-
Name
Tributylphosphine
- EINECS 213-651-2
- CAS No. 998-40-3
- Article Data94
- CAS DataBase
- Density 0.81 g/mL at 25 °C(lit.)
- Solubility Insoluble in water. Miscible with ether, methanol, ethanol and benzene.
- Melting Point -65 °C
- Formula C12H27P
- Boiling Point 244.8 °C at 760 mmHg
- Molecular Weight 202.32
- Flash Point 112.2 °C
- Transport Information UN 3254
- Appearance colourless liquid
- Safety 41-36/37/39-26-16-45-43-17-5
- Risk Codes 36/38-34-21/22-17-11-37
-
Molecular Structure
-
Hazard Symbols
 C;
C;  F;
F;  Xi
Xi
- Synonyms Phosphine, tributyl-;Tributylfosfin;Tri-n-butylphosphine;
- PSA 13.59000
- LogP 4.86860
Synthetic route

-
-
998-40-3
tributylphosphine

| Conditions | Yield |
|---|---|
| With methanol; 4 A molecular sieve In 1,4-dioxane at 115℃; for 28h; | 100% |

| Conditions | Yield |
|---|---|
| Stage #1: n-Butyl chloride With magnesium; methyl iodide In tetrahydrofuran; 5,5-dimethyl-1,3-cyclohexadiene at 40 - 50℃; for 2.7h; Inert atmosphere; Stage #2: With phosphorus trichloride In tetrahydrofuran; 5,5-dimethyl-1,3-cyclohexadiene at 5℃; for 4h; Temperature; Inert atmosphere; | 90.3% |
-
-
55425-72-4
(2,2'-bipyridyl)(1,5-cyclooctadiene)nickel

-
-
13165-72-5
chlorosulfite de methyle

-
A
-
121837-93-2
(bipy)Ni(Cl)(metallasulfinat-Me)

-
B
-
998-40-3
tributylphosphine

-
C
-
5259-72-3, 10060-40-9, 111-78-4
1,5-dicyclooctadiene

| Conditions | Yield |
|---|---|
| In tetrahydrofuran inert atmosphere; cooling soln. of Ni-compd. (-78°C), addn. of org. compd., stirring (room temp.); filtn., washing (hexane), drying; elem. anal.; | A 83% B n/a C n/a |

-
-
72315-65-2
tributyl(methylthio)phosphonium trifluoromethanesulfonate

-
-
998-40-3
tributylphosphine

| Conditions | Yield |
|---|---|
| With Hexamethylphosphorous triamide In dichloromethane | 80% |

| Conditions | Yield |
|---|---|
| With phenylsilane In benzene-d6 at 100℃; for 6h; Kinetics; Temperature; Time; Solvent; Inert atmosphere; Sealed tube; | 72% |
| Stage #1: Tributylphosphine oxide With [Fe(CO)(1,3-bis(diphenylphosphino)propane)H(NO)]; phenylsilane; N-ethyl-N,N-diisopropylamine In toluene at 100℃; for 18h; Schlenk technique; Stage #2: With sodium hydroxide In methanol; water; toluene at 20℃; for 1h; Schlenk technique; chemoselective reaction; | 55% |
| With 1,1,3,3-Tetramethyldisiloxane; titanium(IV) isopropylate In various solvent(s) at 100℃; for 10h; |

| Conditions | Yield |
|---|---|
| With phosphorous; tris(2,2'-bipyridine)nickel(II) tetrafluoroborate In N,N-dimethyl-formamide at 50℃; Electrochemical reaction; Mg anode; | A 11% B 55% |
| With phosphorous In N,N-dimethyl-formamide Electrochemical reaction; Mg anode; | A 11% B 55% |
| With phosphorus; potassium hydroxide; hydrazine hydrate; N-benzyl-N,N,N-triethylammonium chloride In toluene at 80 - 85℃; for 5h; | A 6% B 31% |
-
-
17343-82-7
tri-n-butyl(ethoxycarbonylmethylene)phosphorane

-
A
-
998-40-3
tributylphosphine

-
B
-
814-29-9
Tributylphosphine oxide

-
C
-
623-91-6
diethyl Fumarate

| Conditions | Yield |
|---|---|
| With water; 1-fluoro-2,4,6-trimethylpyridinium trifluoromethanesulfonate In tetrahydrofuran -30 deg C, 30 min; -30 deg C to RT; | A n/a B n/a C 48% |

-
-
51266-28-5
(Bu3P)2Ni(cyclo-octa-1,5-diene)

-
-
13360-57-1
dimethylamino sulfonyl chloride

-
B
-
998-40-3
tributylphosphine

-
C
-
5259-72-3, 10060-40-9, 111-78-4
1,5-dicyclooctadiene

| Conditions | Yield |
|---|---|
| In tetrahydrofuran inert atmosphere; cooling soln. of Ni-compd. (-78°C), addn. of sulfonyl chloride, stirring (room temp.); filtn., washing (THF), drying; elem. anal.; | A 40% B n/a C n/a |

-
-
51266-28-5
(Bu3P)2Ni(cyclo-octa-1,5-diene)

-
-
98-09-9
benzenesulfonyl chloride

-
B
-
998-40-3
tributylphosphine

-
C
-
5259-72-3, 10060-40-9, 111-78-4
1,5-dicyclooctadiene

| Conditions | Yield |
|---|---|
| In tetrahydrofuran inert atmosphere; cooling soln. of Ni-compd. (-78°C), addn. of sulfonyl chloride, stirring (room temp.); filtn., washing (THF), drying; elem. anal.; | A 39% B n/a C n/a |

-
-
51266-28-5
(Bu3P)2Ni(cyclo-octa-1,5-diene)

-
-
124-63-0
methanesulfonyl chloride

-
B
-
998-40-3
tributylphosphine

-
C
-
5259-72-3, 10060-40-9, 111-78-4
1,5-dicyclooctadiene

| Conditions | Yield |
|---|---|
| In tetrahydrofuran inert atmosphere; cooling soln. of Ni-compd. (-78°C), addn. of sulfonyl chloride, stirring (room temp.); filtn., washing (THF), drying; elem. anal.; | A 36% B n/a C n/a |

-
-
1332456-13-9
tributyl(4-ethoxy-3-methyl-4-oxobut-1-en-2-yl)phosphonium trifluoroacetate

-
-
672-66-2
Dimethyl(phenyl)phosphine

-
A
-
998-40-3
tributylphosphine

-
B
-
1332456-56-0
C2F3O2(1-)*C15H22O2P(1+)

-
E
-
1332456-55-9
C2F3O2(1-)*C19H38O2P(1+)

| Conditions | Yield |
|---|---|
| With 1,8-diazabicyclo[5.4.0]undec-7-ene In chloroform-d1 at 20℃; for 1h; optical yield given as %de; | A 26% B n/a C n/a D 35% E 8% |

-
-
51266-28-5
(Bu3P)2Ni(cyclo-octa-1,5-diene)

-
-
98-59-9
p-toluenesulfonyl chloride

-
B
-
998-40-3
tributylphosphine

-
C
-
5259-72-3, 10060-40-9, 111-78-4
1,5-dicyclooctadiene

| Conditions | Yield |
|---|---|
| In tetrahydrofuran inert atmosphere; cooling soln. of Ni-compd. (-78°C), addn. of sulfonyl chloride, stirring (room temp.); filtn., washing (THF), drying; elem. anal.; | A 33% B n/a C n/a |

-
-
121719-01-5
(N,N,N',N'-tetramethylethylenediamine)Ni(1,5-cyclooctadiene)

-
-
13165-72-5
chlorosulfite de methyle

-
A
-
121986-10-5
(TMED)Ni(Cl)(metallasulfinat-Me)

-
B
-
998-40-3
tributylphosphine

-
C
-
5259-72-3, 10060-40-9, 111-78-4
1,5-dicyclooctadiene

| Conditions | Yield |
|---|---|
| In tetrahydrofuran inert atmosphere; cooling soln. of Ni-compd. (-78°C), addn. of org. compd., stirring (room temp.); filtn., pptn. on addn. of hexane, washing, drying; elem. anal.; | A 4% B n/a C n/a |


| Conditions | Yield |
|---|---|
| With phosphan |

| Conditions | Yield |
|---|---|
| With phosphorus trichloride | |
| With diethyl ether; phosphorus trichloride at 0℃; zum Schluss auf dem Wasserbad; |
-
-
60-29-7
diethyl ether

-
-
58758-29-5
tributylphosphine-carbon disulfide adduct

-
A
-
75-15-0
carbon disulfide

-
B
-
998-40-3
tributylphosphine

-
-
64-17-5
ethanol

-
-
58758-29-5
tributylphosphine-carbon disulfide adduct

-
A
-
75-15-0
carbon disulfide

-
B
-
998-40-3
tributylphosphine


| Conditions | Yield |
|---|---|
| With phosphorus trichloride |
-
-
53130-29-3
α-Tri-n-butylphosphonium-phenylisothiocyanat

-
A
-
998-40-3
tributylphosphine

-
B
-
103-72-0
phenyl isothiocyanate

| Conditions | Yield |
|---|---|
| In acetone at 24℃; Equilibrium constant; other solvents; |


| Conditions | Yield |
|---|---|
| With ammonia; sodium at -78℃; for 3h; | 100 % Chromat. |
| With [AlH3(triethylamine)] In hexane at 20℃; for 0.166667h; Inert atmosphere; Schlenk technique; | 92 %Chromat. |

| Conditions | Yield |
|---|---|
| With W2Cl4(PBu3)4 In (2)H8-toluene Thermodynamic data; ΔH(excit.); ΔS(excit.); |
-
-
106-98-9
1-butylene

-
A
-
1732-74-7
n-butylphosphine

-
B
-
1732-72-5
di-n-butylphosphine

-
C
-
998-40-3
tributylphosphine

| Conditions | Yield |
|---|---|
| at 25℃; UV-Licht.Irradiation; |

-
-
106-98-9
1-butylene

-
-
110-05-4
di-tert-butyl peroxide

-
A
-
1732-74-7
n-butylphosphine

-
B
-
1732-72-5
di-n-butylphosphine

-
C
-
998-40-3
tributylphosphine

| Conditions | Yield |
|---|---|
| at 122℃; |


| Conditions | Yield |
|---|---|
| With W2Cl4(PBu3)4 In (2)H8-toluene Thermodynamic data; ΔH(excit.); ΔS(excit.); |
-
-
201230-82-2
carbon monoxide

-
A
-
998-40-3
tributylphosphine

| Conditions | Yield |
|---|---|
| With <(OC)4Mn(μ-PEt2)2Mn(CO)3(PnBu3)> In decalin at 100℃; under 832.6 Torr; Equilibrium constant; var. temp.; |

| Conditions | Yield |
|---|---|
| With phosphan Irradiation.UV-Bestrahlung; |

| Conditions | Yield |
|---|---|
| With dihydrogen peroxide In dichloromethane; water at 20℃; for 3h; Glovebox; Inert atmosphere; | 100% |
| With polymeric complex of oxodiperoxochromium(VI) compound and pyrazine (Pyz-CrO5)n In dichloromethane for 0.15h; Ambient temperature; | 99% |
| With n-butyllithium; dinitrogen monoxide; 1,3-bis[(diphenylphosphino)propane]dichloronickel(II) In hexane; toluene at 20℃; under 760 Torr; for 0.5h; Oxidation; | 98% |
-
-
4360-63-8
2-bromomethyl-1,3-dioxolane

-
-
998-40-3
tributylphosphine

-
-
115754-62-6
(1,3-dioxolan-2-ylmethyl)tributylphosphonium bromide

| Conditions | Yield |
|---|---|
| at 90℃; for 96h; | 100% |
| at 90℃; for 72h; Yield given; |
-
-
998-40-3
tributylphosphine

-
-
84387-62-2
1-chloromethyl-1H-1,2,4-triazole

| Conditions | Yield |
|---|---|
| In acetonitrile for 16h; Heating; | 100% |
-
-
998-40-3
tributylphosphine

-
-
139462-37-6
[(R)-3-(4-Bromomethyl-oxazol-2-yl)-butyl]-carbamic acid tert-butyl ester

-
-
139461-92-0
[2-((R)-3-tert-Butoxycarbonylamino-1-methyl-propyl)-oxazol-4-ylmethyl]-tributyl-phosphonium; bromide

| Conditions | Yield |
|---|---|
| In N,N-dimethyl-formamide at 60℃; | 100% |
-
-
998-40-3
tributylphosphine

-
-
39181-26-5
tri-n-butylphosphine selenide

| Conditions | Yield |
|---|---|
| With 4-phenyl-1,2,3-selenadiazole In benzene at 20℃; for 1h; | 100% |
| With selenium In toluene for 0.5h; Heating; Yield given; | |
| With diphenyl diselenide; palladium diacetate for 0.25h; microwave irradiation; |
-
-
998-40-3
tributylphosphine

-
-
204378-94-9
methyl (E)-5-bromo-2-<(E)-ethylidene>-4-methylpent-3-enoate

| Conditions | Yield |
|---|---|
| In acetonitrile Heating; | 100% |
| In acetonitrile for 0.333333h; Heating; Yield given; |
-
-
998-40-3
tributylphosphine

| Conditions | Yield |
|---|---|
| In acetonitrile at 0℃; for 13h; | 100% |
-
-
998-40-3
tributylphosphine

-
-
380909-81-9
4-((2R,4R,6R)-4-(tert-Butyl-dimethyl-silanyloxy)-6-{(2R,6R)-6-[2-(tert-butyl-diphenyl-silanyloxy)-ethyl]-4-methylene-tetrahydro-pyran-2-ylmethyl}-tetrahydro-pyran-2-yl)-2-chloromethyl-oxazole

| Conditions | Yield |
|---|---|
| In N,N-dimethyl-formamide for 16h; | 100% |
| 93% |
-
-
998-40-3
tributylphosphine

-
-
28868-76-0
2-chlorodimethylmalonate

| Conditions | Yield |
|---|---|
| Stage #1: tributylphosphine; 2-chlorodimethylmalonate In tetrahydrofuran at 20℃; for 0.0333333h; Argon atmosphere; Stage #2: With triethylamine In tetrahydrofuran for 1h; Argon atmosphere; | 100% |
| With triethylamine In ethyl acetate at 20℃; for 1h; | 98.6% |
-
-
998-40-3
tributylphosphine

-
-
56-93-9
benzyltrimethylammonium chloride

-
-
1224-56-2
tributylbenzylphosphonium chloride

| Conditions | Yield |
|---|---|
| at 100℃; for 0.166667h; Irradiation; | 100% |

| Conditions | Yield |
|---|---|
| at 20 - 150℃; for 11h; | 100% |

| Conditions | Yield |
|---|---|
| at 60 - 150℃; for 7h; | 100% |
-
-
874817-67-1
dimethyloxonium tris(trifluoromethyl)borate

-
-
998-40-3
tributylphosphine

| Conditions | Yield |
|---|---|
| In dichloromethane at 20℃; | 100% |
| In dichloromethane at 20℃; | 100% |
-
-
998-40-3
tributylphosphine

-
-
199620-14-9, 13007-92-6
chromium(0) hexacarbonyl

-
-
18497-59-1
Cr(CO)5(P(n-Bu)3)

| Conditions | Yield |
|---|---|
| In diethylene glycol dimethyl ether mole ratio of educts 1.1, in boiling diglyme;; | 100% |
| In diethylene glycol dimethyl ether mole ratio of educts 1.1, in boiling diglyme;; | 100% |
| In decalin Kinetics; at 130.7 °C; |

-
-
998-40-3
tributylphosphine

-
-
97671-41-5
mercury bis(phenyltellurolate)

| Conditions | Yield |
|---|---|
| In dichloromethane molar ratio of Hg(PBu3)2(ClO4)2/Hg(TePh)2/PBu3 = 1:3:2; not isolated; | 100% |

-
-
12146-37-1, 124717-04-0
(bicyclo[2.2.1]hepta-2,5-diene)tetracarbonylmolybdenum(0)

-
-
998-40-3
tributylphosphine

-
-
15748-70-6, 16244-54-5, 17652-79-8
cis-{molybdenum(0)(carbonyl)4(P(n-butyl)3)2}

-
B
-
121-46-0
bicyclo[2.2.1]hepta-2,5-diene

| Conditions | Yield |
|---|---|
| In tetrahydrofuran reaction in a calorimeter under argon; | A 100% B n/a |


| Conditions | Yield |
|---|---|
| In dichloromethane N2 atmosphere; addn. of phosphine to Rh complex, stirring (30 min., room temp.); evapn. (vac.), recrystn. (CH2Cl2/hexane); | 100% |
-
-
122677-54-7
fac-Re(CO)3(N,N'-dimethylpiperazine-2,3-dithione)Br

-
-
998-40-3
tributylphosphine

-
-
2923-28-6
silver trifluoromethanesulfonate

| Conditions | Yield |
|---|---|
| In tetrahydrofuran An equimolar amt. of AgOTF was added to the soln. of the rhenium complex. After filtn. excess P(nBu)3 was added to the filtrate under exclusion of light.; | 100% |


| Conditions | Yield |
|---|---|
| In tetrahydrofuran N2; instantaneous reaction;; after 4 h filtration; washing with THF; elem. anal.;; | 100% |
-
-
998-40-3
tributylphosphine

-
-
261359-74-4, 260995-06-0, 261359-76-6
[Ru(η5,η1-C5H2Me2CO2(CH2)2PPh2)(CH3CN)2][PF6]

-
-
342643-71-4, 340269-30-9
[(CH3(C5H2)CH3)Ru(CH3CN)P(C4H9)3COOCH2CH2P(C6H5)2](1+)*PF6(1-)=[(CH3(C5H2)CH3)Ru(CH3CN)P(C4H9)3COOCH2CH2P(C6H5)2]PF6

| Conditions | Yield |
|---|---|
| In acetone 1:1.1 mixt. stirred at room temp. for 3 h; NMR; | 100% |
-
-
998-40-3
tributylphosphine

-
-
260995-15-1, 380606-14-4
[Ru(η5,η1-C5H2(Me)(t-Bu)CO2(CH2)2PPh2)(CH3CN)2][PF6]

| Conditions | Yield |
|---|---|
| In acetone 1:1.1 mixt. stirred at room temp. for 3 h; NMR; | 100% |
-
-
998-40-3
tributylphosphine

-
-
188797-37-7
carbonyl(tetraphenyltetrabenzoporphyrinato)ruthenium(II)

-
-
188797-48-0
bis(tri-n-butylphosphine)(tetraphenyltetrabenzoporphyrinato)ruthenium(II)

| Conditions | Yield |
|---|---|
| In dichloromethane detd. by UV / Vis and IR spectroscopy; | 100% |
| In chloroform-d1 detd. by (1)H NMR spectroscopy; | 100% |
-
-
998-40-3
tributylphosphine

-
-
15841-27-7
silver 1,3-diphenyl-1,3-propandionate

-
-
879555-31-4
(nBu3P)Ag(1,3-diphenyl-1,3-propanedionate)

| Conditions | Yield |
|---|---|
| In diethyl ether under N2, Ag-compd. was dissolved in Et2O, 1 equiv. of P-compd. was added, stirring at 25 °C for 2 h; soln. was filtered through silica, volatiles were removed in vac., elem.anal.; | 100% |
| In dichloromethane under N2, 1 equiv. of P-compd., 25 °C; |
-
-
998-40-3
tributylphosphine

-
-
175551-77-6
methyl 2-(bromomethyl)oxazole-4-carboxylate

-
-
1222085-97-3
Br(1-)*C18H33NO3P(1+)

| Conditions | Yield |
|---|---|
| In acetonitrile at 20℃; for 1.5h; | 100% |

| Conditions | Yield |
|---|---|
| In diethyl ether (Ar), Ag acetate suspnd. in Et2O, reacted with 3 equiv. of (n-Bu)3P at 25°C for 1 h under stirring; evapd., elem. anal.; | 100% |
-
-
998-40-3
tributylphosphine

-
-
563-63-3
silver(I) acetate

-
-
1224594-74-4
tetra(tri-n-butyl phosphine)silver(I) acetate

| Conditions | Yield |
|---|---|
| In diethyl ether (Ar), Ag acetate suspnd. in Et2O, reacted with 4 equiv. of (n-Bu)3P at 25°C for 1 h under stirring; evapd., elem. anal.; | 100% |
-
-
998-40-3
tributylphosphine

-
-
5398-64-1
2-benzylidene-1,3-diphenylpropane-1,3-dione

-
-
1233127-23-5
C34H44O2P(1+)*Cl(1-)

| Conditions | Yield |
|---|---|
| With hydrogenchloride In 1,4-dioxane at 27 - 30℃; for 0.0166667h; Inert atmosphere; | 100% |
-
-
110-57-6
trans-1,4-dichlorobut-2-ene

-
-
998-40-3
tributylphosphine

-
-
36288-32-1, 134839-26-2
(E)-but-2-ene-1,4-diylbis(tributylphosphonium) dichloride

| Conditions | Yield |
|---|---|
| In 5,5-dimethyl-1,3-cyclohexadiene at 20℃; for 18h; Inert atmosphere; Reflux; | 100% |
-
-
998-40-3
tributylphosphine

-
-
1533434-49-9
(E)-((6-bromohex-4-en-1-yl)oxy)(tertbutyl)dimethylsilane

-
-
1533434-50-2
C24H52OPSi(1+)*Br(1-)

| Conditions | Yield |
|---|---|
| In acetonitrile at 20℃; for 12h; | 100% |
Tributylphosphane Chemical Properties
Molecular Structure:
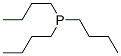
Molecular Formula: C12H27P
Molecular Weight: 202.316541 g/mol
Appearance: colorless pale yellow clear liquid
Melting Point: -65 °C
Boiling Point: 150 °C at 50 mm Hg
Flash Point: 99 °F
Solubility: methanol, ether, ethanol, benzene
Insolubility: water
Density: 0.81 g/mL at 25 °C
Vapor density: 9
Refractive index:n: 20/D 1.462
Storage temp.: 2-8°C
Sensitive: Moisture, Air
BRN: 1738261
EINECS: 213-651-2
IUPAC Name: tributylphosphane
Synonyms of Tributylphosphine (CAS NO.998-40-3): TRIBUTYLPHOSPHINE(998-40-3): Tri-n-butylphosphine ; 4-16-00-01699 (Beilstein Handbook Reference) ; AI3-28071 ; BRN 1738261 ; EINECS 213-651-2 ; NSC 91700 ; Tri-n-butylphosphine ; Tributylfosfin ; Tributylfosfin [Czech] ; Tributylphosphane ; Tributylphosphane [UN3254] [Spontaneously combustible] ; Tris(butyl)phosphine ; UN3254 ; Phosphine, tributyl-
Categories: Tertiary Phosphines;Ligand;Mitsunobu Reaction;Phosphine Ligands;Phosphines (Mitsunobu Reaction);Synthetic Organic Chemistry
Tributylphosphane Uses
Tributylphosphine (CAS NO.998-40-3) is used as main ligand of homogeneous catalyst in the petrochemical. It is also used as reducing agent, olefin polymerization catalyst.
Tributylphosphane Production
Tributylphosphine (CAS NO.998-40-3) is prepared industrially by the reaction of phosphine with butene.
PH3 + 3CH2=CHCH2CH3 → P(CH2CH2CH2CH3)3
It can be prepared in the laboratory by reaction of the appropriate Grignard reagent with phosphorus trichloride.
3BuMgCl + PCl3 → PBu3 + 3MgCl2
Tributylphosphane Toxicity Data With Reference
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| rabbit | LD50 | skin | 2gm/kg (2000mg/kg) | "Toxicology of Drugs and Chemicals," Deichmann, W.B., New York, Academic Press, Inc., 1969Vol. -, Pg. 600, 1969. | |
| rat | LD50 | oral | 750mg/kg (750mg/kg) | "Toxicology of Drugs and Chemicals," Deichmann, W.B., New York, Academic Press, Inc., 1969Vol. -, Pg. 600, 1969. |
Tributylphosphane Consensus Reports
Reported in EPA TSCA Inventory.
Tributylphosphane Safety Profile
Safety Information of Tributylphosphine (CAS NO.998-40-3):
Hazard Codes:Xi  ,F
,F  ,C
,C 
Risk Statements:36/38-34-21/22-17-11-37
36/38:Irritating to eyes and skin
11:Highly flammable
17:Spontaneously flammable in air
21/22:Harmful in contact with skin and if swallowed
34:Causes burns
Safety Statements:41-36/37/39-26-16-45-43-17-5
41:In case of fire and/or explosion do not breathe fumes
16:Keep away from sources of ignition - No smoking
26:In case of contact with eyes, rinse immediately with plenty of water and seek medical advice
36/37/39:Wear suitable protective clothing, gloves and eye/face protection
43:In case of fire, use ... (indicate in the space the precise type of fire-fighting equipment. If water increases the risk add - Never use water)
45:In case of accident or if you feel unwell, seek medical advice immediately (show label where possible)
17:Keep away from combustible material
5:Keep contents under ... (appropriate liquid to be specified by the manufacturer)
RIDADR:UN 3254 4.2/PG 1
WGK Germany:3
RTECS:SZ3270000
F:10-13-23
HazardClass:4.2
PackingGroup:I
Moderately toxic by ingestion. Slightly toxic by skin contact. When heated to decomposition it emits toxic vapors of POx.
Tributylphosphane Specification
First Aid Measures of Tributylphosphine (CAS NO.998-40-3):
Eyes:Immediately flush eyes with plenty of water for at least 15 minutes, occasionally lifting the upper and lower eyelids. Get medical aid.
Skin:Get medical aid. Flush skin with plenty of water for at least 15 minutes while removing contaminated clothing and shoes.
Ingestion:Do NOT induce vomiting. If conscious and alert, rinse mouth and drink 2-4 cupfuls of milk or water.
Inhalation:Remove from exposure and move to fresh air immediately. If not breathing, give artificial respiration. If breathing is difficult, give oxygen. Get medical aid.
Storage of Tributylphosphine (CAS NO.998-40-3):
Keep away from heat, sparks, and flame. Do not store near combustible materials. Store in a cool, dry place. Store in a tightly closed container. Flammables-area. Store under nitrogen.
Related Products
- Tributylphosphane
- 99844-02-7
- 99846-86-3
- 99846-93-2
- 99-85-4
- 99854-17-8
- 99857-17-7
- 99857-72-4
- 99-86-5
- 99873-43-5
- 99-87-6
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View