-
Name
Hexanedioic acid,1-ethyl ester
- EINECS 210-966-7
- CAS No. 626-86-8
- Article Data38
- CAS DataBase
- Density 1.098 g/cm3
- Solubility Soluble in water.
- Melting Point 28-29 °C(lit.)
- Formula C8H14O4
- Boiling Point 285.72 °C at 760 mmHg
- Molecular Weight 174.197
- Flash Point 110.837 °C
- Transport Information
- Appearance solid
- Safety 24/25
- Risk Codes
-
Molecular Structure
- Hazard Symbols
- Synonyms Adipicacid, ethyl ester (6CI,7CI);Adipic acid, monoethyl ester (8CI);Hexanedioicacid, monoethyl ester (9CI);1,4-Butanedicarboxylic acid monoethyl ester;6-Ethoxy-6-oxohexanoic acid;Ethyl hydrogen adipate;Ethyl hydrogenhexanedioate;Monoethyl adipate;Monoethyl hexanedioate;NSC 8877;
- PSA 63.60000
- LogP 1.19450
Synthetic route

| Conditions | Yield |
|---|---|
| With acetic acid In chloroform; water at 60℃; for 8h; Reagent/catalyst; Temperature; Solvent; | 98.4% |
| With aluminum oxide at 25℃; for 48h; Time; Green chemistry; chemoselective reaction; | 28% |
| With sulfuric acid |

-
-
626-86-8
adipinic acid monoethyl ester

| Conditions | Yield |
|---|---|
| With palladium 10% on activated carbon; hydrogen In ethanol at 20℃; under 760.051 Torr; for 1.5h; Inert atmosphere; | 95% |

| Conditions | Yield |
|---|---|
| With iodosylbenzene; potassium bromide In water at 20℃; for 2h; Oxidation; | 92% |

| Conditions | Yield |
|---|---|
| With potassium bromide; poly(diacetoxyiodo)styrene In water at 80℃; for 7h; | A 92% B n/a |

| Conditions | Yield |
|---|---|
| With iron(III) perchlorate for 2h; Ambient temperature; | 90% |
-
-
542-69-8
1-iodo-butane

-
-
78437-79-3
Hexanedioic acid ethyl ester 6-(2-methoxy-ethyl)-pyridin-2-yl ester

-
A
-
626-86-8
adipinic acid monoethyl ester

-
B
-
4144-61-0
6-oxo-decanoic acid ethyl ester

| Conditions | Yield |
|---|---|
| With nickel dichloride; zinc In N,N-dimethyl-formamide at 50℃; for 5h; | A 8% B 86% |


| Conditions | Yield |
|---|---|
| Stage #1: ethyl 5-bromovalerate With chloro-trimethyl-silane; iodine; ethylene dibromide; lithium chloride; zinc In tetrahydrofuran at 50℃; Inert atmosphere; Stage #2: carbon dioxide With [Ni(PCy3)2]2(N2) In tetrahydrofuran; toluene at 0℃; under 760.051 Torr; Inert atmosphere; Stage #3: With hydrogenchloride In tetrahydrofuran; water; ethyl acetate; toluene | 86% |

| Conditions | Yield |
|---|---|
| With 4,5,6,7-tetrafluoroisobenzofuran-1,3-dione at 135℃; for 60h; | 83% |
-
-
124-04-9
Adipic acid

-
-
105-37-3
Ethyl propionate

-
A
-
626-86-8
adipinic acid monoethyl ester

-
B
-
141-28-6
diethyl adipate

| Conditions | Yield |
|---|---|
| With Dowex 50Wx2 In octane at 70℃; for 4h; Esterification; | A 72% B 7% |

-
-
14660-52-7
ethyl 5-bromovalerate

-
-
124-38-9
carbon dioxide

-
-
584-08-7
potassium carbonate

-
A
-
539-82-2
ethyl n-valerate

-
B
-
626-86-8
adipinic acid monoethyl ester

| Conditions | Yield |
|---|---|
| With nickel(II) bromide dimethoxyethane; 2.9-dimethyl-1,10-phenanthroline; diethyl 2,6-dimethyl-1,4-dihydropyridine-3,5-dicarboxylate; C60H36N2 In N,N-dimethyl-formamide at 20℃; for 24h; Molecular sieve; Irradiation; | A 25 %Spectr. B 66% |

-
-
14660-52-7
ethyl 5-bromovalerate

-
-
124-38-9
carbon dioxide

-
-
584-08-7
potassium carbonate

-
-
626-86-8
adipinic acid monoethyl ester

| Conditions | Yield |
|---|---|
| With nickel(II) bromide dimethoxyethane; 2.9-dimethyl-1,10-phenanthroline; diethyl 2,6-dimethyl-1,4-dihydropyridine-3,5-dicarboxylate; C60H36N2 In N,N-dimethyl-formamide at 20℃; for 24h; Molecular sieve; Irradiation; | 66% |

-
-
124-04-9
Adipic acid

-
-
64-17-5
ethanol

-
A
-
626-86-8
adipinic acid monoethyl ester

-
B
-
141-28-6
diethyl adipate

| Conditions | Yield |
|---|---|
| With Diethyl carbonate at 180℃; under 10343.2 Torr; for 5h; Inert atmosphere; | A 65% B 35% |
| With tetrachloromethane; sulfuric acid |


| Conditions | Yield |
|---|---|
| With potassium hydroxide In ethanol at 0℃; for 16h; | 60% |
| With potassium hydroxide at 25℃; | |
| With potassium hydroxide durch partielle Verseifung; |
-
-
4450-39-9
ethyl 5-cyanovalerate

-
A
-
652-11-9
3,4,5,6-tetrafluorophthalimide

-
B
-
626-86-8
adipinic acid monoethyl ester

| Conditions | Yield |
|---|---|
| With 4,5,6,7-tetrafluoroisobenzofuran-1,3-dione at 135℃; for 96h; | A n/a B 56% |

| Conditions | Yield |
|---|---|
| With potassium hydroxide; 18-crown-6 ether In ethanol; benzene 1.) room temperature, 2 h, 2.) reflux, 20 h; | 50% |
-
-
64-17-5
ethanol

-
-
533-60-8
cyclohexanone-2-ol

-
A
-
626-86-8
adipinic acid monoethyl ester

-
B
-
141-28-6
diethyl adipate

| Conditions | Yield |
|---|---|
| With potassium peroxomonosulfate at 20℃; for 18h; | A 25% B 49% |

-
-
64-17-5
ethanol

-
-
874-23-7
2-acetylcyclohexanone

-
A
-
626-86-8
adipinic acid monoethyl ester

-
B
-
141-28-6
diethyl adipate

| Conditions | Yield |
|---|---|
| With potassium peroxomonosulfate at 20℃; for 18h; | A 48% B 35% |

-
-
64-17-5
ethanol

-
-
765-87-7
cyclohexane-1,2-dione

-
A
-
626-86-8
adipinic acid monoethyl ester

-
B
-
141-28-6
diethyl adipate

| Conditions | Yield |
|---|---|
| With potassium peroxomonosulfate at 20℃; for 18h; | A 28% B 44% |

-
-
124-04-9
Adipic acid

-
-
64-17-5
ethanol

-
-
141-28-6
diethyl adipate

-
-
626-86-8
adipinic acid monoethyl ester

| Conditions | Yield |
|---|---|
| With hydrogenchloride; dibutyl ether |


| Conditions | Yield |
|---|---|
| With ethanol; water |
-
-
855597-17-0
O1-benzoyl-monoperoxyadipic acid-6-ethyl ester

-
-
121-69-7
N,N-dimethyl-aniline

-
A
-
366-29-0
N,N,N',N'-tetramethylbenzidine

-
B
-
626-86-8
adipinic acid monoethyl ester

-
C
-
65-85-0
benzoic acid

| Conditions | Yield |
|---|---|
| Pyrolysis; |


| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: dry ammonia gas 2: 83 percent / tetrafluorophthalic anhydride / 60 h / 135 °C View Scheme |

| Conditions | Yield |
|---|---|
| In water; benzene |
-
-
27983-42-2
5-carboethoxypentanal

-
-
626-86-8
adipinic acid monoethyl ester

| Conditions | Yield |
|---|---|
| With air at 21℃; for 2h; |
-
-
626-86-8
adipinic acid monoethyl ester

-
-
103421-61-0
3-amino-1-methyl-5-phenyl-1,3-dihydro-2H-1,4-benzodiazepin-2-one

-
-
1449305-59-2
C24H27N3O4

| Conditions | Yield |
|---|---|
| With benzotriazol-1-ol In chloroform at 20℃; for 18h; | 98% |
-
-
626-86-8
adipinic acid monoethyl ester

-
-
616-38-6
carbonic acid dimethyl ester

-
-
18891-13-9
adipic acid ethyl methyl ester

| Conditions | Yield |
|---|---|
| With potassium carbonate In dimethyl sulfoxide at 90℃; for 16h; Inert atmosphere; Green chemistry; | 98% |

| Conditions | Yield |
|---|---|
| With dmap; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride In dichloromethane at 20℃; Schlenk technique; Inert atmosphere; | 98% |

| Conditions | Yield |
|---|---|
| With thionyl chloride for 1.5h; Heating; | 96% |
| With oxalyl dichloride for 2h; Heating; | 73% |
| With thionyl chloride |

| Conditions | Yield |
|---|---|
| With N,N'-dimethylaminopyridine; di-tert-butyl dicarbonate In nitromethane at 50℃; for 16h; | 96% |

| Conditions | Yield |
|---|---|
| microwave irradiation; | 95% |
-
-
626-86-8
adipinic acid monoethyl ester

-
-
31139-36-3
dibenzyl dicarbonate

| Conditions | Yield |
|---|---|
| magnesium(II) perchlorate In nitromethane at 40℃; for 16h; | 93% |
-
-
626-86-8
adipinic acid monoethyl ester

-
-
4525-33-1
dimethyl dicarbonate

-
-
18891-13-9
adipic acid ethyl methyl ester

| Conditions | Yield |
|---|---|
| magnesium(II) perchlorate In nitromethane at 40℃; for 16h; | 91% |
-
-
7719-09-7
thionyl chloride

-
-
626-86-8
adipinic acid monoethyl ester

-
-
1071-71-2
ethyl 6-chloro-6-oxohexanoate

| Conditions | Yield |
|---|---|
| 89% |
-
-
626-86-8
adipinic acid monoethyl ester

-
-
75-08-1
ethanethiol

-
-
78647-27-5
S-ethyl O-ethyl adipic acid diester

| Conditions | Yield |
|---|---|
| With dmap; diisopropyl-carbodiimide In dichloromethane at 0 - 20℃; for 24h; Steglich Esterification; | 89% |

| Conditions | Yield |
|---|---|
| Stage #1: adipinic acid monoethyl ester; Buprenorphine With dmap; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride In dichloromethane at 20℃; Stage #2: With ethanol; acetyl chloride at 0℃; for 2h; | 89% |
-
-
626-86-8
adipinic acid monoethyl ester

-
-
98-80-6
phenylboronic acid

-
-
4248-25-3
ethyl 6-oxo-6-phenylhexanoate

| Conditions | Yield |
|---|---|
| With P(p-CH3OC6H4)3; water; dimethyl dicarbonate; palladium diacetate In tetrahydrofuran at 20℃; | 88% |
-
-
626-86-8
adipinic acid monoethyl ester

| Conditions | Yield |
|---|---|
| With benzotriazol-1-ol; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride In dichloromethane at 20℃; for 12h; | 87% |
-
-
626-86-8
adipinic acid monoethyl ester

-
-
3963-62-0
2,2-Diphenylethylamine

-
-
573986-16-0
5-(2,2-diphenyl-ethylcarbamoyl)-pentanoic acid ethyl ester

| Conditions | Yield |
|---|---|
| With dmap; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride In dichloromethane | 86% |
-
-
626-86-8
adipinic acid monoethyl ester

-
-
101403-24-1
(S)-(5-methoxy-1,2,3,4-tetrahydro-naphthalen-2-yl)-propylamine hydrochloride

-
-
1403822-89-8
(S)-ethyl 6-((5-methoxy-1,2,3,4-tetrahydronaphthalen-2-yl)(propyl)amino)-6-oxohexanoate

| Conditions | Yield |
|---|---|
| Stage #1: adipinic acid monoethyl ester With benzotriazol-1-ol; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride; triethylamine In dichloromethane at 20℃; for 1h; Inert atmosphere; Stage #2: (S)-(5-methoxy-1,2,3,4-tetrahydro-naphthalen-2-yl)-propylamine hydrochloride In dichloromethane at 20℃; for 20h; Inert atmosphere; | 86% |

| Conditions | Yield |
|---|---|
| With dicyclohexyl-carbodiimide In N,N-dimethyl-formamide for 20h; Ambient temperature; | 85% |
-
-
626-86-8
adipinic acid monoethyl ester

-
-
126-81-8
dimedone

-
-
244161-53-3
6-(4,4-dimethyl-2,6-dioxo-cyclohexylidene)-6-hydroxy-hexanoic acid ethyl ester

| Conditions | Yield |
|---|---|
| Stage #1: adipinic acid monoethyl ester With dmap; diisopropyl-carbodiimide In N,N-dimethyl-formamide for 0.416667h; Stage #2: dimedone In N,N-dimethyl-formamide for 48h; | 84% |
-
-
626-86-8
adipinic acid monoethyl ester

-
-
6638-79-5
N,O-dimethylhydroxylamine*hydrochloride

-
-
163559-10-2
ethyl 5-(N-methoxy-N-methylcarbamoyl)pentanoate

| Conditions | Yield |
|---|---|
| Stage #1: adipinic acid monoethyl ester With trichloroacetonitrile; triphenylphosphine In dichloromethane at 20℃; for 1h; Inert atmosphere; Stage #2: N,O-dimethylhydroxylamine*hydrochloride With triethylamine In dichloromethane at 20℃; for 2h; Inert atmosphere; | 83% |
| With 2-bromo-1-methyl-pyridinium iodide; triethylamine In dichloromethane for 6h; Heating; | 82% |
| With 4-methyl-morpholine; 4-(4,6-dimethoxy-1,3,5-triazin-2-yl)-4-methylmorpholinium chloride In acetonitrile at 20℃; for 1.5h; | 82% |

| Conditions | Yield |
|---|---|
| With copper(I) thiophene-2-carboxylate; (4,4'-di-tert-butyl-2,2'-dipyridyl)-bis-(5-methyl-2-(4-fluorophenyl)pyridine(-1H))-iridium(III) hexafluorophosphate; N,N,N′,N′-tetramethyl-N″-tert-butylguanidine; bathophenanthroline; iodomesitylene diacetate In 1,4-dioxane at 20℃; for 1h; Inert atmosphere; Irradiation; regioselective reaction; | 80% |
-
-
13061-96-6
dihydroxy-methyl-borane

-
-
626-86-8
adipinic acid monoethyl ester

-
-
18891-13-9
adipic acid ethyl methyl ester

| Conditions | Yield |
|---|---|
| With pyridine; copper (II) carbonate hydroxide at 90℃; for 24h; Chan-Lam Coupling; | 78% |

| Conditions | Yield |
|---|---|
| With trichlorophosphate for 0.5h; Reflux; | 77.3% |
| With trichlorophosphate at 80℃; for 2h; | 77.3% |
| With trichlorophosphate at 85℃; for 4h; | 42% |
| With trichlorophosphate at 85℃; for 4h; | 41.85% |
-
-
85825-79-2
6-methyltetrahydropyran-2,4-dione

-
-
626-86-8
adipinic acid monoethyl ester

| Conditions | Yield |
|---|---|
| With dmap; triethylamine; dicyclohexyl-carbodiimide In dichloromethane for 8h; Acylation; | 77% |
Monoethyl Adipate Chemical Properties
Empirical Formula: C8H14O4
Molecular Weight: 174.1944
EINECS: 210-966-7
Index of Refraction: 1.447
Density: 1.097 g/cm3
Flash Point: 110.8 °C
Enthalpy of Vaporization: 57.74 kJ/mol
Boiling Point: 285.7 °C at 760 mmHg
Vapour Pressure: 0.000709 mmHg at 25 °C
Melting point: 28-29 °C(lit.)
Appearance: Solid
Product categories of Adipic acid monoethyl ester (CAS NO.626-86-8): Ester series
Structure of Adipic acid monoethyl ester (CAS NO.626-86-8):
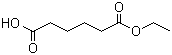
IUPAC Name: 6-Ethoxy-6-oxohexanoic acid
Canonical SMILES: CCOC(=O)CCCCC(=O)O
InChI: InChI=1S/C8H14O4/c1-2-12-8(11)6-4-3-5-7(9)10/h2-6H2,1H3,(H,9,10)
InChIKey: UZNLHJCCGYKCIL-UHFFFAOYSA-N
Monoethyl Adipate Uses
Adipic acid monoethyl ester (CAS NO.626-86-8) has been used in organic synthesis, pharmaceutical intermediates, solvents, etc..
Monoethyl Adipate Toxicity Data With Reference
| 1. | orl-uns LD50:4100 mg/kg | GTPZAB Gigiena Truda i Professionalnye Zabolevaniia. Labor Hygiene and Occupational Diseases. 21 (10)(1977),39. |
Carcinogenicity of Adipic acid monoethyl ester (CAS NO.626-86-8) hasn't been listed as a carcinogen by NTP, IARC,ACGIH, or CA Prop 65. You can see actual entry in RTECS for complete information.
Monoethyl Adipate Consensus Reports
Reported in EPA TSCA Inventory.
Monoethyl Adipate Safety Profile
Mildly toxic by ingestion. When heated to decomposition it emits acrid smoke and irritating vapors.
Safety Statements:S24/25:Avoid contact with skin and eyes.
Monoethyl Adipate Specification
Adipic acid monoethyl ester ,its cas register number is 626-86-8. It also can be called Monoethyl Adipate ; Hexanedioic acid, monoethyl ester acid monoethyl ester . It is hazardous,so the first aid measures and others should be known. Such as: When on the skin: first,should flush skin with plenty of water immediately for at least 15 minutes while removing contaminated clothing. Secondly,Get medical aid. Or in the eyes: Flush eyes with plenty of water for at least 15 minutes, occasionally lifting the upper and lower eyelids. Then get medical aid soon. While ,it's Inhaled: Remove from exposure and move to fresh air immediately.Give artificial respiration while not breathing. When breathing is difficult, give oxygen. And as soon as to get medical aid. Then you have the ingesting of the product : If victim is conscious and alert, give 2-4 cupfuls of milk or water.
In addition, Adipic acid monoethyl ester (CAS NO.626-86-8) can be stable under normal temperature and pressure conditions. You must not take it with strong oxidants. And also prevent it to broken down into hazardous decomposition products: irritating and toxic fumes and gases.
Related Products
- Monoethyl Adipate
- Monoethyl butylphosphonate anhydride with diethyl phosphate
- Monoethyl fumarate
- Monoethyl itaconate
- Monoethyl maleate
- Monoethyl phthalate
- Monoethyldichlorothiophosphate
- Monoethylphenyltriazene
- 626-87-9
- 626-88-0
- 62689-07-0
- 626-89-1
- 62689-96-7
- 62690-13-5
- 626-93-7
- 6269-44-9
- 6269-46-1
- 626-97-1
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View