-
Name
N-METHYLPHENETHYLAMINE
- EINECS 209-632-3
- CAS No. 589-08-2
- Article Data87
- CAS DataBase
- Density 0.93 g/mL at 25 °C(lit.)
- Solubility
- Melting Point 0.93oC
- Formula C9H13N
- Boiling Point 201.739 °C at 760 mmHg
- Molecular Weight 135.209
- Flash Point 73.889 °C
- Transport Information UN 2735 8/PG 3
- Appearance clear colorless to light yellow liquid
- Safety 26-36/37/39-45-61-39
- Risk Codes 34-42/43-52/53-41-22
-
Molecular Structure
-
Hazard Symbols
 C,
C, Xn
Xn
- Synonyms Phenethylamine,N-methyl- (8CI);(2-(Methylamino)ethyl)benzene;(2-Phenylethyl)methylamine;N-(Phenylethyl)methylamine;N-Methyl-2-phenylethanamine;N-Methyl-N-(2-phenylethyl)amine;N-Methyl-b-phenethylamine;N-Methyl-b-phenylethylamine;N-Methylbenzeneethanamine;N-Methylphenethylamine;N-Phenethylmethylamine;
- PSA 12.03000
- LogP 1.83940
Synthetic route
-
-
109-73-9
N-butylamine

-
A
-
70038-63-0
butyl-(3-methyl-3H-benzothiazol-2-ylidene)-amine

-
B
-
589-08-2
N-Methyl-N-phenethylamine

| Conditions | Yield |
|---|---|
| In dichloromethane at 25℃; for 3h; | A 95% B 85% |

-
-
25566-59-0
N-methyl-N-(2-phenethyl)-4-methylbenzenesulfonamide

-
-
589-08-2
N-Methyl-N-phenethylamine

| Conditions | Yield |
|---|---|
| With sodium tetrahydroborate; methoxybenzene In ethanol Irradiation; other reducing agent; | 91% |
| With sodium tetrahydroborate; methoxybenzene In ethanol for 1h; Product distribution; Quantum yield; Irradiation; other solvents, reagents, reagents ratio; | 91% |
| With sodium tetrahydroborate; methoxybenzene In ethanol Mechanism; Rate constant; Quantum yield; Irradiation; effect of other reducing agents, other electron donors, other solvent; | 91% |
| With hydrogenchloride at 180℃; Hydrolysis; | |
| With tetrabutylammonium perchlorate In N,N-dimethyl-formamide various p-tolylsulfonamides, investigation of the deprotection by cathodic cleveage; |

| Conditions | Yield |
|---|---|
| Stage #1: 2-phenylethyl mesylate; methylamine In methanol; water at 140℃; under 15514.9 Torr; for 0.166667h; Flow reactor; Stage #2: With potassium hydroxide In water Solvent; | 90% |
-
-
25566-59-0
N-methyl-N-(2-phenethyl)-4-methylbenzenesulfonamide

-
A
-
536-57-2
p-toluene sulfinic acid

-
B
-
1858-86-2, 59153-38-7, 59203-02-0, 112458-71-6
Ethyl p-toluenesulfinate

-
C
-
672-78-6
methyl p-toluene sulfinate

-
D
-
589-08-2
N-Methyl-N-phenethylamine

| Conditions | Yield |
|---|---|
| With sodium tetrahydroborate; 1,5-dimethoxynaphthalene In ethanol Irradiation; | A 71% B n/a C n/a D 89% |


| Conditions | Yield |
|---|---|
| Stage #1: phenylacetaldehyde; methylamine With titanium(IV) isopropylate In methanol at 20℃; for 5h; Stage #2: With sodium tetrahydroborate In methanol at 20℃; for 2h; | 88% |
| With alkali durch Reduktion des Kondensationsprodukts mit Natrium und Alkohol; | |
| With formic acid; N-terminal His6-tagged formate dehydrogenase from Candida boidinii; Rs-amine dehydrogenase (originating from the enzyme engineeringof phenylalanine dehydrogenases from Rhodococcus sp. M4); NAD In aq. buffer at 30℃; for 48h; pH=8.5; Enzymatic reaction; |

-
-
589-08-2
N-Methyl-N-phenethylamine

| Conditions | Yield |
|---|---|
| With potassium phosphate; tris(2,2-bipyridine)ruthenium(II) hexafluorophosphate; ascorbic acid In water; acetonitrile at 20℃; for 16h; Sealed tube; Irradiation; Inert atmosphere; | 87% |

| Conditions | Yield |
|---|---|
| With sodium tetrahydroborate; bromine In 1,2-dimethoxyethane at -10 - 20℃; for 48h; | 82% |
| With hydrogenchloride; arsenic(III) trioxide at 35 - 40℃; Electrolysis.an Bleikathoden; | |
| With antimony pentoxide; sulfuric acid Electrolysis.an Bleikathoden; |
-
-
415929-57-6
N,N-dimethyl-4-((methyl(phenethyl)amino)methyl)aniline

-
-
589-08-2
N-Methyl-N-phenethylamine

| Conditions | Yield |
|---|---|
| With water; trifluoroacetic acid In acetonitrile at 120℃; for 5h; Sealed tube; | 75% |
-
-
67-56-1
methanol

-
-
6926-44-9
phenethyl azide

-
A
-
589-08-2
N-Methyl-N-phenethylamine

-
B
-
1126-71-2
N,N-dimethylphenethylamine

| Conditions | Yield |
|---|---|
| With trans-RuCl(phenpyra-Me)(PPh3)2PF6; sodium hydroxide at 125℃; for 36h; Sealed tube; Inert atmosphere; Glovebox; | A 10 %Chromat. B 75% |

-
-
78190-66-6
N-<1-Phenyl-2,2,2-trichlor-aethyl>-methylamin

-
-
589-08-2
N-Methyl-N-phenethylamine

| Conditions | Yield |
|---|---|
| With lithium aluminium tetrahydride In diethyl ether for 10h; Heating; | 71% |
-
-
25566-59-0
N-methyl-N-(2-phenethyl)-4-methylbenzenesulfonamide

-
A
-
80-40-0
ethyl ester of p-toluenesulfonic acid

-
B
-
589-08-2
N-Methyl-N-phenethylamine

| Conditions | Yield |
|---|---|
| With 1,5-dimethoxynaphthalene; sodium ethanolate In ethanol Irradiation; | A 45% B 62% |
-
-
67-56-1
methanol

-
-
6926-44-9
phenethyl azide

-
A
-
64-04-0
phenethylamine

-
B
-
589-08-2
N-Methyl-N-phenethylamine

| Conditions | Yield |
|---|---|
| With trans-RuCl(phenpyra-Me)(PPh3)2PF6; sodium hydroxide at 125℃; for 5h; Sealed tube; Inert atmosphere; Glovebox; | A 10 %Chromat. B 60% |


| Conditions | Yield |
|---|---|
| With boron trifluoride diethyl etherate In tetrahydrofuran | 56% |
-
-
383865-57-4
4-methoxy-7-morpholin-4-yl-benzothiazol-2-yl-amine

-
B
-
589-08-2
N-Methyl-N-phenethylamine

| Conditions | Yield |
|---|---|
| A 53% B n/a |
-
-
23069-99-0
N-(2-phenylethyl)formamide

-
-
589-08-2
N-Methyl-N-phenethylamine

| Conditions | Yield |
|---|---|
| With lithium aluminium tetrahydride In tetrahydrofuran Reflux; | 34% |
| With lithium aluminium tetrahydride; diethyl ether | |
| Multi-step reaction with 2 steps 1: potassium; toluene 2: concentrated hydrochloric acid / Hydrolysis View Scheme |

| Conditions | Yield |
|---|---|
| at 20℃; Erwaermen des Reaktionsprodukts mit Wasser; |
-
-
16766-83-9
2-(2-phenylethyl)-1,2-benzisothiazol-3(2H)-one 1,1-dioxide

-
-
589-08-2
N-Methyl-N-phenethylamine

| Conditions | Yield |
|---|---|
| ueber mehrere Stufen; |

| Conditions | Yield |
|---|---|
| beim Erhitzen ueber den Schmelzpunkt; | |
| beim Erhitzen ueber den Schmelzpunkt; |
-
-
27566-66-1
N-methyl-N-phenethylcyanamide

-
-
589-08-2
N-Methyl-N-phenethylamine

| Conditions | Yield |
|---|---|
| With hydrogenchloride at 160℃; | |
| Multi-step reaction with 2 steps 1: aqueous alcoholic sulfuric acid 2: hydrochloric acid / 160 °C View Scheme |
-
-
412274-51-2
N-(2-bromo-2-phenylethyl)-N,4-dimethylbenzenesulfonamide

-
-
589-08-2
N-Methyl-N-phenethylamine

| Conditions | Yield |
|---|---|
| With pentan-1-ol; sodium |
-
-
77130-13-3
N-Methylthiophenylacetamide

-
-
589-08-2
N-Methyl-N-phenethylamine

| Conditions | Yield |
|---|---|
| With hydrogenchloride at 30 - 40℃; Electrolysis.an Bleikathoden; | |
| With sodium tetrahydroborate; triethyloxonium fluoroborate 1.) CH2Cl2, 25 deg C, 10-20 min; 2.) anhydrous methanol 2-3 h; Yield given. Multistep reaction; |
-
-
52516-20-8
2-(3-Chlorophenyl)-N-methylethan-1-amine

-
A
-
824-21-5
N-methylindoline

-
B
-
589-08-2
N-Methyl-N-phenethylamine

| Conditions | Yield |
|---|---|
| With diethyl ether; phenyllithium; diethylamine |
-
-
90784-34-2
N-methyl-N-phenethylformamide

-
-
589-08-2
N-Methyl-N-phenethylamine

| Conditions | Yield |
|---|---|
| With hydrogenchloride Hydrolysis; | |
| With hydrogenchloride; methanol for 18h; Heating; |
-
-
73355-67-6
N-methyl-N-phenethyl-urea

-
-
589-08-2
N-Methyl-N-phenethylamine

| Conditions | Yield |
|---|---|
| With hydrogenchloride at 160℃; |
-
-
84395-76-6
(E)-N-benzyl-N-methyl-2-phenylethen-1-amine

-
-
589-08-2
N-Methyl-N-phenethylamine

| Conditions | Yield |
|---|---|
| With palladium on activated charcoal; ethanol Hydrogenation; |

| Conditions | Yield |
|---|---|
| With ethanol at 100℃; | |
| With benzene at 100℃; |

| Conditions | Yield |
|---|---|
| nachfolgende Hydrolyse; | |
| und Erwaermen des Reaktionsprodukts mit wasserhaltigem Aethanol; |
-
-
52516-17-3
2-(2-chlorochlorophenyl)-N-methylethanamine

-
-
591-51-5
phenyllithium

-
A
-
824-21-5
N-methylindoline

-
B
-
589-08-2
N-Methyl-N-phenethylamine

-
-
103-63-9
1-phenyl-2-bromoethane

-
-
74-89-5
methylamine

-
A
-
13977-33-8
N-methyl-N-phenethyl-2-phenylethan-1-amine

-
B
-
589-08-2
N-Methyl-N-phenethylamine

| Conditions | Yield |
|---|---|
| With ethanol | |
| In ethanol at 0 - 20℃; for 40h; Overall yield = 90 percent; Overall yield = 1.4 g; |

-
-
50-00-0
formaldehyd

-
-
1589-82-8
phenylmagnesium bromide

-
-
74-89-5
methylamine

-
-
589-08-2
N-Methyl-N-phenethylamine

| Conditions | Yield |
|---|---|
| With diethyl ether |

-
-
598-21-0
2-Bromoacetyl bromide

-
-
589-08-2
N-Methyl-N-phenethylamine

-
-
73391-97-6
N-methyl-N-2-phenylethyl-2-bromoacetamide

| Conditions | Yield |
|---|---|
| In dichloromethane at -25 - 20℃; | 100% |
| With potassium carbonate In dichloromethane at 0 - 20℃; for 3h; |
-
-
147577-61-5
(2R)-2-(N-(tert-butoxycarbonyl)-N-methylamino)-3-(2-naphthyl)-propionic acid

-
-
589-08-2
N-Methyl-N-phenethylamine

-
-
202810-83-1
N-methyl-N-((1R)-1-(N-methyl-N-phenethylcarbamoyl)-2-(2-naphthyl)ethyl)carbamic acid tert-butyl ester

| Conditions | Yield |
|---|---|
| Stage #1: (2R)-2-(N-(tert-butoxycarbonyl)-N-methylamino)-3-(2-naphthyl)-propionic acid With 1-hydroxy-7-aza-benzotriazole; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride In dichloromethane; N,N-dimethyl-formamide at 0℃; for 0.333333h; Addition; Stage #2: N-Methyl-N-phenethylamine In dichloromethane; N,N-dimethyl-formamide at 0 - 20℃; for 16h; Condensation; | 100% |
| With 1-hydroxy-7-aza-benzotriazole; N-(3-dimethylaminopropyl)-N'-ethylcarbodiimide hydrochloride In dichloromethane; N,N-dimethyl-formamide at 0 - 20℃; for 16.3h; | 1.89 g |
-
-
589-08-2
N-Methyl-N-phenethylamine

-
-
742104-52-5
(5α,7α)-17-(cyclopropylmethyl)-4,5-epoxy-3,6-dimethoxy-6,14-ethenomorphinan-7-carboxyl chloride

-
-
214064-60-5
C34H40N2O4

| Conditions | Yield |
|---|---|
| With triethylamine In chloroform at 20℃; | 100% |

| Conditions | Yield |
|---|---|
| With di-isopropyl azodicarboxylate; polymer-bound triphenylphosphine In tetrahydrofuran at 20℃; for 12h; | 99% |
| Stage #1: N-Methyl-N-phenethylamine With (acryloyloxymethyl)polystyrene In N,N-dimethyl-formamide at 20℃; for 18h; Solid phase reaction; Michael addition; Stage #2: allyl bromide In N,N-dimethyl-formamide at 20℃; for 18h; Solid phase reaction; quaternization; Stage #3: With N-ethyl-N,N-diisopropylamine In dichloromethane at 20℃; for 5h; Solid phase reaction; Hofmann elimination; | 81% |
| With sodium hydrogencarbonate In N,N-dimethyl-formamide at 70℃; for 2h; | 50% |
| Yield given. Multistep reaction; |

-
-
589-08-2
N-Methyl-N-phenethylamine

| Conditions | Yield |
|---|---|
| Stage #1: (1S,2S)-(+)-2-(methyl(phenethyl)amino)-1-phenylpropan-1-ol With methanesulfonyl chloride; triethylamine In diethyl ether at 0 - 20℃; Stage #2: With triethylamine In diethyl ether at 0 - 20℃; Stage #3: N-Methyl-N-phenethylamine In diethyl ether at 20℃; for 16h; | 99% |

| Conditions | Yield |
|---|---|
| With sodium acetate In methanol at 20℃; Inert atmosphere; Schlenk technique; | 99% |
-
-
68-12-2, 33513-42-7
N,N-dimethyl-formamide

-
-
589-08-2
N-Methyl-N-phenethylamine

-
-
90784-34-2
N-methyl-N-phenethylformamide

| Conditions | Yield |
|---|---|
| at 150℃; for 96h; Sealed tube; Inert atmosphere; Green chemistry; | 99% |
| at 150℃; for 96h; Inert atmosphere; Sealed tube; | 99% |
| With cobalt(II) acetate at 150℃; for 3h; Inert atmosphere; Sealed tube; | 95% |
-
-
589-08-2
N-Methyl-N-phenethylamine

-
-
170114-35-9
1-(2-Deoxy-β-D-erythro-pentofuranosyl)-4-(pentafluorophenyloxy)-2(1H)-pyrimidinone

| Conditions | Yield |
|---|---|
| In 1,4-dioxane at 80℃; | 98% |

| Conditions | Yield |
|---|---|
| With bis(1,5-cyclooctadiene)nickel (0); sodium t-butanolate; 1,3-bis[2,6-bis(1-methylethyl)phenyl]-1,3-dihydro-2H-imidazol-2-ylidene monohydrochloride In toluene at 100℃; for 20h; Inert atmosphere; Schlenk technique; | 98% |
-
-
10328-92-4
N-Methylisatoic anhydride

-
-
589-08-2
N-Methyl-N-phenethylamine

| Conditions | Yield |
|---|---|
| With isatoic anhydride resin In dichloromethane Ambient temperature; 1.) 14 h; 2.) 90 min; | 97% |
-
-
2130-96-3
O-benzyl-N-tert-butoxycarbonyl-L-tyrosine

-
-
589-08-2
N-Methyl-N-phenethylamine

-
-
173899-80-4
N-[Boc-(O-benzyl)-L-tyrosyl]-N-methyl-2-phenylethylamine

| Conditions | Yield |
|---|---|
| 97% |
-
-
100-39-0
benzyl bromide

-
-
589-08-2
N-Methyl-N-phenethylamine

-
-
10479-24-0
N-(2-phenethyl)benzylmethylamine

| Conditions | Yield |
|---|---|
| With di-isopropyl azodicarboxylate; polymer-bound triphenylphosphine In tetrahydrofuran at 20℃; for 12h; | 97% |
| Stage #1: N-Methyl-N-phenethylamine With REM resin at 20℃; for 2h; Stage #2: benzyl bromide In N,N-dimethyl-formamide for 18h; Stage #3: With N-ethyl-N,N-diisopropylamine In dichloromethane | 95% |
| Stage #1: N-Methyl-N-phenethylamine With REM resin at 20℃; for 3h; Michael addition; Stage #2: benzyl bromide In N,N-dimethyl-formamide at 20℃; for 18h; Stage #3: With potassium carbonate; N-ethyl-N,N-diisopropylamine In dichloromethane at 20℃; for 6h; |
-
-
865538-79-0
phenyl 1H-indazol-5-ylcarbamate

-
-
589-08-2
N-Methyl-N-phenethylamine

-
-
1357473-65-4
3-(1H-indazol-5-yl)-1-methyl-1-phenethylurea

| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine In tetrahydrofuran; N,N-dimethyl-formamide at 90℃; for 14h; | 97% |

-
-
589-08-2
N-Methyl-N-phenethylamine

| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine In isopropyl alcohol at 100℃; | 97% |

-
-
589-08-2
N-Methyl-N-phenethylamine

| Conditions | Yield |
|---|---|
| at 70℃; for 2h; | 97% |

| Conditions | Yield |
|---|---|
| With palladium 10% on activated carbon In N,N-dimethyl acetamide under 760.051 Torr; for 16h; Sealed tube; Heating; Schlenk technique; | 96.2% |

-
-
12081-54-8
tetraethyldiborane(6)

-
-
589-08-2
N-Methyl-N-phenethylamine

-
-
22100-27-2
{methyl-(2-phenylethyl)amino}diethylborane

| Conditions | Yield |
|---|---|
| 3 h at 70-100°C; | 96% |
| 3 h at 70-100°C; | 96% |
-
-
626-60-8
3-Chloropyridine

-
-
589-08-2
N-Methyl-N-phenethylamine

-
-
1445086-57-6
N-methyl-N-phenethylpyridin-3-amine

| Conditions | Yield |
|---|---|
| With C43H58ClO2PPd; sodium t-butanolate; ruphos In tetrahydrofuran at 85℃; for 6h; Inert atmosphere; | 96% |
| With C30H43O2P*C13H13NO3PdS; sodium t-butanolate; ruphos In tetrahydrofuran at 85℃; for 24h; | 80% |
| With methanesulfonato(2-dicyclohexylphosphino-2’,6’-di-i-propoxy-1,1’-biphenyl)(2’-methylamino-1,1‘-biphenyl-2-yl)palladium(II); sodium t-butanolate; ruphos In tetrahydrofuran at 85℃; for 24h; Inert atmosphere; | 80% |
-
-
56146-83-9
chlorosulfonyl-acetic acid methyl ester

-
-
589-08-2
N-Methyl-N-phenethylamine

-
-
233283-81-3
2-[methyl(phenethyl)sulfamoyl]acetic acid methyl ester

| Conditions | Yield |
|---|---|
| Stage #1: chlorosulfonyl-acetic acid methyl ester; N-Methyl-N-phenethylamine With triethylamine In tetrahydrofuran at 20℃; Stage #2: With hydrogenchloride In water pH=7; | 96% |
-
-
52805-48-8
2-(Benzyloxy)-1-naphthaldehyde

-
-
589-08-2
N-Methyl-N-phenethylamine

| Conditions | Yield |
|---|---|
| With sodium tris(acetoxy)borohydride; acetic acid In 1,2-dichloro-ethane at 20℃; for 2.5h; | 96% |

| Conditions | Yield |
|---|---|
| With manganese(II) chloride tetrahydrate at 150℃; for 10h; Sealed tube; Microwave irradiation; Inert atmosphere; | 96% |
| With cobalt(II) diacetate tetrahydrate at 150℃; for 3h; Sealed tube; Inert atmosphere; | 83% |

| Conditions | Yield |
|---|---|
| With 1,4-diaza-bicyclo[2.2.2]octane; titanium(IV) isopropylate; hydrogen; iodine; triethylamine In tetrahydrofuran; dichloromethane; 1,2-dichloro-ethane at 20℃; under 30402 Torr; for 20h; | 96% |
-
-
536-74-3
phenylacetylene

-
-
589-08-2
N-Methyl-N-phenethylamine

-
-
220501-09-7
N-methyl-N-phenethyl-2-phenylacetamide

| Conditions | Yield |
|---|---|
| With potassium hexafluorophosphate; 4-methylpyridine-1-oxide; chloro(cyclopentadienyl)bis(triphenylphosphine)ruthenium (II) In 1,2-dichloro-ethane at 100℃; for 24h; Catalytic behavior; Reagent/catalyst; Solvent; Temperature; Inert atmosphere; Sealed tube; | 96% |
-
-
32387-21-6
1-methyl-3-indolecarboxylic acid

-
-
589-08-2
N-Methyl-N-phenethylamine

| Conditions | Yield |
|---|---|
| With PS-carbodiimide; benzotriazol-1-ol In 1-methyl-pyrrolidin-2-one at 100℃; for 0.0833333h; microwave irradiation; | 95% |

| Conditions | Yield |
|---|---|
| With di-isopropyl azodicarboxylate; polymer-bound triphenylphosphine In tetrahydrofuran at 20℃; for 12h; | 95% |
-
-
589-08-2
N-Methyl-N-phenethylamine

-
-
183208-35-7
5-bromo-1H-pyrrolo[2,3-b]pyridine

-
-
1248587-68-9
N-methyl-N-phenethyl-1H-pyrrolo[2,3-b]pyridin-5-amine

| Conditions | Yield |
|---|---|
| With RuPhos palladacycle; lithium hexamethyldisilazane; ruphos In tetrahydrofuran at 65℃; for 4h; Inert atmosphere; Sealed vial; | 95% |

| Conditions | Yield |
|---|---|
| With manganese(II) chloride tetrahydrate at 150℃; for 10h; Sealed tube; Microwave irradiation; Inert atmosphere; | 95% |
| With cobalt(II) diacetate tetrahydrate at 150℃; for 3h; Sealed tube; Inert atmosphere; | 46% |
-
-
64596-35-6
phenyl (benzoyloxy)carbamate

-
-
589-08-2
N-Methyl-N-phenethylamine

| Conditions | Yield |
|---|---|
| With 1H-imidazole In tetrahydrofuran at 100℃; for 2h; Microwave irradiation; Sealed tube; | 95% |

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetone at 20℃; for 3h; | 95% |
N-Methylphenethylamine Specification
The Benzeneethanamine,N-methyl-, with the CAS registry number 589-08-2, is also known as N-Methyl-N-(2-phenylethyl)amine. It belongs to the product categories of Amines and Anilines; Phenyls & Phenyl-Het; Phenyls & Phenyl-Het. Its EINECS registry number is 209-632-3. This chemical's molecular formula is C9H13N and molecular weight is 135.21. What's more, both its IUPAC name and systematic name are the same which is called N-Methyl-2-phenylethanamine. It should be stored in a cool, dry and well-ventilated place. It is a chemical compound derived from phenethylamine. Its N-substitution results in less metabolism by monoamine oxidase than phenethylamine itself.
Physical properties about Benzeneethanamine,N-methyl- are: (1)ACD/LogP: 1.848; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): -1.24; (4)ACD/LogD (pH 7.4): -0.87; (5)ACD/BCF (pH 5.5): 1.00; (6)ACD/BCF (pH 7.4): 1.00; (7)ACD/KOC (pH 5.5): 1.00; (8)ACD/KOC (pH 7.4): 1.00; (9)#H bond acceptors: 1; (10)#H bond donors: 1; (11)#Freely Rotating Bonds: 3 ; (12)Polar Surface Area: 12.03 Å2; (13)Index of Refraction: 1.509; (14)Molar Refractivity: 44.03 cm3; (15)Molar Volume: 147.574 cm3; (16)Polarizability: 17.455×10-24cm3; (17)Surface Tension: 32.228 dyne/cm; (18)Density: 0.916 g/cm3; (19)Flash Point: 73.889 °C; (20)Enthalpy of Vaporization: 43.795 kJ/mol; (21)Boiling Point: 201.739 °C at 760 mmHg; (22)Vapour Pressure: 0.303 mmHg at 25 °C.
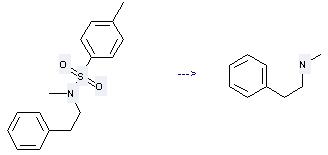
Preparation of Benzeneethanamine,N-methyl-: this chemical can be prepared by N-methyl-N-phenethyl-toluene-4-sulfonamide. This reaction needs reagent concentrated hydrochloric acid at temperature of 180 °C.
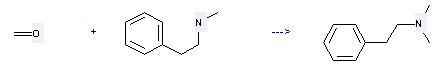
Uses of Benzeneethanamine,N-methyl-: it is used to produce other chemicals. For example, it can react with formaldehyde to get dimethyl-phenethyl-amine. The reaction occurs with reagent NaBH4 and solvents tetrahydrofuran, trifluoroacetic acid at the temperature of 25 °C. The reaction time is 24 hours. The yield is 87 %.
When you are dealing with this chemical, you should be very careful. This chemical is inflammation to the skin or other mucous membranes and may destroy living tissue on contacting. What's more, it may cause burns. Therefore, you should wear suitable protective clothing, gloves and eye/face protection. This material and its container must be disposed of as hazardous waste. In case of contacting with eyes, you should rinse immediately with plenty of water and seek medical advice. And in case of accident or if you feel unwell you should seek medical advice immediately.
You can still convert the following datas into molecular structure:
(1) SMILES: N(CCc1ccccc1)C
(2) InChI: InChI=1S/C9H13N/c1-10-8-7-9-5-3-2-4-6-9/h2-6,10H,7-8H2,1H3
(3) InChIKey: SASNBVQSOZSTPD-UHFFFAOYSA-N
The toxicity data is as follows:
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| mouse | LD50 | parenteral | 180mg/kg (180mg/kg) | Naunyn-Schmiedeberg's Archiv fuer Experimentelle Pathologie und Pharmakologie. Vol. 195, Pg. 647, 1940. | |
| mouse | LDLo | intraperitoneal | 190mg/kg (190mg/kg) | Journal of Pharmacology and Experimental Therapeutics. Vol. 106, Pg. 341, 1952. | |
| rat | LDLo | intraperitoneal | 180mg/kg (180mg/kg) | Naunyn-Schmiedeberg's Archiv fuer Experimentelle Pathologie und Pharmakologie. Vol. 195, Pg. 647, 1940. | |
| rat | LDLo | oral | 1400mg/kg (1400mg/kg) | Naunyn-Schmiedeberg's Archiv fuer Experimentelle Pathologie und Pharmakologie. Vol. 195, Pg. 647, 1940. |
Related Products
- N-Methylphenethylamine
- 58909-39-0
- 58909-56-1
- 589-10-6
- 5891-21-4
- 5891-41-8
- 5891-45-2
- 589-15-1
- 589-16-2
- 58917-25-2
- 58917-26-3
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View