 This product is a nationally controlled contraband, and the Lookchem platform doesn't provide relevant sales information.
This product is a nationally controlled contraband, and the Lookchem platform doesn't provide relevant sales information.
-
Name
Toluene
- EINECS 203-625-9
- CAS No. 108-88-3
- Article Data2432
- CAS DataBase
- Density 0.872 g/cm3
- Solubility Insoluble in water, miscible with benzene, alcohol, ether and other organic solvents
- Melting Point -95 °C
- Formula C7H8
- Boiling Point 110.602 °C at 760 mmHg
- Molecular Weight 92.1405
- Flash Point 10 °C
- Transport Information UN 1294 3/PG 2
- Appearance Colourless liquid with a benzene-like odour
- Safety 36/37-46-62-45
- Risk Codes 11-38-48/20-63-65-67-39/23/24/25-23/24/25
-
Molecular Structure
-
Hazard Symbols
 F,
F, Xn,
Xn, T
T
- Synonyms 1-Methylbenzene;Antisal 1a;CP 25;CP 25 (solvent);Methacide;Methylbenzene;Methylbenzol;NSC 406333;Phenylmethane;Toluol;Toluene(8CI);
- PSA 0.00000
- LogP 1.99500
Synthetic route

| Conditions | Yield |
|---|---|
| With hydrogen; Lindlar's catalyst In benzene under 760 Torr; for 0.5h; Ambient temperature; | 100% |
| With sulfuric acid; aluminium; nickel dichloride In water at 25℃; Product distribution; | 98% |
| With sodium tetrahydroborate; cetyltributylphosphonium bromide In water; toluene at 40℃; for 0h; Product distribution; | 96% |

| Conditions | Yield |
|---|---|
| With hydrogenchloride; aluminium; nickel dichloride In ethanol at 25℃; Product distribution; | 100% |
| With palladium dichloride In methanol at 40℃; for 24h; Green chemistry; chemoselective reaction; | 99% |
| With palladium on activated charcoal; hydrogen In tetrahydrofuran at 25℃; under 760.051 Torr; for 1.5h; | 99% |

| Conditions | Yield |
|---|---|
| With (1,1'-bis(diphenylphosphino)ferrocene)palladium(II) dichloride; cesium fluoride In 2-pentanol at 100℃; for 12h; Inert atmosphere; | 100% |
| With lithium aluminium tetrahydride In 1,2-dimethoxyethane at 35℃; for 5h; ultrasonic acceleration of reduction; | 95% |
| With 2,2'-azobis(isobutyronitrile); poly(n-hexylsilane) In benzene-d6 at 82 - 85℃; for 3h; sealed; | 99 % Spectr. |

| Conditions | Yield |
|---|---|
| With water; sodium iodide; nickel dichloride; zinc; sonication In N,N,N,N,N,N-hexamethylphosphoric triamide at 60℃; for 3h; | 100% |
| With water; sodium iodide; nickel dichloride; zinc; sonication In N,N,N,N,N,N-hexamethylphosphoric triamide at 60℃; for 3h; Product distribution; | 100% |
| With palladium on ceria; sodium hydroxide In isopropyl alcohol at 40℃; for 24h; Temperature; Solvent; Irradiation; Inert atmosphere; Sealed tube; | 97% |

| Conditions | Yield |
|---|---|
| With potassium hydroxide; isopropyl alcohol; polymer supported Na2PdCl4 at 82℃; for 0.666667h; Rate constant; further catalysts; influence of the nature of catalyst on the rate of dehalogenation; | 100% |
| With tetrakis(triphenylphosphine) palladium(0); formaldehyd; caesium carbonate In dimethyl sulfoxide at 80℃; for 12h; | 99% |
| With lithium aluminium tetrahydride In 1,2-dimethoxyethane at 35℃; for 5h; ultrasonic acceleration of reduction; | 97% |

| Conditions | Yield |
|---|---|
| With sodium tetrahydroborate; cetyltributylphosphonium bromide In water; toluene at 18℃; for 0h; Product distribution; | 100% |
| With ammonium chloride; zinc In tetrahydrofuran; water at 20℃; for 4h; | 97% |
| With sodium tetrahydroborate In tetrahydrofuran for 0.0833333h; Ambient temperature; | 90% |
-
-
77924-17-5
1-Phenyl-5-p-tolyloxy-1H-tetrazole

-
A
-
5097-82-5
1-phenyl-5-hydroxytetrazole

-
B
-
108-88-3
toluene

| Conditions | Yield |
|---|---|
| With hydrazine hydrate; palladium on activated charcoal In ethanol; water; benzene for 3.5h; Ambient temperature; | A n/a B 100% |
| With hydrazine hydrate; palladium on activated charcoal In ethanol; benzene Mechanism; Ambient temperature; | A n/a B 100% |
| With sodium hypophosphite; palladium on activated charcoal In ethanol; benzene at 80℃; Relative steady-state rates of cleavage, relative extrapolated interceps; |

| Conditions | Yield |
|---|---|
| With tetrakis(triphenylphosphine)palladium dichloride In benzene at 80℃; for 7h; | 100% |

| Conditions | Yield |
|---|---|
| With hydrogen; palladium on activated charcoal In water; isopropyl alcohol at 20℃; under 5171.62 Torr; for 1h; | 100% |

-
-
108-88-3
toluene

| Conditions | Yield |
|---|---|
| With perchloric acid In acetonitrile Kinetics; | 100% |
-
-
60528-58-7
(η6-toluene)bis(η1-pentafluorophenyl)cobalt(II)

-
A
-
434-90-2
decafluorobiphenyl

-
B
-
7440-48-4
cobalt

-
C
-
108-88-3
toluene

| Conditions | Yield |
|---|---|
| In neat (no solvent, solid phase) pyrolysis at 150°C; | A 93% B n/a C 100% |


| Conditions | Yield |
|---|---|
| With zeolite Y with a silica-alumina at 250℃; for 5h; Temperature; Inert atmosphere; | 100% |

| Conditions | Yield |
|---|---|
| With Ni0.85Rh0.15; hydrogen In water at 95℃; under 760.051 Torr; for 16h; Reagent/catalyst; | A 100% B 5% C 79% |
| With isopropyl alcohol at 150℃; under 7500.75 Torr; for 48h; Inert atmosphere; Autoclave; | A 100% B 12.2% C 87.5% |
| With isopropyl alcohol at 150℃; under 7500.75 Torr; for 3h; Catalytic behavior; Temperature; Inert atmosphere; Autoclave; | A 100% B 73.1% C 22.4% |
| With Ru0.6Ni0.4; hydrogen In water at 95℃; under 760.051 Torr; for 16h; Reagent/catalyst; | A 98% B 61% C 6% |
| With hydrogen In n-heptane at 140℃; under 750.075 Torr; for 6h; Catalytic behavior; | A 34 %Chromat. B 12 %Chromat. C 22 %Chromat. |


| Conditions | Yield |
|---|---|
| With hydrogen In n-heptane at 160℃; under 750.075 Torr; for 6h; Catalytic behavior; | A 96 %Chromat. B 100% |

| Conditions | Yield |
|---|---|
| With hydrogen In water; ethyl acetate at 50℃; under 15001.5 Torr; for 5h; | 99% |
| With hydrogen In water at 25℃; for 1h; | 99% |
| With palladium on activated charcoal; hydrogen In methanol at 25℃; under 760.051 Torr; for 0.833333h; | 99% |

| Conditions | Yield |
|---|---|
| With Ni0.85Ru0.15; hydrogen In water at 95℃; under 760.051 Torr; for 16h; Reagent/catalyst; | A 99% B 45% |
| With 0.5%Pd/TiO2; isopropyl alcohol In water at 24.84℃; for 2h; Inert atmosphere; Sealed tube; Irradiation; | A 99% B 99% |
| With formic acid In water at 120℃; Green chemistry; | A 98% B 96% |

| Conditions | Yield |
|---|---|
| With copper nickel; pyrographite In 1,2-dimethoxyethane at 85℃; for 20h; | A 99% B 1% |
| With titanium(III) citrate; Tris buffer; tetra(n-butyl)ammonium hydroxide; vitamin B-12 In ethanol for 1h; pH=8; | A 98% B 1% |
| With potassium phosphate; (4,4'-di-tert-butyl-2,2'-dipyridyl)-bis-(2-phenylpyridine(-1H))-iridium(III) hexafluorophosphate; diethyl 2,6-dimethyl-1,4-dihydropyridine-3,5-dicarboxylate In N,N-dimethyl-formamide at 20℃; for 4h; Inert atmosphere; Irradiation; | A 85% B 6% |
-
-
132636-69-2
3-(4-Chloro-2-methyl-phenoxy)-benzo[d]isothiazole 1,1-dioxide

-
A
-
108-88-3
toluene

-
B
-
81-07-2
saccharin

| Conditions | Yield |
|---|---|
| With sodium hypophosphite; palladium on activated charcoal In water; benzene for 3h; Heating; | A 99% B n/a |

| Conditions | Yield |
|---|---|
| With formic acid In water at 120℃; Green chemistry; | A 97% B 99% |
| With 9,10-dihydroanthracene at 224.84℃; Kinetics; Product distribution; | |
| With hydrogen In hexane at 260℃; under 5250.53 Torr; for 10h; Autoclave; | A 93.6 %Chromat. B 82.9 %Chromat. |
| With isopropyl alcohol at 260℃; under 15001.5 Torr; for 4h; Autoclave; Inert atmosphere; Green chemistry; | A 85 %Chromat. B 88 %Chromat. |
-
-
78709-76-9
{bis(triphenylphosphine)nitrogen}{HW(CO)5}

-
-
100-39-0
benzyl bromide

-
A
-
15131-04-1
bromopentacarbonyl tungstate(0)(1-)

-
B
-
108-88-3
toluene

| Conditions | Yield |
|---|---|
| In tetrahydrofuran Kinetics; 26.0°C, 20-fold excess of alkyl halide; second-order rate const. is given; anal. of the reaction mixt. by gas chromy.; | A n/a B 99% |


| Conditions | Yield |
|---|---|
| With methyl iodide; bis(triphenylphosphine)nickel(II) chloride In tetrahydrofuran addn. of Ni-catalyst and alkyl halide to soln. of PhYbI (prepd. in situ from Yb metal and PhI in THF), agitation (25°C); GLC; | A n/a B 99% |
| With methyl iodide; tetrakis(triphenylphosphine) palladium(0) In tetrahydrofuran addn. of Ni-catalyst and alkyl halide to soln. of PhYbI (prepd. in situ from Yb metal and PhI in THF), agitation (25°C); GLC; | A n/a B 97% |

| Conditions | Yield |
|---|---|
| With palladium dichloride In methanol at 40℃; for 12h; Inert atmosphere; Green chemistry; chemoselective reaction; | 99% |
| With methanol; toluene-4-sulfonic acid at 25℃; for 7.5h; Reagent/catalyst; Inert atmosphere; Sealed tube; UV-irradiation; | 95% |
| With samarium diiodide; water In tetrahydrofuran; decane at 20℃; | |
| With diisobutylaluminium hydride; sodium t-butanolate; bis(1,5-cyclooctadiene)nickel(0); 1,3-bis[(2,6-diisopropyl)phenyl]imidazolinium chloride In tetrahydrofuran at 80℃; for 16h; Product distribution / selectivity; Inert atmosphere; | 82 %Chromat. |
-
-
946-80-5
(benzyloxy)benzene

-
A
-
82166-21-0
methyl cyclohexane

-
B
-
108-88-3
toluene

-
C
-
108-93-0
cyclohexanol

| Conditions | Yield |
|---|---|
| With hydrogen In isopropyl alcohol at 120℃; for 2h; | A 28% B 72% C 99% |
| With Rh0.6Ni0.4; hydrogen In water at 95℃; under 760.051 Torr; for 16h; Reagent/catalyst; | A 35% B 63% C 95% |
| With 10% Pd/C; hydrogen In hexane at 160℃; under 30003 Torr; for 2h; Autoclave; | |
| With isopropyl alcohol at 170℃; for 15h; Sealed tube; | A 0.68 mmol B 0.28 mmol C 0.94 mmol |


| Conditions | Yield |
|---|---|
| With sodium hydroxide at 24.84℃; under 760.051 Torr; for 6h; Inert atmosphere; UV-irradiation; Sealed tube; | 99% |

| Conditions | Yield |
|---|---|
| With sodium hydroxide at 24.84℃; under 760.051 Torr; for 6h; Inert atmosphere; UV-irradiation; Sealed tube; | 99% |

| Conditions | Yield |
|---|---|
| With 0.5%Pd/TiO2; isopropyl alcohol In water at 24.84℃; for 2h; Inert atmosphere; Sealed tube; Irradiation; | A 99% B 97% |

| Conditions | Yield |
|---|---|
| With 0.5%Pd/TiO2; isopropyl alcohol In water at 24.84℃; for 2h; Inert atmosphere; Sealed tube; Irradiation; | A 99% B 99% |

| Conditions | Yield |
|---|---|
| With 0.5%Pd/TiO2; isopropyl alcohol In water at 24.84℃; for 2h; Inert atmosphere; Sealed tube; Irradiation; | A 99% B 99% |

| Conditions | Yield |
|---|---|
| With hydrogen In n-heptane at 140℃; under 750.075 Torr; for 6h; Catalytic behavior; | A 99% B 99% |

| Conditions | Yield |
|---|---|
| platinum; potassium oxide at 360℃; | A 98% B 2% |
| platinum; potassium oxide at 360℃; Product distribution; other content of catalyst; | A 98% B 2% |
-
-
38136-29-7
4-methylvaleroyl chloride

-
-
108-88-3
toluene

-
-
21847-98-3
4-methyl-1-(4'-methylphenyl)pentan-1-one

| Conditions | Yield |
|---|---|
| Friedel Crafts Acylation; | 100% |
| With aluminium trichloride at 75 - 80℃; for 1h; | 35% |
| With aluminium trichloride | |
| Friedel-Crafts reaction; |

| Conditions | Yield |
|---|---|
| With gallium(III) trichloride at 20℃; for 24h; | 100% |
| With aluminium trichloride at 20℃; for 24h; | 68% |
| With aluminium trichloride Behandeln bei Siedetemperatur; |

| Conditions | Yield |
|---|---|
| With bromine In tetrachloromethane for 1.5h; Ambient temperature; | 100% |
| With manganese(IV) oxide; bromine In dichloromethane at 0℃; for 1h; Product distribution; Further Variations:; Solvents; Temperatures; reaction time; | 100% |
| With bromine; sodium t-butanolate In cyclohexane Heating; | 100% |

| Conditions | Yield |
|---|---|
| With laccase from Coriolus versicolor MTCC-138; 2,2'-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid) diammonium salt In 1,4-dioxane for 0.5h; pH=4.5; Green chemistry; Enzymatic reaction; | 100% |
| With nickel-doped graphene carbon nitride nanoparticles; air In ethanol at 25℃; for 8h; Reagent/catalyst; Solvent; Irradiation; Green chemistry; | 98% |
| With water at 20℃; for 3h; Reagent/catalyst; | 98% |
-
-
108-88-3
toluene

-
-
82166-21-0
methyl cyclohexane

| Conditions | Yield |
|---|---|
| With hydrogen; [(norbornadiene)rhodium(I)chloride]2; phosphinated polydiacetylene In n-heptane at 30℃; under 60800 Torr; for 1.5h; | 100% |
| With hydrogen; Ni-Tc on γ-Al2O3 at 175 - 250℃; under 760 Torr; Product distribution; dependence of catalytic activity on the reduction temperature; enhanced activity of bimetallic catalysts; | 100% |
| With hydrogen; Rh on carbon In methanol at 20℃; under 760.051 Torr; for 0.5h; | 100% |

| Conditions | Yield |
|---|---|
| With methyl cyanoformate; sodium decatungstate In acetonitrile at 8℃; for 90h; Irradiation; | 100% |
| With di-tert-butyl peroxide; sodium acetate at 120℃; for 10h; Schlenk technique; Green chemistry; | 94% |
| With di-tert-butyl peroxide; sodium acetate at 120℃; for 10h; | 94% |

| Conditions | Yield |
|---|---|
| Ce3+ exchanged Y-fanjasite at 150℃; for 48h; | 100% |
-
-
1711-05-3
m-anisoyl chloride

-
-
108-88-3
toluene

-
-
82520-37-4
(4-methylphenyl)-(3-methoxyphenyl)methanone

| Conditions | Yield |
|---|---|
| Stage #1: m-anisoyl chloride; toluene With aluminum (III) chloride In dichloromethane at 20℃; for 4h; Stage #2: With hydrogenchloride In dichloromethane; water | 100% |
| With aluminium trichloride | 84% |
| With aluminium trichloride In nitromethane for 0.75h; Ambient temperature; | 84% |
| With aluminium trichloride In 1,2-dichloro-benzene | |
| With aluminium trichloride In dichloromethane at 20℃; |

| Conditions | Yield |
|---|---|
| With α-manganese oxide; oxygen at 290℃; Temperature; [1,2]-Wittig Rearrangement; Inert atmosphere; | 100% |
| With oxygen at 258℃; under 760.051 Torr; Kinetics; Reagent/catalyst; Temperature; Inert atmosphere; | 100% |
| With oxygen at 230 - 240℃; for 0.5h; Reagent/catalyst; Temperature; Inert atmosphere; | 99.9% |

| Conditions | Yield |
|---|---|
| Stage #1: toluene; 3-methyl-3-phenyl-2-methyleneoxetane With n-butyllithium; N,N,N,N,-tetramethylethylenediamine In hexane at 0℃; for 1.5h; Stage #2: chloro-trimethyl-silane In hexane at 0℃; for 0.55h; | 100% |

-
-
108-88-3
toluene

-
-
25015-63-8
4,4,5,5-tetramethyl-[1,3,2]-dioxaboralane

-
-
195062-57-8
p-tolylboronic pinacol ester

| Conditions | Yield |
|---|---|
| [Ir(COD)(1,3-dicyclohexylimidazolidin-2-ylidene)2]CF3CO2 at 40℃; for 10h; | 100% |
| (η4-1,5-cyclooctadiene)bis(1,3-dimethylimidazolin-2-ylidene)iridium(I) trifluoracetate In toluene byproducts: H2; (N2); using Schlenk techniques; dissolving of 2 mmol pinacolborane and 1.5 mol% of Ir(COD)(C3H2N2Me2)2CF3CO2 in toluene; stirring and heating at40°C for 12 h; monitoring by GC-MS; removal of solvent under vac. at room temp.; chromy. over silica gel, eluting with CH2Cl2; | 100% |
| (η4-1,5-cyclooctadiene)(1,1'-dimethyl-3,3'-o-xylylene-diimidazolin-2,2'-diylidene)iridium(I) trifluoroacetate In toluene (N2); using Schlenk techniques; dissolving of 2 mmol pinacolborane and 1.5 mol% of Ir(COD)(1,1'-dimethyl-3,3'-o-xylylene-diimidazolin-2,2'-diylidene)2(CF3CO2) in toluene; stirring and heating at 40°C for 12 h; monitoring by GC-MS; removal of solvent under vac. at room temp.; chromy. over silica gel, eluting with CH2Cl2; | 100% |
-
-
199620-14-9, 13007-92-6
chromium(0) hexacarbonyl

-
-
108-88-3
toluene

-
-
12083-24-8
tricarbonyl(η(6)-toluene)chromium

| Conditions | Yield |
|---|---|
| With catalyst: dimethyl succinate In decalin byproducts: CO; refluxing for 2.3 h (catalyst: dimethyl succinate); freezing out at -18°C or quick chromy. of the decaline soln. on a SiO2 column; | 100% |
| With catalyst: butyl acetate In decalin byproducts: CO; refluxing for 3 h (catalyst: butyl acetate); freezing out at -18°C or quick chromy. of the decaline soln. on a SiO2 column; | 99% |
| With acetic acid In decalin refluxing of toluene with the Cr compd. (4:1) and 25 microliter AcOH for 4 h; | 98.7% |
-
-
51031-50-6
heptafluoropropane-2-sulphenic acid chloride

-
-
108-88-3
toluene

-
A
-
754-62-1
bis(perfluoroisopropyl) disulfide

-
B
-
68408-97-9
heptafluoropropane-2-thiol

-
C
-
68409-03-0
benzyl perfluoroisopropyl sulfide

-
D
-
100-44-7
benzyl chloride

| Conditions | Yield |
|---|---|
| A n/a B n/a C 4% D 100% | |
| A n/a B n/a C 4% D 100% |

-
-
32308-83-1
nonafluoro-tert-butanesulfenyl chloride

-
-
108-88-3
toluene

-
A
-
32308-82-0
nonafluoro-tert-butanethiol

-
B
-
100-44-7
benzyl chloride

| Conditions | Yield |
|---|---|
| A 100% B 100% | |
| A 100% B 100% |


| Conditions | Yield |
|---|---|
| In tetrahydrofuran; toluene stirring Mn2(CO)10 with Me3NO*2H2O in THF (18 h), solvent removal; recrystn. (hot toluene); | 100% |
| In toluene byproducts: trimethylammonium carbonate; treatment of Mn2(CO)10 with 6 equivs. of Me3NO, boiling of product in MePh; hot filtration, crystn. (5°C); second crop from mother liquor; elem. anal.; | 98% |

-
-
86197-44-6
bis(pentafluorophenyl)(η6-anisole)cobalt(II)

-
-
108-88-3
toluene

-
-
60528-58-7
(η6-toluene)bis(η1-pentafluorophenyl)cobalt(II)

| Conditions | Yield |
|---|---|
| In chloroform-d1 byproducts: anisole; mol. ratio 1/10; not isolated, detected by NMR; | 100% |
| In chloroform-d1 byproducts: anisole; mol. ratio 1/1; not isolated, detected by NMR; | 85% |

| Conditions | Yield |
|---|---|
| In toluene byproducts: HCl; under N2; 2-tert-butyl-4-methylphenol in toluene added to TiCl4 (molar ratio 1:1) in toluene; refluxed for 13 h; filtered; solvent removed; dried under vac. for 4 h; elem. anal.; | 100% |


| Conditions | Yield |
|---|---|
| In hexane | 100% |
| In hexane; toluene B(C6F5)3 and AlMe3 in 1:3 toluene/hexanes mixt.; | 99% |
| In hexane; toluene (inert atm.); reaction of borane deriv. with trimethylaluminium in hexane/toluene (3:1); | 99% |


-
-
108-88-3
toluene

| Conditions | Yield |
|---|---|
| In toluene 100 mL vessel filled with a soln. of Rh(C8H12)diphosphiteBF4 in toluene, placed into an autoclave, autoclave purged 3 times with H2 and pressurised to the appropriate pressure (5 bar), after react. time of 3 h at room temp. autoclave depressurised; concentration to dryness, elem. anal.; | 100% |
Toluene History
The name Toluene (CAS NO.108-88-3) was derived from the older name toluol, which refers to tolu balsam, an aromatic extract from the tropical Colombian tree Myroxylon balsamum, from which it was first isolated. It was originally named by J?ns Jakob Berzelius.
Toluene Consensus Reports
Community Right-To-Know List. Reported in EPA TSCA Inventory. EPA Genetic Toxicology Program.
Toluene Standards and Recommendations
OSHA PEL: TWA 100 ppm; STEL 150 ppm
ACGIH TLV: TWA 50 ppm (skin); Not Classifiable as a Human Carcinogen; BEI: 1.6 g/g creatinine of hippuric acid in urine at end of shift; 0.05 mg/L toluene in venous blood prior to last shift of workweek; BEI: BEI: 0.05 mg/L toluene in venous blood prior to last shift of work ek; 0.5 mg/L o-cresol in urine at end of shift
DFG MAK: 50 ppm (190 mg/m3); BAT: 340 μg/dL in blood at end of shift
NIOSH REL: (Toluene) TWA 100 ppm; CL 200 ppm/10M
DOT Classification: 3; Label: Flammable Liquid
Toluene Analytical Methods
For occupational chemical analysis use NIOSH: Hydrocarbons, Aromatic, 1501; Hydrocarbons, Bp: 36–126°C, 1500.
Toluene Specification
Toluene, also called Methylbenzene, is a colorless, flammable liquid with a benzenelike odor. It is insoluble in water, but miscible with benzene, alcohol, ether and other organic solvents. Toluene was first isolated as aromatic extract from the tropical Colombian tree Myroxylon balsamum. In chemical, it is a mono-substituted benzene derivative commonly used as an industrial solvent for the manufacturing of paints, chemicals, pharmaceuticals, and rubber. And it is not only an important organic solvent, but also dissolving a number of notable inorganic chemicals such as sulfur.
Physical properties about Toluene are: (1)ACD/LogP: 2.72; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): 2.72; (4)ACD/LogD (pH 7.4): 2.72; (5)ACD/BCF (pH 5.5): 68.71; (6)ACD/BCF (pH 7.4): 68.71; (7)ACD/KOC (pH 5.5): 718.68; (8)Index of Refraction: 1.5; (9)Molar Refractivity: 31.078 cm3; (10)Molar Volume: 105.709 cm3; (11)Polarizability: 12.32 10-24cm3; (12)Surface Tension: 28.8169994354248 dyne/cm; (13)Density: 0.872 g/cm3; (14)Flash Point: 10 °C; (15)Enthalpy of Vaporization: 33.48 kJ/mol; (16)Boiling Point: 110.602 °C at 760 mmHg; (17)Vapour Pressure: 27.7119998931885 mmHg at 25°C
Preparation of Toluene: Toluene can be produced by many methods.
1. It is derived from coal tar, and commercial grades usually contain small amounts of benzene as an impurity.
2. It can be obtained by benzene from Friedel–Crafts reaction. Benzene reacts with CH3+ and AlCl4- which are got by CH3Cl and AlCl3 to form toluene. In this reaction, the AlCl3 is used as catalyst.
CH3Cl + AlCl3 → CH3+ + AlCl4-
C6H5H + CH3+ + AlCl4- → C6H5CH3 + HCl + AlCl3
3. It can be obtained by cresol from distillation with zinc dust.
C6H4CH3OH (cresol) + Zn → C6H5CH3 (toluene) + ZnO
4. It can be obtained by bromobenzene from Wurtz-Fittig reaction. When bromobenzene and methyl bromide react with sodium metal in dry ether solution, toluene is obtained.
C6H5Br + CH3Br + 2Na → C6H5CH3 + 2NaBr
5. It can be obtained by decarboxylation from toluic acid. Heat the sodium salt of toluic acid (o-, m-, p-) with soda lime, then toluene is synthesized.
C6H4CH3COONa (sodium toluate) + NaOH → C6H5CH3 (toluene) + Na2CO3
Uses of Toluene:
1. Toluene is not only widely used as organic solvent, but also used as raw material to synthetize pharmaceuticals, paints, resins, dyes, explosives, pesticides ansd so on. Moreover, it used as standard material for chromatography and analytical reagent.
2. Toluene is also used as raw material in production of benzene and many other chemical products. For example: it can react with tert-butyl hydroperoxide to get benzyl-tert-butyl peroxide. This reaction needs reagent RuIII-Montmorillonite K10 and solvent acetonitrile by heating. The reaction time is 4 hours. The yield is 60%.
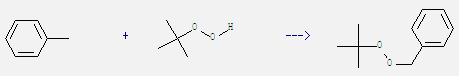
3. It is used as leaching agent of plant components. Used in internal combustion engines, toluene can be used as an octane booster in gasoline fuels. And it has also been used as a coolant for its good heat transfer capabilities in sodium cold traps used in nuclear reactor system loops.
When you are using this chemical, please be cautious about it as the following: Toluene is toxic by inhalation, in contact with skin and if swallowed. And it also has danger of very serious irreversible effects. The vapours of toluene may cause drowsiness and dizziness. If you want to contact this product, you must wear suitable protective clothing and gloves. In case of accident or if you feel unwell, seek medical advice immediately (show the label whenever possible.) If swallowed, do not induce vomitting; seek medical advice immediately and show this container or label. So toluene should be sealed in the container and stored in the cool and dry place which must be away from oxidant.
You can still convert the following datas into molecular structure:
1.Smiles:c1(ccccc1)C
2.InChI:InChI=1S/C7H8/c1-7-5-3-2-4-6-7/h2-6H,1H3
The toxicity data is as follows:
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| frog | LDLo | subcutaneous | 920mg/kg (920mg/kg) | AUTONOMIC NERVOUS SYSTEM: OTHER (DIRECT) PARASYMPATHOMIMETIC BEHAVIORAL: CONVULSIONS OR EFFECT ON SEIZURE THRESHOLD BEHAVIORAL: CHANGES IN MOTOR ACTIVITY (SPECIFIC ASSAY) | Naunyn-Schmiedeberg's Archiv fuer Experimentelle Pathologie und Pharmakologie. Vol. 130, Pg. 250, 1928. |
| guinea pig | LCLo | inhalation | 1600ppm (1600ppm) | BEHAVIORAL: GENERAL ANESTHETIC BEHAVIORAL: SOMNOLENCE (GENERAL DEPRESSED ACTIVITY) BEHAVIORAL: IRRITABILITY | Journal of Industrial Hygiene. Vol. 10, Pg. 261, 1928. |
| guinea pig | LD50 | intraperitoneal | 500mg/kg (500mg/kg) | Neurotoxicology. Vol. 2, Pg. 567, 1981. | |
| human | LDLo | oral | 50mg/kg (50mg/kg) | Gekkan Yakuji. Pharmaceuticals Monthly. Vol. 22, Pg. 883, 1980. | |
| human | TCLo | inhalation | 200ppm (200ppm) | BRAIN AND COVERINGS: RECORDINGS FROM SPECIFIC AREAS OF CNS BLOOD: CHANGES IN BONE MARROW NOT INCLUDED ABOVE BEHAVIORAL: ANTIPSYCHOTIC | JAMA, Journal of the American Medical Association. Vol. 123, Pg. 1106, 1943. |
| mammal (species unspecified) | LC50 | inhalation | 30gm/m3 (30000mg/m3) | Gigiena Truda i Professional'nye Zabolevaniya. Labor Hygiene and Occupational Diseases. Vol. 32(10), Pg. 25, 1988. | |
| mammal (species unspecified) | LD50 | oral | 4gm/kg (4000mg/kg) | Gigiena Truda i Professional'nye Zabolevaniya. Labor Hygiene and Occupational Diseases. Vol. 32(10), Pg. 25, 1988. | |
| mammal (species unspecified) | LDLo | intraperitoneal | 1750mg/kg (1750mg/kg) | BEHAVIORAL: MUSCLE CONTRACTION OR SPASTICITY) LUNGS, THORAX, OR RESPIRATION: OTHER CHANGES | American Journal of Hygiene. Vol. 7, Pg. 276, 1927. |
| man | LDLo | oral | 719uL/kg (0.719mL/kg) | CARDIAC: OTHER CHANGES LUNGS, THORAX, OR RESPIRATION: ACUTE PULMONARY EDEMA LIVER: OTHER CHANGES | Forensic Science International. Vol. 41, Pg. 255, 1989. |
| man | TCLo | inhalation | 100ppm (100ppm) | BEHAVIORAL: "HALLUCINATIONS, DISTORTED PERCEPTIONS" BEHAVIORAL: CHANGES IN MOTOR ACTIVITY (SPECIFIC ASSAY) BEHAVIORAL: CHANGES IN PSYCHOPHYSIOLOGICAL TESTS | Work, Environment, Health. Vol. 9, Pg. 131, 1972. |
| mouse | LC50 | inhalation | 400ppm/24H (400ppm) | Neurotoxicology. Vol. 2, Pg. 567, 1981. | |
| mouse | LD50 | intraperitoneal | 59mg/kg (59mg/kg) | Neurotoxicology. Vol. 2, Pg. 567, 1981. | |
| mouse | LD50 | subcutaneous | 2250mg/kg (2250mg/kg) | Neurotoxicology. Vol. 8, Pg. 237, 1987. | |
| mouse | LD50 | unreported | 2gm/kg (2000mg/kg) | Gigiena i Sanitariya. For English translation, see HYSAAV. Vol. 45(12), Pg. 64, 1980. | |
| rabbit | LCLo | inhalation | 55000ppm/40M (55000ppm) | Journal of Industrial Hygiene and Toxicology. Vol. 26, Pg. 69, 1944. | |
| rabbit | LD50 | skin | 14100uL/kg (14.1mL/kg) | American Industrial Hygiene Association Journal. Vol. 30, Pg. 470, 1969. | |
| rabbit | LDLo | intravenous | 130mg/kg (130mg/kg) | Haematologica. Vol. 58, Pg. 491, 1973. | |
| rat | LC50 | inhalation | 49gm/m3/4H (49000mg/m3) | Gigiena Truda i Professional'nye Zabolevaniya. Labor Hygiene and Occupational Diseases. Vol. 32(10), Pg. 23, 1988. | |
| rat | LD50 | intraperitoneal | 1332mg/kg (1332mg/kg) | Environmental Research. Vol. 40, Pg. 411, 1986. | |
| rat | LD50 | intravenous | 1960mg/kg (1960mg/kg) | Medicina del Lavoro. Industrial Medicine. Vol. 54, Pg. 486, 1963. | |
| rat | LD50 | oral | 636mg/kg (636mg/kg) | Neurotoxicology. Vol. 2, Pg. 567, 1981. | |
| rat | LD50 | unreported | 6900mg/kg (6900mg/kg) | Gigiena i Sanitariya. For English translation, see HYSAAV. Vol. 45(12), Pg. 64, 1980. |
Related Products
- Toluene
- Toluene ethylsulfonamide
- Toluene, m-(diacetoxyiodo)-
- Toluene-2,3-diamine
- Toluene-2,4,6-triyl triisocyanate
- Toluene-2,4,6-triyltriamine
- Toluene-2,4-dicarbamicacid, bis(2,2,3,3,4,4,5,5,6,6,7,7,8,8,9,9,10,10,11,11-eicosafluoroundecyl)ester (8CI)
- Toluene-2,4-dicarbamicacid, bis(o-fluorophenyl) ester (8CI)
- Toluene-2,4-disulfonic acid, {.alpha.-[4-(ethylamino)-m-tolyl]-.alpha.-[4-(ethylimino)-3-methyl-} 2,5-cyclohexadien-1-ylidene]-5-hydroxy-, monosodium salt
- Toluene-2-sulphonic acid
- 108887-44-1
- 108-89-4
- 108894-40-2
- 1088965-37-0
- 1088994-18-6
- 1088994-22-2
- 108900-05-6
- 108902-83-6
- 108906-15-6
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View