-
Name
Methallyl alcohol
- EINECS 208-161-0
- CAS No. 513-42-8
- Article Data95
- CAS DataBase
- Density 0.829 g/cm3
- Solubility 11 g/100 mL in water
- Melting Point -50 °C
- Formula C4H8O
- Boiling Point 116.3 °C at 760 mmHg
- Molecular Weight 72.1069
- Flash Point 34.7 °C
- Transport Information UN 2614 3/PG 3
- Appearance clear colourless liquid
- Safety 26-36-37/39-16
- Risk Codes 10-36/37/38
-
Molecular Structure
-
Hazard Symbols
 Xi
Xi
- Synonyms 2-Methallylalcohol;2-Methyl-2-propen-1-ol;2-Methyl-2-propenol;2-Methyl-3-hydroxy-1-propene;2-Methylallyl alcohol;2-Methylprop-1-en-3-ol;3-Hydroxy-2-methylpropene;Isopropenyl carbinol;Methallylalcohol;NSC 30674;NSC 404204;b-Methallyl alcohol;b-Methylallyl alcohol;
- PSA 20.23000
- LogP 0.55480
Synthetic route

| Conditions | Yield |
|---|---|
| With sodium hydrogencarbonate; acrylonitrile; hydroquinone In water at 40 - 67℃; for 5.5h; Reagent/catalyst; Temperature; | 99.05% |
| With t-butylnitrite; water; sodium hydrogencarbonate; acrylonitrile at 45 - 67℃; for 3h; Catalytic behavior; Reagent/catalyst; | 99.05% |
| With 5,5-Dimethyl-1-pyrroline N-oxide; phenyl tert-butylmethyldimethoxysilane; water; sodium acetate; sodium carbonate; acetonitrile at 45 - 67℃; for 2.5h; Catalytic behavior; Reagent/catalyst; | 99.02% |
-
-
78-85-3
2-methylpropenal

-
-
108-93-0
cyclohexanol

-
A
-
108-94-1
cyclohexanone

-
B
-
513-42-8
3-hydroxy-2-methyl-1-propene

| Conditions | Yield |
|---|---|
| With TEMPOL; SnO2#Nb2O5 at 90℃; under 3000.3 Torr; for 10h; Inert atmosphere; | A 95.33% B n/a |

-
-
820-57-5
methallyl formate

-
A
-
77287-34-4, 77287-35-5, 60100-09-6
formamide

-
B
-
513-42-8
3-hydroxy-2-methyl-1-propene

| Conditions | Yield |
|---|---|
| With aluminum oxide; ammonia at 0 - 5℃; Gas phase; Large scale; | A 850 g B 95% |

| Conditions | Yield |
|---|---|
| With hydrogen In water at 70℃; under 11251.1 Torr; for 1.5h; Temperature; Reagent/catalyst; Pressure; | 94% |
| With hydrogen In water at 70℃; under 3000.3 Torr; Temperature; Reagent/catalyst; Pressure; | 89% |
| With sodium tetrahydroborate; pyrographite In tetrahydrofuran; water at 20℃; for 0.0166667h; | 85% |
-
-
127-09-3
sodium acetate

-
-
563-47-3
3-Chloro-2-methylpropene

-
A
-
820-71-3
methallyl acetate

-
B
-
513-42-8
3-hydroxy-2-methyl-1-propene

| Conditions | Yield |
|---|---|
| With sodium D-gluconate; copper(II) sulfate In water for 4h; Reagent/catalyst; Reflux; | A 92% B 5.5% |

-
-
558-30-5
2-methyl-1,2-epoxypropane

-
A
-
78-84-2
isobutyraldehyde

-
B
-
513-42-8
3-hydroxy-2-methyl-1-propene

| Conditions | Yield |
|---|---|
| lithium phosphate at 210℃; for 3h; Kinetics; Thermodynamic data; 110 - 180 degC , ΔG, ΔH, E(activ.); | A 5.79% B 85.1% |
| With hydrogen; lithium phosphate at 210℃; for 3h; | A 5.79% B 85.1% |
| With 3-chlorobenzoate; methanesulfonic acid In dichloromethane-d2 at 26.5℃; Product distribution; various conc. of methanesulfonic acid in the presence and absence of 3-chlorobenzoic acid; | A 10% B 20% |
| With 3-chlorobenzoate; methanesulfonic acid In dichloromethane-d2 at 26.5℃; | A 10% B 20% |

| Conditions | Yield |
|---|---|
| With lithium aluminium tetrahydride In diethyl ether at -30℃; for 4h; Reduction; | 83% |
| With lithium aluminium tetrahydride In diethyl ether at 0℃; for 1h; | 72% |

-
-
513-42-8
3-hydroxy-2-methyl-1-propene

| Conditions | Yield |
|---|---|
| With tellurium; lithium triethylborohydride In tetrahydrofuran for 0.166667h; Ambient temperature; | 70% |

| Conditions | Yield |
|---|---|
| With ZnO/MgZrOx mixed oxide with 4.3 Mg/Zr atomic ratio on MgO nanaosheet at 275℃; Inert atmosphere; | 17% |
-
-
54004-34-1
2,5-dimethyl-3,4-dihydro-2H-pyran-2-methanol

-
-
513-42-8
3-hydroxy-2-methyl-1-propene

| Conditions | Yield |
|---|---|
| With aluminum oxide at 500℃; |

| Conditions | Yield |
|---|---|
| With potassium carbonate |

| Conditions | Yield |
|---|---|
| With aluminum oxide; copper(II) sulfate at 225℃; under 760 Torr; |

| Conditions | Yield |
|---|---|
| With lithium aluminium tetrahydride; diethyl ether | |
| With diisobutylaluminium hydride In hexane; dichloromethane at -78℃; for 1.5h; Inert atmosphere; |
-
-
78-85-3
2-methylpropenal

-
A
-
78-83-1
2-methyl-propan-1-ol

-
B
-
78-84-2
isobutyraldehyde

-
C
-
67-64-1
acetone

-
D
-
513-42-8
3-hydroxy-2-methyl-1-propene

| Conditions | Yield |
|---|---|
| With magnesium oxide; isopropyl alcohol; boron trioxide at 270℃; other α,β-unsaturated corbonyl compounds, other alcohols; |

-
-
59085-70-0
2-methyl-3-prop-2-enyl phenylselenide

-
-
513-42-8
3-hydroxy-2-methyl-1-propene

| Conditions | Yield |
|---|---|
| With dihydrogen peroxide In pyridine; dichloromethane at 0℃; for 0.333333h; Yield given; |
-
-
75-16-1
methylmagnesium bromide

-
-
107-19-7
propargyl alcohol

-
A
-
89732-64-9
(Z)-(3-<2H>)-2-methylprop-2-en-1-ol

-
B
-
513-42-8
3-hydroxy-2-methyl-1-propene

| Conditions | Yield |
|---|---|
| With hydrogenchloride; copper(l) iodide; water-d2 1.) THF, -60 deg C up to RT, 2.) THF, Et2O, H2O, pH=7; Yield given. Multistep reaction. Yields of byproduct given. Title compound not separated from byproducts; |

-
-
78-85-3
2-methylpropenal

-
A
-
78-83-1
2-methyl-propan-1-ol

-
B
-
78-84-2
isobutyraldehyde

-
C
-
513-42-8
3-hydroxy-2-methyl-1-propene

| Conditions | Yield |
|---|---|
| With hydrogen; AuZr(O) at 89.9℃; Product distribution; pulse reactor; other temperatures; | A 6.7 % Turnov. B 42.9 % Turnov. C 47.2 % Turnov. |

-
-
598-09-4
2-chloromethyl-2-methyloxiran

-
A
-
29526-99-6
1-methylcyclopropanol

-
B
-
513-42-8
3-hydroxy-2-methyl-1-propene

| Conditions | Yield |
|---|---|
| With magnesium In tetrahydrofuran Heating; Title compound not separated from byproducts; |
-
-
53250-10-5
2-(2-methylallyloxy)tetrahydro-2H-pyran

-
-
513-42-8
3-hydroxy-2-methyl-1-propene

| Conditions | Yield |
|---|---|
| zeolite HSZ-330 In methanol for 2h; Ambient temperature; |
-
-
115-11-7
isobutene

-
A
-
563-80-4
3-methyl-butan-2-one

-
B
-
67-64-1
acetone

-
C
-
513-42-8
3-hydroxy-2-methyl-1-propene

| Conditions | Yield |
|---|---|
| at 200 - 240℃; under 58840.6 Torr; |


-
-
513-42-8
3-hydroxy-2-methyl-1-propene

| Conditions | Yield |
|---|---|
| With potassium hydroxide | |
| With water; potassium carbonate man destilliert die erhaltene Loesung und Schuettelt das Destillat mit K2CO3, wobei das Isopropenylcarbinol abgeschieden wird, das man ueber CaO entwaessert; |
-
-
78-85-3
2-methylpropenal

-
-
64-19-7
acetic acid

-
A
-
67470-03-5
meso-2,5-dimethyl-hexa-1,5-diene-3,4-diol

-
B
-
513-42-8
3-hydroxy-2-methyl-1-propene

-
-
78-85-3
2-methylpropenal

-
-
557-20-0
diethylzinc

-
A
-
125637-07-2
(R)-3-hydroxy-2-methylpent-1-ene

-
B
-
98168-21-9
(+)-(R)-2-methylpent-1-en-3-ol

-
C
-
513-42-8
3-hydroxy-2-methyl-1-propene

| Conditions | Yield |
|---|---|
| With (1R,2S)-(+)-1-cyclohexyl-2-phenyl-2-(N-morpholino)ethanol In hexane; toluene at 20℃; for 3h; Title compound not separated from byproducts.; |


| Conditions | Yield |
|---|---|
| at 260.4℃; under 49.5 Torr; Kinetics; Further Variations:; Pressures; Temperatures; also in the presence of free radical inhibitors; |

| Conditions | Yield |
|---|---|
| In methanol; potassium hydroxide |
-
-
1396267-65-4
3-[(ethoxycarbonyl)oxy]-2-methylpropyl propanoate

-
A
-
802294-64-0
propionic acid

-
B
-
513-42-8
3-hydroxy-2-methyl-1-propene

| Conditions | Yield |
|---|---|
| at 626.84℃; Kinetics; Further Variations:; Temperatures; |
-
-
1073930-96-7
2-methylallyl 1-naphthyl selenide

-
A
-
78-85-3
2-methylpropenal

-
B
-
1787-80-0
2-bis[3,5bis(trifluoromethyl)phenyl]diselane

-
C
-
513-42-8
3-hydroxy-2-methyl-1-propene

| Conditions | Yield |
|---|---|
| With oxygen In n-heptane at 20℃; for 0.25h; Reactivity; Time; UV-irradiation; |

-
-
78-39-7
Triethyl orthoacetate

-
-
513-42-8
3-hydroxy-2-methyl-1-propene

-
-
4911-54-0
ethyl 4-methyl-4-pentenoate

| Conditions | Yield |
|---|---|
| With propionic acid for 8h; Reflux; | 100% |
| With propionic acid at 118℃; for 6h; Johnson orthoester Claisen rearrangement; | 98% |
| With acetic acid at 150 - 170℃; Inert atmosphere; | 87% |
-
-
124-63-0
methanesulfonyl chloride

-
-
513-42-8
3-hydroxy-2-methyl-1-propene

-
-
142820-20-0
2-methylallyl methanesulfonate

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane at -15℃; for 1h; Green chemistry; | 100% |
| With triethylamine In tetrahydrofuran for 0.25h; Ambient temperature; | |
| With triethylamine In dichloromethane at -20℃; for 1.5h; | |
| With triethylamine In diethyl ether at -78 - 0℃; Inert atmosphere; |

| Conditions | Yield |
|---|---|
| With tetrabutylphosphonium peroxotantalate; dihydrogen peroxide In water at 0℃; for 3.5h; Schlenk technique; chemoselective reaction; | 100% |
| With C16H36N(1+)*H2NbO5(1-); dihydrogen peroxide In water at 0℃; for 4h; chemoselective reaction; | 96.3% |
| With 3-chloro-benzenecarboperoxoic acid In dichloromethane at 0 - 20℃; for 18h; | 83% |
-
-
104-88-1
4-chlorobenzaldehyde

-
-
513-42-8
3-hydroxy-2-methyl-1-propene

-
A
-
106536-22-5
3,3-bis(3-phenyl-2-propen-1-yl)pentane-2,4-dione

-
B
-
106536-23-6
(E)-1-chloro-4-(3-methylbuta-1,3-dien-1-yl)benzene

| Conditions | Yield |
|---|---|
| With triphenylphosphine; palladium(II) acetylacetonate In 1,4-dioxane for 50h; Heating; | A 100% B 21% C n/a |

-
-
824-94-2
p-methoxybenzyl chloride

-
-
513-42-8
3-hydroxy-2-methyl-1-propene

-
-
189366-67-4
1-((methallyloxy)methyl)-4-methoxybenzene

| Conditions | Yield |
|---|---|
| With dmap; triethylamine In dichloromethane at 20℃; | 100% |
| With sodium hydroxide 1.) THF, mineral oil, DMF, 0 deg C, 30 min; 2.) THF, 2 h; | 97% |
| With sodium hydride In tetrahydrofuran; N,N-dimethyl-formamide at 0℃; 1.) 30 min, 2.) 2 h; |

| Conditions | Yield |
|---|---|
| With dimesitylammonium pentafluorobenzenesulfonate In n-heptane at 80℃; for 3h; | 100% |
| With Iron(III) isopropoxide; zirconium(IV) tetraisopropoxide In n-heptane for 15h; | 99% |
| dimesitylammonium pentafluorobenzenesulfonate In n-heptane at 80℃; for 3h; Product distribution / selectivity; | 99% |
-
-
1049038-08-5
[(4-methoxyphenyl)sulfonyl]azanyl sulfamate

-
-
513-42-8
3-hydroxy-2-methyl-1-propene

-
-
1049038-15-4
C11H16N2O6S2

| Conditions | Yield |
|---|---|
| With triphenylphosphine; diethylazodicarboxylate In tetrahydrofuran at 25℃; Mitsunobu reaction; Cooling with ice; | 100% |

| Conditions | Yield |
|---|---|
| With triphenylphosphine; diethylazodicarboxylate In tetrahydrofuran at 20℃; Mitsunobu reaction; Cooling with ice; | 100% |

| Conditions | Yield |
|---|---|
| With sodium hydride In tetrahydrofuran at 0 - 20℃; for 20h; | 100% |

| Conditions | Yield |
|---|---|
| Stage #1: N-(p-toluenesulfonyl)aziridine; 3-hydroxy-2-methyl-1-propene In dichloromethane at 0℃; for 0.166667h; Stage #2: With Amberlyst-15 In dichloromethane at 0 - 20℃; for 4h; | 100% |
-
-
545-06-2
trichloroacetonitrile

-
-
513-42-8
3-hydroxy-2-methyl-1-propene

-
-
84820-25-7
2-methyl-2-propenyl trichloroacetimidate

| Conditions | Yield |
|---|---|
| With 1,8-diazabicyclo[5.4.0]undec-7-ene In dichloromethane at 0℃; for 22h; Inert atmosphere; | 99% |
| (i) Na, (ii) /BRN= 605572/; Multistep reaction; | |
| With sodium hydride In tetrahydrofuran at 0℃; for 2h; | |
| With sodium hydride In diethyl ether at 0℃; Esterification; |

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane at 0 - 20℃; | 99% |
| With triethylamine In tetrahydrofuran at 0 - 20℃; for 26h; | 72% |
| With pyridine In dichloromethane at 0 - 23℃; for 2.25h; Inert atmosphere; Autoclave; | 70% |

| Conditions | Yield |
|---|---|
| With sodium hydrogencarbonate; acrylonitrile; hydroquinone In water at 40 - 67℃; for 5.5h; Reagent/catalyst; Temperature; | 99.05% |
| With t-butylnitrite; water; sodium hydrogencarbonate; acrylonitrile at 45 - 67℃; for 3h; Catalytic behavior; Reagent/catalyst; | 99.05% |
| With 5,5-Dimethyl-1-pyrroline N-oxide; phenyl tert-butylmethyldimethoxysilane; water; sodium acetate; sodium carbonate; acetonitrile at 45 - 67℃; for 2.5h; Catalytic behavior; Reagent/catalyst; | 99.02% |
-
-
78-85-3
2-methylpropenal

-
-
108-93-0
cyclohexanol

-
A
-
108-94-1
cyclohexanone

-
B
-
513-42-8
3-hydroxy-2-methyl-1-propene

| Conditions | Yield |
|---|---|
| With TEMPOL; SnO2#Nb2O5 at 90℃; under 3000.3 Torr; for 10h; Inert atmosphere; | A 95.33% B n/a |

-
-
820-57-5
methallyl formate

-
A
-
77287-34-4, 77287-35-5, 60100-09-6
formamide

-
B
-
513-42-8
3-hydroxy-2-methyl-1-propene

| Conditions | Yield |
|---|---|
| With aluminum oxide; ammonia at 0 - 5℃; Gas phase; Large scale; | A 850 g B 95% |

| Conditions | Yield |
|---|---|
| With hydrogen In water at 70℃; under 11251.1 Torr; for 1.5h; Temperature; Reagent/catalyst; Pressure; | 94% |
| With hydrogen In water at 70℃; under 3000.3 Torr; Temperature; Reagent/catalyst; Pressure; | 89% |
| With sodium tetrahydroborate; pyrographite In tetrahydrofuran; water at 20℃; for 0.0166667h; | 85% |
-
-
127-09-3
sodium acetate

-
-
563-47-3
3-Chloro-2-methylpropene

-
A
-
820-71-3
methallyl acetate

-
B
-
513-42-8
3-hydroxy-2-methyl-1-propene

| Conditions | Yield |
|---|---|
| With sodium D-gluconate; copper(II) sulfate In water for 4h; Reagent/catalyst; Reflux; | A 92% B 5.5% |

-
-
558-30-5
2-methyl-1,2-epoxypropane

-
A
-
78-84-2
isobutyraldehyde

-
B
-
513-42-8
3-hydroxy-2-methyl-1-propene

| Conditions | Yield |
|---|---|
| lithium phosphate at 210℃; for 3h; Kinetics; Thermodynamic data; 110 - 180 degC , ΔG, ΔH, E(activ.); | A 5.79% B 85.1% |
| With hydrogen; lithium phosphate at 210℃; for 3h; | A 5.79% B 85.1% |
| With 3-chlorobenzoate; methanesulfonic acid In dichloromethane-d2 at 26.5℃; Product distribution; various conc. of methanesulfonic acid in the presence and absence of 3-chlorobenzoic acid; | A 10% B 20% |
| With 3-chlorobenzoate; methanesulfonic acid In dichloromethane-d2 at 26.5℃; | A 10% B 20% |

| Conditions | Yield |
|---|---|
| With lithium aluminium tetrahydride In diethyl ether at -30℃; for 4h; Reduction; | 83% |
| With lithium aluminium tetrahydride In diethyl ether at 0℃; for 1h; | 72% |

-
-
513-42-8
3-hydroxy-2-methyl-1-propene

| Conditions | Yield |
|---|---|
| With tellurium; lithium triethylborohydride In tetrahydrofuran for 0.166667h; Ambient temperature; | 70% |

| Conditions | Yield |
|---|---|
| With ZnO/MgZrOx mixed oxide with 4.3 Mg/Zr atomic ratio on MgO nanaosheet at 275℃; Inert atmosphere; | 17% |
-
-
54004-34-1
2,5-dimethyl-3,4-dihydro-2H-pyran-2-methanol

-
-
513-42-8
3-hydroxy-2-methyl-1-propene

| Conditions | Yield |
|---|---|
| With aluminum oxide at 500℃; |

| Conditions | Yield |
|---|---|
| With potassium carbonate |

| Conditions | Yield |
|---|---|
| With aluminum oxide; copper(II) sulfate at 225℃; under 760 Torr; |

| Conditions | Yield |
|---|---|
| With lithium aluminium tetrahydride; diethyl ether | |
| With diisobutylaluminium hydride In hexane; dichloromethane at -78℃; for 1.5h; Inert atmosphere; |
-
-
78-85-3
2-methylpropenal

-
A
-
78-83-1
2-methyl-propan-1-ol

-
B
-
78-84-2
isobutyraldehyde

-
C
-
67-64-1
acetone

-
D
-
513-42-8
3-hydroxy-2-methyl-1-propene

| Conditions | Yield |
|---|---|
| With magnesium oxide; isopropyl alcohol; boron trioxide at 270℃; other α,β-unsaturated corbonyl compounds, other alcohols; |

-
-
59085-70-0
2-methyl-3-prop-2-enyl phenylselenide

-
-
513-42-8
3-hydroxy-2-methyl-1-propene

| Conditions | Yield |
|---|---|
| With dihydrogen peroxide In pyridine; dichloromethane at 0℃; for 0.333333h; Yield given; |
-
-
75-16-1
methylmagnesium bromide

-
-
107-19-7
propargyl alcohol

-
A
-
89732-64-9
(Z)-(3-<2H>)-2-methylprop-2-en-1-ol

-
B
-
513-42-8
3-hydroxy-2-methyl-1-propene

| Conditions | Yield |
|---|---|
| With hydrogenchloride; copper(l) iodide; water-d2 1.) THF, -60 deg C up to RT, 2.) THF, Et2O, H2O, pH=7; Yield given. Multistep reaction. Yields of byproduct given. Title compound not separated from byproducts; |

-
-
78-85-3
2-methylpropenal

-
A
-
78-83-1
2-methyl-propan-1-ol

-
B
-
78-84-2
isobutyraldehyde

-
C
-
513-42-8
3-hydroxy-2-methyl-1-propene

| Conditions | Yield |
|---|---|
| With hydrogen; AuZr(O) at 89.9℃; Product distribution; pulse reactor; other temperatures; | A 6.7 % Turnov. B 42.9 % Turnov. C 47.2 % Turnov. |

-
-
598-09-4
2-chloromethyl-2-methyloxiran

-
A
-
29526-99-6
1-methylcyclopropanol

-
B
-
513-42-8
3-hydroxy-2-methyl-1-propene

| Conditions | Yield |
|---|---|
| With magnesium In tetrahydrofuran Heating; Title compound not separated from byproducts; |
-
-
53250-10-5
2-(2-methylallyloxy)tetrahydro-2H-pyran

-
-
513-42-8
3-hydroxy-2-methyl-1-propene

| Conditions | Yield |
|---|---|
| zeolite HSZ-330 In methanol for 2h; Ambient temperature; |
-
-
115-11-7
isobutene

-
A
-
563-80-4
3-methyl-butan-2-one

-
B
-
67-64-1
acetone

-
C
-
513-42-8
3-hydroxy-2-methyl-1-propene

| Conditions | Yield |
|---|---|
| at 200 - 240℃; under 58840.6 Torr; |


-
-
513-42-8
3-hydroxy-2-methyl-1-propene

| Conditions | Yield |
|---|---|
| With potassium hydroxide | |
| With water; potassium carbonate man destilliert die erhaltene Loesung und Schuettelt das Destillat mit K2CO3, wobei das Isopropenylcarbinol abgeschieden wird, das man ueber CaO entwaessert; |
Methallyl alcohol Chemical Properties
The Molecular Weight of ISOPROPENYL CARBINOL(513-42-8): 72.11
Molecular Structure :
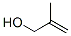
EINECS: 208-161-0
Melting point: -50 °C
Boiling Point: 116.3 °C at 760 mmHg
Flash Point: 34.7 °C
Index of Refraction: 1.412
Molar Refractivity: 21.68 cm3
Molar Volume: 86.9 cm3
Polarizability: 8.59 10-24 cm3
Surface Tension: 24.3 dyne/cm
Density: 0.829 g/cm3
Enthalpy of Vaporization: 41.34 kJ/mol
Vapour Pressure: 9.15 mmHg at 25°C
storage temp.: Flammables area
Water Solubility: 11 g/100 mL
refractive index: n20/D 1.426(lit.)
IUPAC Name: 2-methylprop-2-en-1-ol
Synonyms: 2-methyl-2-propen-1-o;3-Hydroxy-2-methylpropene;METHALLYL ALCOHOL;ISOPROPENYLCARBINOL;beta-methylallylalcohol;CH2=C(CH3)CH2OH;methacrylalcohol;
Methallyl alcohol Uses
Methallyl alcohol Toxicity Data With Reference
| 1. | skn-rbt 500 mg MOD | SCCUR* Shell Chemical Company. Unpublished Report. (2401 Crow Canyon Rd., San Romon, CA 94583) | ||
| 2. | orl-mus LDLo:500 mg/kg | SCCUR* Shell Chemical Company. Unpublished Report. (2401 Crow Canyon Rd., San Romon, CA 94583) | ||
| 3. | ihl-mus LCLo:2924 ppm/2H | SCCUR* Shell Chemical Company. Unpublished Report. (2401 Crow Canyon Rd., San Romon, CA 94583) | ||
| 4. | skn-rbt LDLo:2000 mg/kg | SCCUR* Shell Chemical Company. Unpublished Report. (2401 Crow Canyon Rd., San Romon, CA 94583) |
Methallyl alcohol Consensus Reports
Methallyl alcohol Safety Profile
The Hazard Codes of ISOPROPENYL CARBINOL(513-42-8):
 Xi
Xi HazardClass 3.2
The Risk Statements information of ISOPROPENYL CARBINOL(513-42-8):
10: Flammable
36/37/38: Irritating to eyes, respiratory system and skin
The Safety Statements information of ISOPROPENYL CARBINOL(513-42-8):
16: Keep away from sources of ignition - No smoking
26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice
36: Wear suitable protective clothing
37/39: Wear suitable gloves and eye/face protection
RIDADR: UN 2614 3/PG 3
WGK Germany: 3
RTECS: UD5250000
PackingGroup: III
Methallyl alcohol Standards and Recommendations
Related Products
- Methallyl acetate
- Methallyl alcohol
- Methallyl methacrylate
- Methallyl-19-nortestosterone
- 51-34-3
- 513-44-0
- 5134-62-3
- 513-48-4
- 513-49-5
- 51350-19-7
- 5135-30-8
- 513-53-1
- 51-35-4
- 51354-80-4
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View