-
Name
m-Toluic acid
- EINECS 202-723-9
- CAS No. 99-04-7
- Article Data350
- CAS DataBase
- Density 1.151 g/cm3
- Solubility water: 1.054 g/mL at 25 °C(lit.)
- Melting Point 108 °C
- Formula C8H8O2
- Boiling Point 263.8 °C at 760 mmHg
- Molecular Weight 136.15
- Flash Point 120 °C
- Transport Information
- Appearance off-white crystalline solid
- Safety 22-24/25-36
- Risk Codes 22
-
Molecular Structure
-
Hazard Symbols
 Xn
Xn
- Synonyms 3-methylbenzoate; zinc(+2) cation;Benzoic acid, 3-methyl-, cadmium salt;beta-Bethylbenzoic acid;Zinc 3-methylbenzoate;m-Methylbenzoate;Barium 3-methylbenzoate;Benzoic acid, 3-methyl-;m-Methylbenzoic acid;Cadmium 3-methylbenzoate;Benzoic acid, 3-methyl-, barium salt;beta-Methylbenzoic acid;Barium m-toluate;3-methylbenzoate;3-methylbenzoic acid;Zinc m-toluate;1/C8H8O2/c1-6-3-2-4-7(5-6)8(9)10/h2-5H,1H3,(H,9,10;cadmium(+2) cation; 3-methylbenzoate;Benzoic acid, 3-methyl-, zinc salt;Cadmium m-toluate;m-Toluicacid;
- PSA 37.30000
- LogP 1.69320
Synthetic route

| Conditions | Yield |
|---|---|
| With 2-mesityl-6,7-dihydro-5H-pyrrolo[2,1-c][1,2,4]triazol-2-ium tetrafluoroborate; 1,3,4,6,7,8-hexahydro-2H-pyrimido[1,2-a]pyrimidine In acetonitrile at 20℃; | 99% |
| With tert.-butylhydroperoxide; water; iodine; sodium hydroxide at 70℃; pH=10; Green chemistry; | 95% |
| With 2-mesityl-6,7-dihydro-5H-pyrrolo[2,1-c][1,2,4]triazol-2-ium tetrafluoroborate; 1,4-diaza-bicyclo[2.2.2]octane; oxygen In tetrahydrofuran at 20℃; for 16h; | 95% |

| Conditions | Yield |
|---|---|
| With amphiphilic resin-supported phosphine-palladium; water; potassium carbonate at 25℃; under 760 Torr; hydroxycarbonylation; | 99% |
| With water; potassium carbonate In acetonitrile at 100℃; under 3750.38 Torr; for 0.0161111h; | 98% |
| With 4,4’‐bis(trimethylammoniummethyl)‐2,2’‐bipyridine; bis-triphenylphosphine-palladium(II) chloride; water; sodium carbonate In water at 100℃; under 7600.51 Torr; for 24h; Catalytic behavior; Autoclave; | 86% |
| With water; palladium diacetate; triethylamine; 4,5-bis(diphenylphos4,5-bis(diphenylphosphino)-9,9-dimethylxanthenephino)-9,9-dimethylxanthene In 1,4-dioxane at 100℃; under 11251.1 Torr; Flow reactor; | 58% |

| Conditions | Yield |
|---|---|
| With water; sodium hydroxide at 100℃; for 4h; Temperature; | 98.5% |
| With hydrogenchloride im Druckrohr; | |
| With steam; thorium dioxide at 420℃; | |
| With sulfuric acid | |
| With nitrilase from Gordonia terrae In aq. phosphate buffer at 35℃; for 1h; pH=8; Enzymatic reaction; |
-
-
61560-94-9
3-methylphenyloxoacetic acid

-
-
99-04-7
m-Toluic acid

| Conditions | Yield |
|---|---|
| With oxone In N,N-dimethyl-formamide for 4h; | 98% |
-
-
73318-83-9
2-oxo-2-(m-tolyl)acetaldehyde

-
-
99-04-7
m-Toluic acid

| Conditions | Yield |
|---|---|
| With oxone In N,N-dimethyl-formamide for 4h; | 98% |
| With tert.-butylhydroperoxide for 7h; | 82% |

| Conditions | Yield |
|---|---|
| With diethylene glycol dimethyl ether at 70℃; for 0.5h; Sonication; | 97% |
| With oxygen; sodium hydroxide In water at 80℃; for 10h; Catalytic behavior; | 97% |
| With tert.-butylhydroperoxide; water; iodine; sodium hydroxide at 70℃; pH=10; Green chemistry; | 90% |

| Conditions | Yield |
|---|---|
| With potassium hydroxide; palladium dichloride In water at 100℃; for 5h; Condensation; | 97% |
-
-
136125-68-3
methyl 3-methylphenylglyoxylate

-
-
99-04-7
m-Toluic acid

| Conditions | Yield |
|---|---|
| With oxone In N,N-dimethyl-formamide for 4h; | 97% |
-
-
108-38-3
m-xylene

-
A
-
121-91-5
isophthalic acid

-
B
-
619-21-6
m-formylphenyl benzoic acid

-
C
-
99-04-7
m-Toluic acid

| Conditions | Yield |
|---|---|
| With dihydrogen peroxide In dimethyl sulfoxide at 70℃; for 10h; Catalytic behavior; Reagent/catalyst; Overall yield = > 99 %; | A 96% B n/a C n/a |
| With cobalt(II) acetate; manganese(II) acetate; acetic acid; 3-benzyl-1-methylimidazolium bromide at 215℃; for 3h; |

-
-
53663-39-1
3-methyl-2-bromobenzoic acid

-
-
99-04-7
m-Toluic acid

| Conditions | Yield |
|---|---|
| With isopropyl alcohol for 5h; Schlenk technique; Inert atmosphere; Irradiation; Heating; | 96% |
| With [RhCl2(p-cymene)]2; cesium acetate In isopropyl alcohol at 20 - 100℃; for 24h; Inert atmosphere; | 94% |
| With [ruthenium(II)(η6-1-methyl-4-isopropyl-benzene)(chloride)(μ-chloride)]2; cesium acetate; isopropyl alcohol at 100℃; Schlenk technique; Inert atmosphere; | 93% |
-
-
15068-35-6
2-chloro-3-methylbenzoic acid

-
-
99-04-7
m-Toluic acid

| Conditions | Yield |
|---|---|
| With isopropyl alcohol for 20h; Schlenk technique; Inert atmosphere; Irradiation; Heating; | 96% |

| Conditions | Yield |
|---|---|
| With iodine; aluminium In acetonitrile at 80℃; for 18h; | 95% |
| With iron(III) chloride hexahydrate In glycerol at 70℃; for 14h; | 93% |
| With magnesium iodide In toluene for 72h; Heating; | 48% |

| Conditions | Yield |
|---|---|
| With copper(l) iodide; potassium carbonate In dimethyl sulfoxide at 100℃; for 24h; Inert atmosphere; | 95% |

| Conditions | Yield |
|---|---|
| With palladium diacetate; triethylamine; dicyclohexyl-carbodiimide; 4,5-bis(diphenylphosphino)-9,9-dimethylxanthene In N,N-dimethyl-formamide at 80℃; for 10h; Inert atmosphere; Sealed tube; | 94% |
| With palladium diacetate; triethylamine; dicyclohexyl-carbodiimide; 4,5-bis(diphenylphosphino)-9,9-dimethylxanthene In N,N-dimethyl-formamide at 100℃; for 20h; Inert atmosphere; Sealed tube; | 82% |

-
-
99-04-7
m-Toluic acid

| Conditions | Yield |
|---|---|
| With H-β zeolite; methoxybenzene In toluene for 2.5h; Heating; | 93% |

| Conditions | Yield |
|---|---|
| With oxygen; sodium t-butanolate In tetrahydrofuran at 20℃; under 750.075 Torr; for 22h; chemoselective reaction; | 93% |
-
-
15226-74-1, 61091-28-9, 61117-58-6
dicobalt octacarbonyl

-
-
32578-31-7
3-tolyl triflate

-
-
99-04-7
m-Toluic acid

| Conditions | Yield |
|---|---|
| With dichloro(1,1'-bis(diphenylphosphanyl)ferrocene)palladium(II)*CH2Cl2; potassium acetate In 1,4-dioxane; water at 20℃; for 16h; Schlenk technique; Inert atmosphere; chemoselective reaction; | 93% |

-
-
99-04-7
m-Toluic acid

| Conditions | Yield |
|---|---|
| With sodium hydroxide In water at 100℃; for 9h; Reflux; chemoselective reaction; | 93% |

| Conditions | Yield |
|---|---|
| With potassium ferrate(VI) In neat (no solvent) for 5h; Milling; | A n/a B 92.8% |
-
-
137932-33-3
benzyl 3-methylbenzoate

-
-
108-88-3
toluene

-
A
-
620-47-3
1-methyl-3-(phenylmethyl)-benzene

-
B
-
620-83-7
1-methyl-4-(phenylmethyl)benzene

-
C
-
713-36-0
2-benzyltoluene

-
D
-
99-04-7
m-Toluic acid

| Conditions | Yield |
|---|---|
| With boron trifluoride diethyl etherate; water at 80℃; for 2h; regioselective reaction; | A n/a B n/a C n/a D 92% |

-
-
38362-90-2
3-nitroxylene

-
-
99-04-7
m-Toluic acid

| Conditions | Yield |
|---|---|
| With water; tetra-(n-butyl)ammonium iodide at 80℃; for 15h; Reagent/catalyst; | 92% |
| With tetra-(n-butyl)ammonium iodide; acetic acid In water at 80℃; for 15h; Reagent/catalyst; | 87% |
| With zinc diacetate; water; tetra-(n-butyl)ammonium iodide at 80℃; for 24h; | 84% |

| Conditions | Yield |
|---|---|
| With oxone; sodium hydrogencarbonate In water; acetonitrile at 20℃; for 7h; | 91% |
-
-
15226-74-1, 61091-28-9, 61117-58-6
dicobalt octacarbonyl

-
-
99-04-7
m-Toluic acid

| Conditions | Yield |
|---|---|
| With dichloro(1,1'-bis(diphenylphosphanyl)ferrocene)palladium(II)*CH2Cl2; potassium acetate In 1,4-dioxane; water at 20℃; for 16h; Schlenk technique; Inert atmosphere; chemoselective reaction; | 91% |

| Conditions | Yield |
|---|---|
| Stage #1: lithium formate With acetic anhydride; N-ethyl-N,N-diisopropylamine In N,N-dimethyl-formamide at 20℃; Stage #2: 3-Iodotoluene With tris(dibenzylideneacetone)dipalladium (0); lithium chloride In N,N-dimethyl-formamide at 80℃; for 5.5h; | 90% |

| Conditions | Yield |
|---|---|
| With sodium cyanide In N,N-dimethyl-formamide at 80℃; for 24h; Molecular sieve; | A 90% B 6% |


| Conditions | Yield |
|---|---|
| Stage #1: m-Fluorotoluene With C78H70Al2Cl4N6P4Rh2; magnesium; ethylene dibromide In tetrahydrofuran at -30℃; for 22h; Inert atmosphere; Glovebox; Stage #2: carbon dioxide In tetrahydrofuran at 20℃; under 760.051 Torr; for 0.5h; | 90% |

| Conditions | Yield |
|---|---|
| With tetra(n-butyl)ammonium hydrogensulfate In N,N-dimethyl-formamide for 2h; Electrochemical reaction; | 88% |

| Conditions | Yield |
|---|---|
| With manganese; 2.9-dimethyl-1,10-phenanthroline; lithium acetate; cobalt(II) bromide In N,N-dimethyl acetamide at 20℃; under 760.051 Torr; for 12h; Inert atmosphere; Schlenk technique; | 88% |
| With tetrabutylammonium tetrafluoroborate In N,N-dimethyl-formamide at 20℃; under 760.051 Torr; for 4h; Electrolysis; | 78% |
| With [2,2]bipyridinyl; lithium chloride; cobalt(II) iodide; zinc In N,N-dimethyl-formamide; acetonitrile at 40℃; under 760.051 Torr; for 14h; Sealed tube; | 45% |

| Conditions | Yield |
|---|---|
| With tert.-butylhydroperoxide; water at 20℃; for 12h; Inert atmosphere; | 87% |
| With cobalt(II) 5,10,15,20-tetraphenylporphyrin; oxygen at 200℃; under 19502 Torr; Pressure; Temperature; Reagent/catalyst; | 78.9% |
| With iron(III) sulfate In water; acetonitrile for 16h; Irradiation; Sealed tube; | 75% |

| Conditions | Yield |
|---|---|
| With N-hydroxyphthalimide; air; cobalt(II) acetate; manganese(II) acetate In acetic acid at 150℃; under 22801.5 Torr; for 3h; | A 87% B 7% |
| Stage #1: m-xylene With hafnium(IV) oxide; N-hydroxy-o-sulphonyl benzamide; C32H12F4FeN8; C44H28N4O8Ru; oxygen at 145℃; under 6000.6 Torr; for 1.6h; Stage #2: With hafnium(IV) oxide; N-hydroxy-o-sulphonyl benzamide; C32H12F4FeN8; C44H28N4O8Ru; oxygen; acetic acid at 176℃; under 13501.4 Torr; for 2.2h; Temperature; Pressure; Reagent/catalyst; | A 84.4% B 15.6% |
| With N-hydroxyphthalimide; oxygen; nitric acid at 110℃; under 760.051 Torr; for 6h; Ionic liquid; | A 71% B n/a |

| Conditions | Yield |
|---|---|
| With sulfuric acid at 20℃; for 15h; Heating / reflux; | 100% |
| With sulfuric acid for 4h; Reflux; | 100% |
| With thionyl chloride at 0 - 20℃; for 5h; | 99% |

| Conditions | Yield |
|---|---|
| Stage #1: m-Toluic acid With N,N-dimethyl-formamide In dichloromethane at 0℃; for 0.0833333h; Stage #2: With oxalyl dichloride In dichloromethane at 20℃; for 12h; | 100% |
| With thionyl chloride; N,N-dimethyl-formamide at 90℃; for 3h; | 99.3% |
| With thionyl chloride; tetra(n-butyl)ammonium hydrogensulfate In 5,5-dimethyl-1,3-cyclohexadiene at 45℃; for 4.5h; Temperature; Reagent/catalyst; Solvent; Large scale; | 98.2% |

| Conditions | Yield |
|---|---|
| With Candida antarctica lipase B immobilised in a macroporous DVB crosslinked polymer (Novozym 435) In cyclohexane at 80℃; for 24h; Enzymatic reaction; | 100% |
| With diphenylphosphinopolystyrene; diethylazodicarboxylate In tetrahydrofuran at 25℃; for 4h; | 90% |
-
-
99-04-7
m-Toluic acid

-
-
31673-44-6
3-methylcyclohexa-2,5-dienecarboxylic acid

| Conditions | Yield |
|---|---|
| With sodium In ammonia | 100% |
| With ammonia; sodium In tetrahydrofuran for 0.5h; | 80% |
| With ammonia; sodium |
-
-
75685-01-7
2,2'-dimethoxy-1,1'-binaphthyl

-
-
99-04-7
m-Toluic acid

| Conditions | Yield |
|---|---|
| With methanesulfonic acid; phosphorus pentoxide at 60℃; for 24h; Friedel-Crafts acylation; | 100% |
-
-
1438397-77-3
N-(2-aminoethyl)-4-methyl-N-(prop-2-yn-1-yl)benzenesulfonamide

-
-
99-04-7
m-Toluic acid

| Conditions | Yield |
|---|---|
| With benzotriazol-1-ol; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride In dichloromethane at 20℃; for 0.5h; | 100% |
-
-
99-04-7
m-Toluic acid

-
-
59646-37-6
2,5-bis(3-methylphenyl)-1,3,4-oxadiazole

| Conditions | Yield |
|---|---|
| With hydrazine dihydrochloride; phosphoric acid; trichlorophosphate In water at 140℃; for 2h; Condensation; | 99.8% |
| With hydrazine dihydrochloride; phosphoric acid; phosphorus pentoxide at 130℃; for 0.0833333h; microwave irradiation; | 97% |
| With sodium tetrahydroborate; hydrazine dihydrochloride; phosphoric acid at 170 - 180℃; Inert atmosphere; Neat (no solvent); | 89% |
-
-
74-88-4
methyl iodide

-
-
99-04-7
m-Toluic acid

-
-
31689-43-7
1-methyl-3-methylcyclohexa-2,5-dienecarboxylic acid

| Conditions | Yield |
|---|---|
| Stage #1: m-Toluic acid With ammonia; lithium In water at -60℃; for 0.5h; Birch reduction; Stage #2: methyl iodide In water regioselective reaction; | 99% |
| (i) Li, liq. NH3, (ii) /BRN= 969135/; Multistep reaction; | |
| Stage #1: m-Toluic acid With lithium In ammonia at -78 - -60℃; Birch reduction; liquid NH3; Stage #2: methyl iodide Cooling with ice; |
-
-
74-83-9
methyl bromide

-
-
99-04-7
m-Toluic acid

-
-
31689-43-7
1-methyl-3-methylcyclohexa-2,5-dienecarboxylic acid

| Conditions | Yield |
|---|---|
| Stage #1: m-Toluic acid With ammonia; lithium at -60℃; Birch reduction; Stage #2: methyl bromide at -60 - 20℃; | 99% |
-
-
99-56-9
4-Nitrophenylene-1,2-diamine

-
-
99-04-7
m-Toluic acid

-
-
1571-91-1
5-nitro-2-(m-tolyl)-1H-benzo[d]imidazole

| Conditions | Yield |
|---|---|
| With polyphosphoric acid (PPA) at 120 - 150℃; for 5h; | 99% |
| With polyphosphoric acid at 120 - 150℃; for 5h; | 99% |

| Conditions | Yield |
|---|---|
| With manganese; 1,3-bis[(diphenylphosphino)propane]dichloronickel(II); 1,3-bis-(diphenylphosphino)propane; 2,2-dimethylpropanoic anhydride; zinc In toluene at 160℃; for 24h; Sealed tube; Inert atmosphere; | 99% |
-
-
30433-91-1
[2-(2-thyenyl)ethyl]amine

-
-
99-04-7
m-Toluic acid

| Conditions | Yield |
|---|---|
| With benzotriazol-1-yloxyl-tris-(pyrrolidino)-phosphonium hexafluorophosphate; N-ethyl-N,N-diisopropylamine In N,N-dimethyl-formamide at 20℃; | 98% |

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 20℃; for 1h; Inert atmosphere; regioselective reaction; | 98% |
-
-
941-55-9
4-toluenesulfonyl azide

-
-
99-04-7
m-Toluic acid

-
-
152126-61-9
N-<(4-Methylphenyl)sulfonyl>-3-methylbenzenecarboxamide

| Conditions | Yield |
|---|---|
| With dicobalt octacarbonyl; tert-butylisonitrile In acetonitrile at 80℃; for 4h; | 98% |
| With copper(l) iodide; methyl ethynyl ketone In acetonitrile at 80℃; for 4h; | 72% |

| Conditions | Yield |
|---|---|
| With dmap; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride; N-ethyl-N,N-diisopropylamine In dichloromethane at 20℃; | 98% |

| Conditions | Yield |
|---|---|
| With dmap; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride; N-ethyl-N,N-diisopropylamine In dichloromethane at 20℃; Cooling; | 98% |
-
-
616-38-6
carbonic acid dimethyl ester

-
-
99-04-7
m-Toluic acid

-
-
99-36-5
1-methoxycarbonyl-3-methylbenzene

| Conditions | Yield |
|---|---|
| With sulfuric acid; acetic acid at 85 - 90℃; for 12h; Neat (no solvent); Large scale reaction; | 97.65% |

| Conditions | Yield |
|---|---|
| With triethylamine at 20℃; for 0.333333h; | 97.5% |
| With triethylamine at 20℃; for 0.333333h; | 97.5% |

| Conditions | Yield |
|---|---|
| Stage #1: m-Toluic acid In dichloromethane at 0℃; for 0.25h; Stage #2: 1,2-diamino-benzene With pyridine In dichloromethane at 20℃; for 6h; Further stages.; | 97% |
| With polyphosphoric acid In toluene at 165℃; for 6h; | 89% |
| With propylene glycol at 170℃; for 18h; | 78.5% |
-
-
501-65-5
diphenyl acetylene

-
-
99-04-7
m-Toluic acid

-
-
93743-65-8
7-methyl-3,4-diphenyl-1H-2-benzopyran-1-one

| Conditions | Yield |
|---|---|
| With dichloro[1,3-di(ethoxycarbonyl)-2,4,5-trimethylcyclopentadienyl]rhodium(III) dimer; copper(II) acetate monohydrate; silver(I) triflimide In dichloromethane at 20℃; for 24h; Sealed tube; | 97% |
| With carbonyl(pentamethylcyclopentadienyl)cobalt diiodide; sodium acetate; copper(II) oxide In 2,2,2-trifluoroethanol at 80℃; for 24h; Schlenk technique; Molecular sieve; Inert atmosphere; | 87% |
| With silver hexafluoroantimonate; [ruthenium(II)(η6-1-methyl-4-isopropyl-benzene)(chloride)(μ-chloride)]2; copper(II) acetate monohydrate at 80℃; for 18h; Green chemistry; regioselective reaction; | 80% |

| Conditions | Yield |
|---|---|
| With 1,1'-carbonyldiimidazole In tetrahydrofuran at 20℃; | 97% |
| Stage #1: m-Toluic acid With oxalyl dichloride; N,N-dimethyl-formamide In dichloromethane at 0 - 20℃; for 4h; Under an atmosphere of argon; Stage #2: N-methoxylamine hydrochloride With potassium carbonate In water; ethyl acetate at 0 - 20℃; for 4h; | 96% |
-
-
95-14-7
1,2,3-Benzotriazole

-
-
99-04-7
m-Toluic acid

-
-
313549-89-2
(1H-benzo-[d][1,2,3]triazol-1-yl)(m-tolyl)methanone

| Conditions | Yield |
|---|---|
| Stage #1: m-Toluic acid With iodine; triphenylphosphine In dichloromethane at 0℃; for 0.5h; Stage #2: 1,2,3-Benzotriazole In dichloromethane at 0℃; for 0.166667h; Stage #3: With triethylamine In dichloromethane at 20℃; for 0.5h; | 97% |
| With thionyl chloride In dichloromethane for 2h; Inert atmosphere; | 95% |
| With thionyl chloride In dichloromethane at 20℃; | 84% |
-
-
68820-12-2
cyclohexyl-N-tosyl aziridine

-
-
99-04-7
m-Toluic acid

| Conditions | Yield |
|---|---|
| With potassium hydroxide In dimethyl sulfoxide at 45℃; for 2h; | 97% |
M-toluic acid Chemical Properties
IUPAC Name: 3-Methylbenzoic acid
Synonyms: 3-Methylbenzoic acid ; Benzoic acid, 3-methyl- ; 3-Methyl-Benzoic Acid ; 3-Toluic acid
Product Categories: Aromatic Carboxylic Acids, Amides, Anilides, Anhydrides & Salts;Organic acids
Molecular Structure of M-toluic acid (CAS NO.99-04-7) : 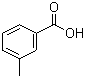
Molecular Formula of M-toluic acid (CAS NO.99-04-7) : C8H8O2
Molecular Weight of M-toluic acid (CAS NO.99-04-7) : 136.15
CAS NO: 99-04-7
EINECS: 202-723-9
Index of Refraction: 1.556
Surface Tension: 45.4 dyne/cm
Density: 1.151 g/cm3
Flash Point: 120 °C
Enthalpy of Vaporization: 53 kJ/mol
Boiling Point: 263.8 °C at 760 mmHg
Vapour Pressure: 0.00506 mmHg at 25°C
Melting point: 108 °C
Water Solubility : <0.1 g/100 mL at 19 ºC
Stability: Stable. Combustible. Incompatible with strong oxidizing agents
General Description :White to yellowish crystals or mostly yellow flaky solid (with some white flakes). Has a floral-honey odor.
M-toluic acid Uses
It serves, among other purposes, as a precursor to DEET (N,N diethyl-m-toluamide) the well-known insect repellent.
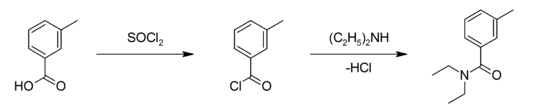
M-toluic acid Production
Preparation Products: 3-Carboxybenzaldehyde-->3-(4-Methylpiperazin-1-ylmethyl)benzoic acid-->Methyl 3-(morpholinomethyl)benzoate ,98%-->METHYL 3-((PYRROLIDIN-1-YL)METHYL)BENZOATE-->3-(BROMOMETHYL)BENZOIC ACID-->3-BENZOYLPHENYLACETIC ACID-->3-Methylbenzophenone-->N,N-Diethyl-3-methylbenzamide
Raw materials :m-Xylene-->1,3-Bisbenzyl-2-oxoimidazolidine-4,5-dicarboxylic acid
M-toluic acid (CAS NO.99-04-7) is derived from m-Xylene catalytic oxidation. cobalt naphthenate as a catalyst , m-Xylene oxidation by using air , the reaction temperature is 125-135 °C, pressure is about 0.25MPa. Per ton of product consumption of 1700kg m-Xylene.
M-toluic acid Toxicity Data With Reference
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| mouse | LD50 | intraperitoneal | 562mg/kg (562mg/kg) | BEHAVIORAL: REGIDITY BEHAVIORAL: MUSCLE CONTRACTION OR SPASTICITY) | Journal of Medicinal Chemistry. Vol. 11, Pg. 1020, 1968. |
| mouse | LD50 | oral | 1630mg/kg (1630mg/kg) | BEHAVIORAL: MUSCLE WEAKNESS LUNGS, THORAX, OR RESPIRATION: RESPIRATORY DEPRESSION GASTROINTESTINAL: NAUSEA OR VOMITING | Gigiena Truda i Professional'nye Zabolevaniya. Labor Hygiene and Occupational Diseases. Vol. 18(7), Pg. 57, 1974. |
| rat | LD | oral | > 5gm/kg (5000mg/kg) | BEHAVIORAL: EXCITEMENT GASTROINTESTINAL: NAUSEA OR VOMITING | Gigiena Truda i Professional'nye Zabolevaniya. Labor Hygiene and Occupational Diseases. Vol. 18(7), Pg. 57, 1974. |
M-toluic acid Safety Profile
Hazard Codes  Xn
Xn
Risk Statements 22
R22:Harmful if swallowed.
Safety Statements 22-24/25-36
S22:Do not breathe dust.
S24/25:Avoid contact with skin and eyes.
S36:Wear suitable protective clothing.
WGK Germany 3
RTECS XU1200000
HS Code 29163900
M-toluic acid Specification
1.Air & Water Reactions :Fine dust dispensed in air in sufficient concentrations, and in the presence of an ignition source is a potential dust explosion hazard. . Insoluble in water.
Reactivity Profile m-Toluic acid is a carboxylic acid. Carboxylic acids donate hydrogen ions if a base is present to accept them. They react in this way with all bases, both organic (for example, the amines) and inorganic. Their reactions with bases, called "neutralizations", are accompanied by the evolution of substantial amounts of heat. Neutralization between an acid and a base produces water plus a salt. Carboxylic acids with six or fewer carbon atoms are freely or moderately soluble in water; those with more than six carbons are slightly soluble in water. Soluble carboxylic acid dissociate to an extent in water to yield hydrogen ions. The pH of solutions of carboxylic acids is therefore less than 7.0. Many insoluble carboxylic acids react rapidly with aqueous solutions containing a chemical base and dissolve as the neutralization generates a soluble salt. Carboxylic acids in aqueous solution and liquid or molten carboxylic acids can react with active metals to form gaseous hydrogen and a metal salt. Such reactions occur in principle for solid carboxylic acids as well, but are slow if the solid acid remains dry. Even "insoluble" carboxylic acids may absorb enough water from the air and dissolve sufficiently in m-Toluic acid to corrode or dissolve iron, steel, and aluminum parts and containers. Carboxylic acids, like other acids, react with cyanide salts to generate gaseous hydrogen cyanide. The reaction is slower for dry, solid carboxylic acids. Insoluble carboxylic acids react with solutions of cyanides to cause the release of gaseous hydrogen cyanide. Flammable and/or toxic gases and heat are generated by the reaction of carboxylic acids with diazo compounds, dithiocarbamates, isocyanates, mercaptans, nitrides, and sulfides. Carboxylic acids, especially in aqueous solution, also react with sulfites, nitrites, thiosulfates (to give H2S and SO3), dithionites (SO2), to generate flammable and/or toxic gases and heat. Their reaction with carbonates and bicarbonates generates a harmless gas (carbon dioxide) but still heat. Like other organic compounds, carboxylic acids can be oxidized by strong oxidizing agents and reduced by strong reducing agents. These reactions generate heat. A wide variety of products is possible. Like other acids, carboxylic acids may initiate polymerization reactions; like other acids, they often catalyze (increase the rate of) chemical reactions. m-Toluic acid is incompatible with strong oxidizers.
2.Fire Hazard Flash point data for m-Toluic acid are not available; however, m-Toluic acid is probably combustible.
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View