic acid

-
M
-
109241-60-3
(6Z,10E,12Z)-octadeca-6,10,12-trienoic acid

| Conditions | Yield |
|---|---|
| With sodium hydroxide In propylene glycol at 160℃; for 2h; |


| Conditions | Yield |
|---|---|
| With potassium hydroxide In methanol | |
| With water; sodium hydroxide In methanol for 1h; Heating; |

-
A
-
60-33-3
linoleic acid

| Conditions | Yield |
|---|---|
| With boric acid-borax buffer; Lipase type XI; Triton X-100 at 38℃; for 1h; |

-
A
-
373-49-9
cis-9-hexadecenoic acid

-
B
-
112-80-1
cis-Octadecenoic acid

-
C
-
112-79-8
Elaidic acid

-
D
-
544-70-7
(9Z,12Z)-octadeca-9,11-dienoic acid

-
E
-
544-70-7, 544-71-8, 872-23-1, 1839-11-8, 2540-56-9
cis-9,trans-11-octadecadienoic acid

-
F
-
7307-45-1
(10Z,12Z)-10,12-Octadecadienoic acid

-
G
-
1072-36-2, 2420-44-2, 2420-56-6, 7307-45-1, 22880-03-1
trans-10,cis-12-octadecadienoic acid

-
H
-
60-33-3
linoleic acid

-
I
-
60-33-3
(9E,12E)-Octadeca-9,12-dienoic acid

-
J
-
29204-02-2
gadoleic acid

-
K
-
506-30-9
Arachidic acid

-
L
-
112-85-6
n-docosanoic acid

-
M
-
544-63-8
n-tetradecanoic acid

-
N
-
57-10-3
1-hexadecylcarboxylic acid

-
O
-
57-11-4
stearic acid

-
P
-
117624-52-9
octadeca-11Z,13Z-dienoic acid

| Conditions | Yield |
|---|---|
| With sodium hydroxide; ethanol; water at 150 - 215℃; under 7500.75 - 24002.4 Torr; for 0 - 6h; Product distribution / selectivity; | |
| With potassium hydroxide; ethanol; water at 150℃; under 7500.75 - 9000.9 Torr; for 0 - 6h; Product distribution / selectivity; | |
| With potassium hydroxide; water In propylene glycol at 150℃; under 7500.75 - 9000.9 Torr; for 0 - 6h; Product distribution / selectivity; |


| Conditions | Yield |
|---|---|
| With hydrogenchloride; water for 1h; Heating / reflux; | |
| With water In n-heptane; tert-butyl methyl ether at 37℃; for 24h; Enzymatic reaction; | |
| With 2C33H37N*H2O7S2; water at 80℃; | 90 %Spectr. |
-
-
62439-44-5
methyl octadec-9,12-diynoic acid

-
A
-
60-33-3
linoleic acid

-
B
-
79050-23-0
[9,10,12,13-2H]-(Z,Z)-9,12-octadecadienoic acid

| Conditions | Yield |
|---|---|
| Stage #1: methyl octadec-9,12-diynoic acid With benzo[1,3,2]dioxaborole at 60℃; for 5h; Stage #2: With deuteroacetic acid; acetic acid for 5h; Heating; Stage #3: With potassium hydroxide In water for 1h; Heating; Further stages. Title compound not separated from byproducts.; |

| Conditions | Yield |
|---|---|
| With Saccharomyces cerevisiae IFO10150 expressing Saccharomyces kluyveri Δ12 fatty acid desaturase Sk-FAD2 In water at 30℃; |

| Conditions | Yield |
|---|---|
| Stage #1: β-sitosteryl glucoside-3'-O-linoleate With sodium hydroxide In tetrahydrofuran; methanol at 50℃; for 2h; Stage #2: With hydrogenchloride In tetrahydrofuran; methanol; water |
-
-
1190855-79-8
neomastoidin A

-
B
-
4219-94-7, 19794-53-7, 32620-11-4, 86118-11-8
DL-3-methoxy-alanine

-
C
-
60-33-3
linoleic acid

| Conditions | Yield |
|---|---|
| With ethanol; potassium hydroxide at 25℃; |


| Conditions | Yield |
|---|---|
| With oxalyl dichloride In dichloromethane at 20℃; | 100% |
| With oxalyl dichloride | 99% |
| With oxalyl dichloride In dichloromethane at 0 - 20℃; for 4h; | 92% |
-
-
60-33-3
linoleic acid

-
-
100-79-8
(R,S)-2,2-dimethyl-1,3-dioxolane-4-methanol

-
-
127592-95-4
(+/-)-2,2-dimethyl-1,3-dioxolan-4-ylmethyl (9Z,12Z)-9,12-octadecadienoate

| Conditions | Yield |
|---|---|
| With dmap In dichloromethane at 20℃; | 100% |
| With dmap; dicyclohexyl-carbodiimide | 89% |
| With dmap; dicyclohexyl-carbodiimide In diethyl ether at 20 - 25℃; for 4.5h; Esterification; |

| Conditions | Yield |
|---|---|
| With dmap In tetrahydrofuran for 0.166667h; | 100% |
-
-
60-33-3
linoleic acid

-
-
22323-82-6
(S)-(+)-(2,2-dimethyl-[1,3]dioxolan-4-yl)methanol

-
-
2518-17-4
(R)-2,2-dimethyl-1,3-dioxolan-4-ylmethyl (9Z,12Z)-9,12-octadecadienoate

| Conditions | Yield |
|---|---|
| With dmap; dicyclohexyl-carbodiimide In dichloromethane at 0 - 20℃; | 100% |
| With N-(3-dimethylaminopropyl)-N-ethylcarbodiimide In tetrahydrofuran at 0℃; |

-
-
60-33-3
linoleic acid

-
-
890843-15-9
N-{2-[(2,6-dimethylphenyl)-amino]-2-oxoethyl}-N,N-diethyl-benzenemethanaminium linolate

| Conditions | Yield |
|---|---|
| In methanol at 30 - 60℃; for 1 - 2h; | 100% |
-
-
60-33-3
linoleic acid

-
-
69256-58-2
1-palmitoyl-3-O-trityl-rac-glycerol

-
-
920271-16-5
1-palmitoyl-2-linoleoyl-3-trityl-glycerol

| Conditions | Yield |
|---|---|
| With dmap; dicyclohexyl-carbodiimide In n-heptane at 20℃; for 3h; | 100% |
| With dicyclohexyl-carbodiimide; dmap In n-heptane at 20℃; for 3h; |
-
-
920112-92-1
1-O-palmitoyl-3-O-tertbutyldimethylsilyl-sn-glycerol

-
-
60-33-3
linoleic acid

-
-
128052-49-3
1-palmitoyl-2-linoleoyl-3-t-butyl-dimethylsilyl-glycerol

| Conditions | Yield |
|---|---|
| With dmap; dicyclohexyl-carbodiimide In n-heptane at 20℃; for 3h; | 100% |
| With dicyclohexyl-carbodiimide; dmap In n-heptane at 20℃; for 3h; |

| Conditions | Yield |
|---|---|
| In acetone for 2h; | 100% |
-
-
60-33-3
linoleic acid

-
-
2450-71-7
Propargylamine

-
-
1312773-81-1
(9Z,12Z)-N-(prop-2-yn-1-yl)octadeca-9,12-dienamide

| Conditions | Yield |
|---|---|
| With dmap; diisopropyl-carbodiimide In dichloromethane at 20℃; for 18h; | 100% |
-
-
60-33-3
linoleic acid

-
-
162558-25-0
N-α-tert-butoxycarbonyl-N-β-(9-fluorenylmethoxycarbonyl)-L-diaminopropionic acid

| Conditions | Yield |
|---|---|
| With dmap; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride In dichloromethane at 20℃; for 17h; | 100% |

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 40 - 50℃; for 5h; | 100% |

| Conditions | Yield |
|---|---|
| Stage #1: doxepin; linoleic acid In tetrahydrofuran at 40 - 50℃; for 5h; Stage #2: 4-amino-n-butyric acid In tetrahydrofuran; methanol at 50℃; for 1h; | 100% |


| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 40 - 50℃; for 5h; | 100% |

| Conditions | Yield |
|---|---|
| Stage #1: linoleic acid; benzydamine In tetrahydrofuran at 40 - 50℃; for 5h; Stage #2: 4-amino-n-butyric acid In tetrahydrofuran; methanol at 50℃; for 1h; | 100% |


| Conditions | Yield |
|---|---|
| With sulfuric acid at 0℃; Reflux; | 99% |
| With 1-hexyl-3-methylimidazolium hydrogen sulphate In cyclohexane at 40℃; for 8h; Reagent/catalyst; Temperature; | 91.07% |
| With lipase from Geotrichum candidum strain NRRL Y-552, 65 kDa, immobilized on poly-hydroxybutyrate (PHB) particles In n-heptane at 35℃; for 2h; pH=7; Concentration; Temperature; Green chemistry; Enzymatic reaction; | 70% |

| Conditions | Yield |
|---|---|
| With dmap; dicyclohexyl-carbodiimide In dichloromethane at 20℃; for 2.5h; | 99% |
-
-
60-33-3
linoleic acid

-
-
19670-51-0, 32899-41-5, 542-44-9, 5309-46-6
(R)-2,3-dihydroxypropyl n-hexadecanoate

-
-
66964-28-1
3-hexadecanoyl-1,2-di(cis-octadec-9,12-dienoyl)-sn-glycerol

| Conditions | Yield |
|---|---|
| With dmap; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride In dichloromethane at 20℃; | 99% |
-
-
60-33-3
linoleic acid

-
-
6638-79-5
N,O-dimethylhydroxylamine*hydrochloride

-
-
577973-81-0
(9Z,12Z)-N-methoxy-N-methyloctadeca-9,12-dienamide

| Conditions | Yield |
|---|---|
| With 1-hydroxybenzimidazole hydrate; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride; triethylamine In dichloromethane at 20℃; | 99% |

| Conditions | Yield |
|---|---|
| In methanol at 50℃; for 2h; Green chemistry; | 99% |

-
-
112-80-1
cis-Octadecenoic acid

-
-
60-33-3
linoleic acid

-
-
488711-07-5
N-methyl-N,N,N-trioctylammonium methylcarbonate

| Conditions | Yield |
|---|---|
| In methanol at 50℃; for 2h; Green chemistry; | 99% |

-
-
112-80-1
cis-Octadecenoic acid

-
-
60-33-3
linoleic acid

-
-
1204316-79-9
1,1,1-trioctyl-1-methylphosphonium methylcarbonate

| Conditions | Yield |
|---|---|
| In methanol at 50℃; for 2h; Green chemistry; | 99% |

-
-
60-33-3
linoleic acid

| Conditions | Yield |
|---|---|
| In methanol at 50℃; for 2h; Green chemistry; | 99% |
-
-
60-33-3
linoleic acid

-
-
488711-07-5
N-methyl-N,N,N-trioctylammonium methylcarbonate

-
-
1416991-40-6
methyltrioctylammonium linoleate

| Conditions | Yield |
|---|---|
| In methanol at 50℃; for 2h; Green chemistry; | 99% |
-
-
60-33-3
linoleic acid

-
-
1204316-79-9
1,1,1-trioctyl-1-methylphosphonium methylcarbonate

| Conditions | Yield |
|---|---|
| In methanol at 50℃; for 2h; Green chemistry; | 99% |
-
-
60-33-3
linoleic acid

| Conditions | Yield |
|---|---|
| Stage #1: linoleic acid With sodium hydride In tetrahydrofuran for 0.5h; Inert atmosphere; Stage #2: 2-((difluoromethyl)thio)-3-methylbenzo[d]thiazol-3-ium trifluoromethanesulfonate In tetrahydrofuran at 20℃; for 2h; Inert atmosphere; | 99% |

| Conditions | Yield |
|---|---|
| With lithium aluminium tetrahydride In tetrahydrofuran at 0℃; for 3h; | 98% |
| With lithium aluminium tetrahydride In tetrahydrofuran Reflux; | 91% |
| With lithium aluminium tetrahydride In tetrahydrofuran at 0 - 25℃; | 91% |
-
-
60-33-3
linoleic acid

-
-
17364-00-0
1,3-dibutanoyloxy-2-propanol

-
-
402562-28-1
2-((9Z,12Z)-octadeca-9,12-dienoyloxy)propane-1,3-diyl dibutyrate

| Conditions | Yield |
|---|---|
| With dmap; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride In tetrahydrofuran; dichloromethane at 0℃; for 4h; | 98% |
-
-
60-33-3
linoleic acid

-
-
6076-30-8
1,2-di-O-palmitoyl-sn-glycerol

-
-
102491-55-4
2,3-dihexadecanoyl-1-cis-octadec-9,12-dienoyl-sn-glycerol

| Conditions | Yield |
|---|---|
| With dmap; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride In dichloromethane at 20℃; | 98% |

| Conditions | Yield |
|---|---|
| With hydrogen In methanol at 20℃; for 4h; | 97.5% |
Linoleic acid Consensus Reports
Reported in EPA TSCA Inventory.
Linoleic acid Specification
The CAS registry number of Linoleic acid is 60-33-3. With the EINECS registry number 200-470-9, its systematic name is (9Z,12Z)-octadeca-9,12-dienoic acid. It is a colorless liquid at room temperature. And it belongs to one of the two families of essential fatty acids that humans and other animals must ingest for good health. In addition, it is used in making soaps, emulsifiers, and quick-drying oils. Besides, it has become increasingly popular in the beauty products industry because of its beneficial properties on the skin.
Physical properties about this chemical are: (1)ACD/LogP: 7.02; (2)# of Rule of 5 Violations: 1; (3)ACD/LogD (pH 5.5): 6.219; (4)ACD/LogD (pH 7.4): 4.424; (5)ACD/BCF (pH 5.5): 20180.996; (6)ACD/BCF (pH 7.4): 323.607; (7)ACD/KOC (pH 5.5): 24903.602; (8)ACD/KOC (pH 7.4): 399.335; (9)#H bond acceptors: 2; (10)#H bond donors: 1; (11)#Freely Rotating Bonds: 14; (12)Polar Surface Area: 37.3 Å2; (13)Index of Refraction: 1.478; (14)Molar Refractivity: 87.121 cm3; (15)Molar Volume: 307.512 cm3; (16)Polarizability: 34.537 ×10-24cm3; (17)Surface Tension: 34.355 dyne/cm; (18)Density: 0.912 g/cm3; (19)Flash Point: 272.976 °C; (20)Enthalpy of Vaporization: 66.597 kJ/mol; (21)Boiling Point: 360.552 °C at 760 mmHg.
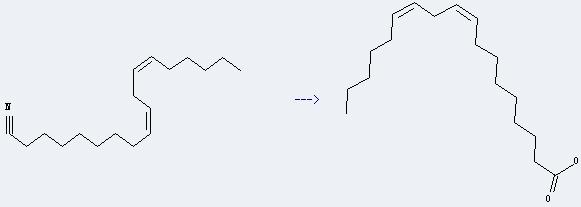
Preparation of Linoleic acid: it can be prepared by octadeca-9c,12c-dienenitrile. This reaction will need reagent NaOH and solvents ethanol and H2O. The yield is about 81% by heating.
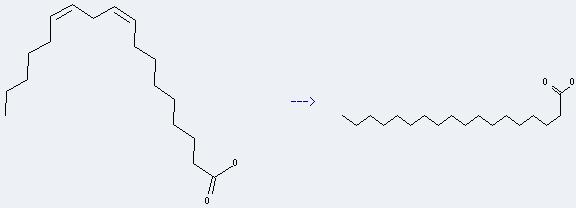
Uses of Linoleic acid: it can be used to get octadecanoic acid. This reaction will need reagent H2, catalyst Rhodium chloride tri(triphenylphosphine-meta-trisulfonate) and solvent H2O. The reaction time is 18 hours with ambient temperature. The yield is about 95%.
When you are using this chemical, please be cautious about it as the following:
This chemical is irritating to eyes. During using it, wear suitable protective clothing and avoid contact with skin and eyes. In case of contact with eyes, rinse immediately with plenty of water and seek medical advice.
You can still convert the following datas into molecular structure:
(1)SMILES: CCCCC/C=C\C/C=C\CCCCCCCC(=O)O
(2)Std. InChI: InChI=1S/C18H32O2/c1-2-3-4-5-6-7-8-9-10-11-12-13-14-15-16-17-18(19)20/h6-7,9-10H,2-5,8,11-17H2,1H3,(H,19,20)/b7-6-,10-9-
(3)Std. InChIKey: OYHQOLUKZRVURQ-HZJYTTRNSA-N
The toxicity data is as follows:
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| mouse | LD50 | intraperitoneal | 280mg/kg (280mg/kg) | Yakugaku Zasshi. Journal of Pharmacy. Vol. 104, Pg. 793, 1984. | |
| mouse | LD50 | oral | > 50gm/kg (50000mg/kg) | United States Patent Document. Vol. #3728459, | |
| rat | LD50 | intraperitoneal | > 50gm/kg (50000mg/kg) | Oyo Yakuri. Pharmacometrics. Vol. 4, Pg. 327, 1970. |
Related Products
- Linoleic acid
- Linoleic acid, hydroperoxy-, methyl ester
- 603-33-8
- 603-34-9
- 603-35-0
- 60335-71-9
- 603-36-1
- 603-40-7
- 603-41-8
- 60-34-4
- 603-44-1
- 6034-46-4
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View