Simagchem Corporation
Welcome to Simagchem, your partner in China as a premier supply of bulk specialty chemicals for industry and life science. We introduce experienced quality product and exceptional JIT service with instant market intelligence in China to benefit our
Cas:1071-46-1
Min.Order:0 Metric Ton
Negotiable
Type:Manufacturers
inquiryDayang Chem (Hangzhou) Co.,Ltd.
DayangChem exported this product to many countries and regions at best price. If you are looking for the material’s manufacturer or supplier in China, DayangChem is your best choice. Pls contact with us freely for getting detailed product s
Cas:1071-46-1
Min.Order:1 Kilogram
FOB Price: $3.0
Type:Lab/Research institutions
inquiryChemwill Asia Co., Ltd.
Our main production base is located in Xuzhou industry park. We are certified both to the ISO 9001 and ISO 14001 Standards, have a safety management system in place.Our R&D team masters core technology for process-design of target building block
Cas:1071-46-1
Min.Order:5 Kiloliter
FOB Price: $1.2 / 5.0
Type:Manufacturers
inquiryLIDE PHARMACEUTICALS LIMITED
LIDE PHARMACEUTICALS LIMITED is a professional chemicals and APIs leading manufacturer in China. Our core business line covers APIs, Intermediates, Herb extract, etc. We are specialized in chemical synthesis, process development of pharmaceu
Cas:1071-46-1
Min.Order:1 Gram
FOB Price: $1.0
Type:Lab/Research institutions
inquiryAlity Chemical Corporation
3-Hydroxy-2-naphthoic acid Basic information Product Name: 3-Hydroxy-2-naphthoic acid Synonyms: 3-hydroxy-2-naphthoicaci;3-Hydroxy-beta-naphthoic acid;developer8;developerbon;Kyselina 3-hydroxy-2-naftoova;kyselina3-hydroxy-2-naftoova;Miketazol Deve
Hebei Nengqian Chemical Import and Export Co., LTD
With our good experience, we offer detailed technical support and advice to assist customers. We communicate closely with customers to establish their quality requirements. Consistent Quality Our plant has strict quality control in each manufacturin
Cas:1071-46-1
Min.Order:1 Kilogram
FOB Price: $100.0 / 150.0
Type:Trading Company
inquiryZhuozhou Wenxi import and Export Co., Ltd
WITH US,YOUR MONEY IN SAFE,YOUR BUSINESS IN SAFE 1)Quick Response Within 12 hours; 2)Quality Guarantee: All products are strictly tested by our QC, confirmed by QA and approved by third party lab in China, USA, Canada, Germany, UK, Italy, France et
Cas:1071-46-1
Min.Order:1 Kilogram
FOB Price: $139.0 / 210.0
Type:Trading Company
inquiryHangzhou Sartort Biopharma Co., Ltd
Appearance:Pale yellow liquid Storage:room temperature Package:200kg/drum Application:Ethyl Hydrogen Malonate has been shown to impair brain mitochondrial succinate and malate transport. It is also found in the extract of Hericum erinaceus mushroom
Cas:1071-46-1
Min.Order:1 Kilogram
FOB Price: $1.0
Type:Lab/Research institutions
inquiryShanghai Upbio Tech Co.,Ltd
1.No Less 8 years exporting experience. Clients can 100% received goods 2.Lower Price with higher quality 3,Free sample 4,We are sincerely responsible for the "product quality" and "After Service" Upbio is Specialized
Cas:1071-46-1
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryHenan Wentao Chemical Product Co., Ltd.
Henan Wentao Chemical Product Co.,Ltd is Located in Zhengzhou High-tech Development Zone with import and export license, We passed ISO 9001:2008 as well, Henan Wentao has developed more than 1000 compounds, which are widely used in the fields of prod
Xiamen Hisunny Chemical Co.,Ltd
Best quality & Attractive price & Professional service; Trial & Pilot & Commercial Hisunny Chemical is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality intermediates, specia
Hebei Quanhe Biotechnology Co. LTD
1. Timely and efficient service to ensure communication with customers 2. Produce products of different specifications and sizes according to your requirements. 3. Quality procedures and standards recognized by SGS. Advanced plant equipment ensures
Cas:1071-46-1
Min.Order:1 Kilogram
FOB Price: $30.0 / 50.0
Type:Lab/Research institutions
inquiryHangzhou Keyingchem Co.,Ltd
Hangzhou KeyingChem Co., Ltd. exported this product to many countries and regions at best price. If you are looking for the material’s manufacturer or supplier in China, KeyingChem is your best choice. Pls contact with us freely for getting det
Cas:1071-46-1
Min.Order:0 Metric Ton
Negotiable
Type:Lab/Research institutions
inquiryHangzhou J&H Chemical Co., Ltd.
J&H CHEM R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. J&H CHEM has some Manufacturing base in Jia
Shanghai Massive Chemical Technology Co., Ltd.
Massive Chemical is certified with ISO9001 and ISO14001 manufacturer for this product. We will offer all documents as requirement for the materials which includes, Certificate of Analysis, Material Safety Data Sheet, and Method of Analysis and
Cas:1071-46-1
Min.Order:1 Gram
FOB Price: $1.0
Type:Lab/Research institutions
inquiryHenan Tianfu Chemical Co., Ltd.
Product Name:Ethyl hydrogen malonateSynonyms:ETHYL HYDROGEN MALONATE;MONOETHYL MALONATE;3-ethoxy-3-oxapropanoic acid;Monoethyl malonic acid;3-ethoxy-3-oxo-propanoic acid;Ethyl hydrogen malonate, 90+%;Ethoxycarbonylacetic acid;Malonic acid 1
Cas:1071-46-1
Min.Order:1 Metric Ton
FOB Price: $20.0
Type:Lab/Research institutions
inquirySiwei Development Group Ltd.
Product name: Ethyl Hydrogen Malonate CAS No.: 1071-46-1 Molecule Formula:C5H8O4 Molecule Weight:132.11 Purity: 98.0% Package: 25kg/drum Description:White powder Manufacture Standards:Enterprise Standard TESTING IT
Cas:1071-46-1
Min.Order:1 Kilogram
Negotiable
Type:Trading Company
inquiryXiamen Jenny Chemical Technology Co., Ltd.
GMP standard, high purity, competitive price, in stock 1. Quick Response: within 6 hours after receiving your email. 2. Quality Guarantee: All products are strictly tested by our QC, confirmed by QA, and approved by a third-party lab in China, USA,
Shanghai Minstar Chemical Co., Ltd
Product Name: Ethyl hydrogen malonate Synonyms: ETHYL HYDROGEN MALONATE;MONOETHYL MALONATE;3-ethoxy-3-oxapropanoic acid;Monoethyl malonic acid;3-ethoxy-3-oxo-propanoic acid;Ethyl hydrogen malonate, 90+%;Ethoxycarbonylacetic acid;Malonic acid 1
Cas:1071-46-1
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryHunan chemfish Pharmaceutical co.,Ltd
Appearance:95%+ Package:R&D,Pilot run Transportation:per client require Port:Express ,Air, Sea
Suzhou Health Chemicals Co., Ltd.
High quality,stable supply chain.Appearance:white/off-white or light yellow Storage:Store in cool and dry place, keep away from strong light and heat. Package:aluminum bottle,glass bottle,PTFE bottle,cardboard drum Application:This product can be use
Win-Win chemical Co.Ltd
Stock products, own laboratory Package:Grams, Kilograms Application:For R&D Transportation:According to customer request Port:Shanghai
Cas:1071-46-1
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryKono Chem Co.,Ltd
high purity lowest priceAppearance:solid or liquid Storage:in sealed air resistant place Package:drum and bag Application:for pharma use Transportation:by sea or air Port:Beijing or Guangzhou
Lonwin Chemical Group Limited
Shanghai Lonwin Chemical company is a subsidiary of Lonwin Industry Group Limited, was established in 2011 and is headquartered in Shanghai, adjacent to China National Convention and Exhibition Center and Hongqiao transportation hub.Lonwinchem is
Cas:1071-46-1
Min.Order:100 Kilogram
FOB Price: $100.0 / 150.0
Type:Other
inquiryGIHI CHEMICALS CO.,LIMITED
high purity,in stock Package:25kg/drum,or as per customers'demand Application:API,or Intermediates,fine chemicals Transportation:air,sea,courier
Cas:1071-46-1
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryShanghai Acmec Biochemical Technology Co., Ltd.
Acmec is a leading manufacturer and supplier of biochemical reagents and life science products. We have over 40,000 items in stock (real-time inventory) and offer discounted prices to registered members of the online store ( www.acmec.com.cn ) Appea
Cas:1071-46-1
Min.Order:1 bottle
Negotiable
Type:Lab/Research institutions
inquiryHenan Allgreen Chemical Co.,Ltd
high quality Storage:Sealed, dry, microtherm , avoid light and smell. Package:According to the demand of customer Application:Organic synthesis Transportation:by air or by sea
Aecochem Corp.
Our clients, like BASF,CHEMO,Brenntag,ASR,Evonik,Merck and etc.Appearance:COA Storage:in stock Application:MSDS/TDS
Xi`an Eastling Biotech Co., Ltd.
high purity lowest priceAppearance:solid or liquid Storage:in sealed air resistant place Package:Foil bag; Drum; Plastic bottle Application:Pharma;Industry;Agricultural Transportation:by sea or air Port:Beijing or Guangzhou
Antimex Chemical Limied
Ansciep Chemical is a professional enterprise manufacturing and distributing fine chemicals and speciality chemicals. We have been dedicated to heterocycle compounds and phenyl rings for tens of years. This is our mature product for export. Our quali
Synthetic route

| Conditions | Yield |
|---|---|
| With hydrogenchloride In water at 5℃; for 1h; | 100% |
| With hydrogenchloride In water at 0℃; for 0.666667h; | 99% |
| With hydrogenchloride In water Inert atmosphere; Cooling with ice; | 91% |

| Conditions | Yield |
|---|---|
| With potassium hydroxide In ethanol; water at 25℃; for 1h; | 94% |
| With potassium hydroxide In tetrahydrofuran at 0℃; for 1h; | 90% |
| Stage #1: diethyl malonate With potassium hydroxide; water In tetrahydrofuran at 0℃; for 1h; Stage #2: With hydrogenchloride; water In tetrahydrofuran at 0℃; Product distribution / selectivity; | 90% |

| Conditions | Yield |
|---|---|
| With potassium phosphate buffer; nitrilase from Alcaligenes faecalis ATCC8750 at 30℃; for 7h; pH=7.3; | 94% |
| With benzene-1,2-dicarboxylic acid at 250℃; under 7600 Torr; for 0.25h; microwave irradiation; | 53% |
| With benzene-1,2-dicarboxylic acid at 240℃; under 3040 Torr; for 1h; | 10% |
| With copper(II) sulfate at 240℃; for 1h; | 9% |

| Conditions | Yield |
|---|---|
| With boric acid Heating; | 80% |
| With sulfuric acid | |
| With sulfuric acid In acetone for 168h; | |
| With boric acid for 18h; Reflux; | |
| at 80℃; |

| Conditions | Yield |
|---|---|
| Stage #1: diethyl malonate With water; potassium hydroxide In tetrahydrofuran at 0℃; for 0.5h; Stage #2: In tetrahydrofuran; water Acidic conditions; | A 8.3% B 76.9% |
| Stage #1: diethyl malonate With water; potassium hydroxide at 0℃; for 6h; Stage #2: Acidic aq. solution; | A 36.6% B 35.6% |

| Conditions | Yield |
|---|---|
| With iron(III) perchlorate for 2h; Ambient temperature; | 76% |
-
-
7597-56-0
ethyl malonamate

-
A
-
60-35-5
acetamide

-
B
-
141-82-2
malonic acid

-
C
-
1071-46-1
hydrogen ethyl malonate

-
D
-
64-19-7
acetic acid

| Conditions | Yield |
|---|---|
| With phthalic anhydride at 240 - 250℃; under 3040 Torr; for 1h; Hydrolysis; | A n/a B n/a C 53% D n/a |


| Conditions | Yield |
|---|---|
| With dmap; bis-triphenylphosphine-palladium(II) chloride; pyrrole; triethylamine In water; toluene UV-irradiation; Autoclave; | 52% |

-
A
-
28856-92-0
(Ethoxycarbonyl)methyl 2-pyridyl sulfide

-
B
-
2127-03-9
2,2'-dipyridyldisulphide

-
C
-
1071-46-1
hydrogen ethyl malonate

| Conditions | Yield |
|---|---|
| In benzene photolysis; Irradiation; | A 48% B 3% C 17% |


| Conditions | Yield |
|---|---|
| With tetraethylammonium chloride; silver; magnesium In acetonitrile at 0℃; under 760.051 Torr; Electrolysis; | 38% |

| Conditions | Yield |
|---|---|
| With diethyl ether; ammonia; sodium amide nachfolgendes Behandeln mit festem CO2; | |
| With triphenylmethyl sodium at 0℃; nachfolgendes Behandeln mit festem CO2; |

| Conditions | Yield |
|---|---|
| With potassium hydroxide Heating; |
-
-
16694-80-7
ethyl (ethylthio)carbonylacetate

-
-
1071-46-1
hydrogen ethyl malonate

| Conditions | Yield |
|---|---|
| With potassium hydroxide In ethanol Heating; |

| Conditions | Yield |
|---|---|
| (i) LDA, (ii) /BRN= 385653/; Multistep reaction; |

| Conditions | Yield |
|---|---|
| for 24h; Heating; | |
| In toluene for 2h; Heating; Yield given; | |
| In toluene for 5h; Heating; |

| Conditions | Yield |
|---|---|
| With water pH 8; |

-
-
32864-38-3
tert-Butyl ethyl malonate

-
-
1071-46-1
hydrogen ethyl malonate

| Conditions | Yield |
|---|---|
| With bromocatecholborane In dichloromethane for 18h; Ambient temperature; Yield given; |

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 5℃; electrochemical oxidation; |

| Conditions | Yield |
|---|---|
| Reaktion des Natriumenolats; |

-
-
1071-46-1
hydrogen ethyl malonate

| Conditions | Yield |
|---|---|
| With hydrogenchloride |

| Conditions | Yield |
|---|---|
| Erwaermen; |

-
-
861351-52-2
oxo-ethane-1,1,2-tricarboxylic acid triethyl ester

-
-
7732-18-5
water

-
A
-
1071-46-1
hydrogen ethyl malonate

-
B
-
144-62-7
oxalic acid

-
-
861351-52-2
oxo-ethane-1,1,2-tricarboxylic acid triethyl ester

-
-
7732-18-5
water

-
A
-
1071-46-1
hydrogen ethyl malonate

-
B
-
144-62-7
oxalic acid

| Conditions | Yield |
|---|---|
| Erhitzen; |


| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 91 percent / KOH / ethanol / 1 h / Heating 2: 100 percent / HCl / H2O / 1 h / 5 °C View Scheme |


| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 50 percent / KOH / ethanol 2: 55 percent / conc. HCl / H2O View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: (i) HCl, benzene, (ii) H2O 2: KOH / ethanol / Heating View Scheme |

-
-
43167-10-8
sodium monoethyl malonate

-
-
1071-46-1
hydrogen ethyl malonate

| Conditions | Yield |
|---|---|
| With hydrogenchloride; water pH=3; | 52.2 g |

| Conditions | Yield |
|---|---|
| at -65℃; |
-
-
1071-46-1
hydrogen ethyl malonate

-
-
103-80-0
phenylacetyl chloride

-
-
718-08-1
ethyl 3-oxo-4-phenylbutyrate

| Conditions | Yield |
|---|---|
| Stage #1: hydrogen ethyl malonate With n-butyllithium In tetrahydrofuran; hexane at -78 - -10℃; Stage #2: phenylacetyl chloride In tetrahydrofuran; hexane at -78℃; for 0.166667h; Further stages.; | 100% |
| With [2,2]bipyridinyl; n-butyllithium In tetrahydrofuran; hexane at -65℃; for 0.0833333h; | 85% |
| 75% |

| Conditions | Yield |
|---|---|
| With dicyclohexyl-carbodiimide In dichloromethane at 25℃; for 0.25h; | 100% |
| With dicyclohexyl-carbodiimide In acetonitrile for 2h; | 66% |
| With diphenyl phosphoryl azide; triethylamine | |
| With 2-chloro-1-methyl-pyridinium iodide; benzotriazol-1-ol; N-ethyl-N,N-diisopropylamine In acetonitrile at 20℃; for 2h; | |
| With dicyclohexyl-carbodiimide In acetonitrile at 25℃; for 1h; |
-
-
1071-46-1
hydrogen ethyl malonate

-
-
88419-56-1
2,4,5-trifluorobenzoyl chloride

-
-
98349-24-7
ethyl 2,4,5-trifluorobenzoylacetate

| Conditions | Yield |
|---|---|
| Stage #1: hydrogen ethyl malonate With n-butyllithium; triphenylmethane In tetrahydrofuran Metallation; Stage #2: 2,4,5-trifluorobenzoyl chloride In tetrahydrofuran at 20℃; for 1h; Acetylation; | 100% |
| Stage #1: hydrogen ethyl malonate With n-butyllithium In tetrahydrofuran at -75 - -5℃; for 0.0333333h; Stage #2: 2,4,5-trifluorobenzoyl chloride In tetrahydrofuran at -75 - 20℃; Stage #3: With hydrogenchloride; water In tetrahydrofuran; ethyl acetate | 89% |
| With n-butyllithium; biquinoline 1.) THF, -30 deg C to - 5 deg C, 2.) -50 deg C, 0.5 h; Yield given. Multistep reaction; |
-
-
1071-46-1
hydrogen ethyl malonate

-
-
37517-78-5
magnesium bis(3-ethoxy-3-oxopropanoate)

| Conditions | Yield |
|---|---|
| With magnesium(II) chloride hexahydrate; potassium hydroxide In water Cooling with ice; | 100% |
| With magnesium In tetrahydrofuran for 4h; Heating / reflux; | |
| With magnesium ethylate In tetrahydrofuran at 20℃; for 2.5h; |
-
-
1071-46-1
hydrogen ethyl malonate

-
-
148151-47-7
(2R)-tetrahydrofuran-2-carboxylic acid chloride

-
-
1161825-32-6
ethyl 3-oxo-3-[(2R)-tetrahydrofuran-2-yl]propanoate

| Conditions | Yield |
|---|---|
| Stage #1: hydrogen ethyl malonate With n-butyllithium In tetrahydrofuran; hexanes at -78 - -5℃; Stage #2: (2R)-tetrahydrofuran-2-carboxylic acid chloride In tetrahydrofuran; hexanes at -65℃; for 1h; | 100% |
-
-
1071-46-1
hydrogen ethyl malonate

-
-
1227685-20-2
(2S,4R)-1-benzyl 4-methyl 4-amino-2-methylpiperidine-1,4-dicarboxylate

| Conditions | Yield |
|---|---|
| With 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride; triethylamine In dichloromethane at 20℃; | 100% |
-
-
1071-46-1
hydrogen ethyl malonate

-
-
619-21-6
m-formylphenyl benzoic acid

-
-
91047-79-9
(E)-3-(3-ethoxy-3-oxoprop-1-en-1-yl)benzoic acid

| Conditions | Yield |
|---|---|
| With piperidine In pyridine at 100℃; for 18h; Inert atmosphere; | 100% |

| Conditions | Yield |
|---|---|
| With benzotriazol-1-ol; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride In N,N-dimethyl-formamide at 0.25℃; for 12h; | 100% |
-
-
1071-46-1
hydrogen ethyl malonate

| Conditions | Yield |
|---|---|
| With pyridine; trichlorophosphate In acetonitrile at 0 - 5℃; for 2h; | 100% |
-
-
120-57-0
piperonal

-
-
1071-46-1
hydrogen ethyl malonate

-
-
24393-66-6
ethyl (E)-3-(benzo[d][1,3]dioxol-5-yl)acrylate

| Conditions | Yield |
|---|---|
| With poly{styrene-co-[4-(N-methyl-N-vinylbenzyl-amino)pyridine]-co-[1-phenyl-3-(4-vinylbenzyl)piperazine]} In N,N-dimethyl-formamide at 50℃; decarboxylative Doebner-Knoevenagel condensation reaction; optical yield given as %de; stereoselective reaction; | 99% |
| With piperidine; pyridine | |
| With piperidine; pyridine at 70℃; for 9h; |
-
-
1071-46-1
hydrogen ethyl malonate

-
-
70685-50-6
(E)-4-(4-Isopropoxy-phenyl)-but-3-en-2-one

-
-
111973-93-4
3-Hydroxy-5-(4-isopropoxy-phenyl)-cyclohex-2-enone

| Conditions | Yield |
|---|---|
| With sodium methylate In methanol | 99% |
-
-
1071-46-1
hydrogen ethyl malonate

-
-
79-30-1
isobutyryl chloride

-
-
7152-15-0
ethyl 4-methyl-3-oxo-pentanoate

| Conditions | Yield |
|---|---|
| With n-butyllithium In tetrahydrofuran | 99% |
| Stage #1: hydrogen ethyl malonate With [2,2]bipyridinyl; n-butyllithium In tetrahydrofuran; hexane at -70 - -10℃; Inert atmosphere; Stage #2: isobutyryl chloride In tetrahydrofuran; hexane at -65℃; for 0.166667h; Inert atmosphere; | 80% |
-
-
1071-46-1
hydrogen ethyl malonate

-
-
123-11-5
4-methoxy-benzaldehyde

-
-
24393-56-4
ethyl (E)-3-(4-methoxyphenyl)prop-2-enoate

| Conditions | Yield |
|---|---|
| With dmap In N,N-dimethyl-formamide at 25℃; Doebner-Knoevenagel reaction; | 99% |
| With dmap In N,N-dimethyl-formamide at 25℃; Knoevenagel reaction; | 99% |
| piperidine; dmap In N,N-dimethyl-formamide at 10 - 20℃; for 24h; Product distribution / selectivity; | 99% |
| With poly{styrene-co-[4-(N-methyl-N-vinylbenzyl-amino)pyridine]-co-[1-phenyl-3-(4-vinylbenzyl)piperazine]} In N,N-dimethyl-formamide at 50℃; decarboxylative Doebner-Knoevenagel condensation reaction; optical yield given as %de; | 97% |
-
-
98-03-3
thiophene-2-carbaldehyde

-
-
1071-46-1
hydrogen ethyl malonate

-
-
13979-15-2
ethyl (E)-3-(thiophen-2-yl)acrylate

| Conditions | Yield |
|---|---|
| With poly{styrene-co-[4-(N-methyl-N-vinylbenzyl-amino)pyridine]-co-[1-phenyl-3-(4-vinylbenzyl)piperazine]} In N,N-dimethyl-formamide at 50℃; decarboxylative Doebner-Knoevenagel condensation reaction; optical yield given as %de; stereoselective reaction; | 99% |
| With dmap |
-
-
1071-46-1
hydrogen ethyl malonate

-
-
623-33-6
glycine ethyl ester hydrochloride

-
-
51925-56-5
ethyl 3-((2-ethoxy-2-oxoethyl)amino)-3-oxopropanoate

| Conditions | Yield |
|---|---|
| With dmap; triethylamine; dicyclohexyl-carbodiimide In dichloromethane at 0 - 20℃; for 6.5h; Inert atmosphere; | 99% |
| Stage #1: hydrogen ethyl malonate; glycine ethyl ester hydrochloride With dmap; triethylamine In dichloromethane at 0℃; for 0.5h; Stage #2: With 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride In dichloromethane at 20℃; for 7h; | 79% |
| With dicyclohexyl-carbodiimide; trimethylamine In dichloromethane at 0 - 20℃; for 4.5h; | 78% |
| With triethylamine; dicyclohexyl-carbodiimide In dichloromethane at 20℃; | 70% |
-
-
500-22-1
3-pyridinecarboxaldehyde

-
-
1071-46-1
hydrogen ethyl malonate

-
-
59607-99-7, 28447-17-8
ethyl (E)-3-(pyridin-3-yl)acrylate

| Conditions | Yield |
|---|---|
| With poly{styrene-co-[4-(N-methyl-N-vinylbenzyl-amino)pyridine]-co-[1-phenyl-3-(4-vinylbenzyl)piperazine]} In N,N-dimethyl-formamide at 50℃; decarboxylative Doebner-Knoevenagel condensation reaction; optical yield given as %de; stereoselective reaction; | 99% |
-
-
1071-46-1
hydrogen ethyl malonate

-
-
1122-91-4
4-bromo-benzaldehyde

-
-
24393-53-1, 136265-11-7, 15795-20-7
ethyl (E)-3-(4-bromophenyl)-2-propenoate

| Conditions | Yield |
|---|---|
| With poly{styrene-co-[4-(N-methyl-N-vinylbenzyl-amino)pyridine]-co-[1-phenyl-3-(4-vinylbenzyl)piperazine]} In N,N-dimethyl-formamide at 50℃; decarboxylative Doebner-Knoevenagel condensation reaction; optical yield given as %de; stereoselective reaction; | 99% |
-
-
1071-46-1
hydrogen ethyl malonate

-
-
135-02-4
ortho-anisaldehyde

-
-
24393-54-2
ethyl 3-(2-methoxyphenyl)prop-2-enoate

| Conditions | Yield |
|---|---|
| With poly{styrene-co-[4-(N-methyl-N-vinylbenzyl-amino)pyridine]-co-[1-phenyl-3-(4-vinylbenzyl)piperazine]} In N,N-dimethyl-formamide at 50℃; decarboxylative Doebner-Knoevenagel condensation reaction; optical yield given as %de; stereoselective reaction; | 99% |
-
-
1071-46-1
hydrogen ethyl malonate

-
-
100-61-8
N-methylaniline

-
-
59050-15-6
ethyl 3-(N-methyl-N-phenylamino)-3-oxopropionate

| Conditions | Yield |
|---|---|
| With 2-chloro-1-methyl-pyridinium iodide; triethylamine In dichloromethane at 0 - 20℃; for 3h; Inert atmosphere; | 99% |
| With dicyclohexyl-carbodiimide In dichloromethane at 20℃; | 90% |
| With 2-chloro-1-methyl-pyridinium iodide; triethylamine In dichloromethane at 0 - 20℃; for 1h; Inert atmosphere; | 77% |

| Conditions | Yield |
|---|---|
| With dmap; triethylamine; dicyclohexyl-carbodiimide In dichloromethane at 0 - 20℃; for 2.5h; Inert atmosphere; | 99% |

| Conditions | Yield |
|---|---|
| Stage #1: hydrogen ethyl malonate; vanillin With piperidine; pyridine at 100℃; for 24h; Knoevenagel Condensation; Stage #2: With hydrogenchloride In water at -10 - 0℃; for 24h; | 98% |
| With piperidine In pyridine at 100℃; for 8h; Knoevenagel Condensation; | 97% |
| With piperidine; pyridine; aniline | |
| With pyridine Knoevenagel Condensation; Reflux; |
-
-
1071-46-1
hydrogen ethyl malonate

-
-
196819-38-2
(S)-N-[1-(5-oxazoyl)ethyl]-1H-indole-3-ethanamine

-
-
196819-50-8
(S)-3-[[2-(1H-indol-3-yl)ethyl][1-(5-oxazoyl)ethyl]amino]-3-oxopropanoic acid ethyl ester

| Conditions | Yield |
|---|---|
| With diethyl cyanophosphonate; triethylamine In N,N-dimethyl-formamide for 2h; Ambient temperature; | 98% |
| With diethyl cyanophosphonate; triethylamine In N,N-dimethyl-formamide at 20℃; for 2h; Condensation; | 98% |

| Conditions | Yield |
|---|---|
| With triethylamine In tetrahydrofuran at 4℃; for 0.5h; | A 98% B n/a |

-
-
57690-96-7
2,2-dimethylbut-3-enoyl chloride

-
-
1071-46-1
hydrogen ethyl malonate

-
-
66248-79-1
ethyl 4,4-dimethyl-3-oxo-hex-5-enoate

| Conditions | Yield |
|---|---|
| Stage #1: hydrogen ethyl malonate With [2,2]bipyridinyl; n-butyllithium In tetrahydrofuran; hexane at -70 - -10℃; Stage #2: 2,2-dimethylbut-3-enoyl chloride In tetrahydrofuran; hexane at -60 - 0℃; for 3h; | 98% |
| With n-butyllithium In tetrahydrofuran; hexane at -65℃; for 1h; | 8.1 g |
| Stage #1: hydrogen ethyl malonate With n-butyllithium In tetrahydrofuran; hexanes at -70 - -10℃; Inert atmosphere; Stage #2: 2,2-dimethylbut-3-enoyl chloride In tetrahydrofuran; hexanes at -60 - 0℃; for 3h; |
-
-
1071-46-1
hydrogen ethyl malonate

-
-
122-85-0
para-acetamidobenzaldehyde

-
-
215258-04-1
ethyl 3-(4-acetylaminophenyl)-2-propenoate

| Conditions | Yield |
|---|---|
| With poly{styrene-co-[4-(N-methyl-N-vinylbenzyl-amino)pyridine]-co-[1-phenyl-3-(4-vinylbenzyl)piperazine]} In N,N-dimethyl-formamide at 50℃; decarboxylative Doebner-Knoevenagel condensation reaction; optical yield given as %de; stereoselective reaction; | 98% |
| With dmap In N,N-dimethyl-formamide at 25℃; for 168h; Knoevenagel reaction; | 92% |
-
-
1071-46-1
hydrogen ethyl malonate

-
-
1750-36-3, 33993-35-0, 538-51-2, 137431-97-1, 137432-04-3
(E)-N-benzylidenebenzenamine

| Conditions | Yield |
|---|---|
| Stage #1: hydrogen ethyl malonate With 1,1'-carbonyldiimidazole In dichloromethane at 25℃; for 1h; Stage #2: (E)-N-benzylidenebenzenamine In dichloromethane at 25℃; for 1h; | 98% |
| Stage #1: hydrogen ethyl malonate With 1,1'-carbonyldiimidazole In dichloromethane at 20℃; for 1h; Staudinger reaction; Inert atmosphere; Stage #2: (E)-N-benzylidenebenzenamine In dichloromethane at 20℃; for 1h; Staudinger reaction; Inert atmosphere; stereoselective reaction; | 79% |
-
-
1071-46-1
hydrogen ethyl malonate

-
-
104-88-1
4-chlorobenzaldehyde

-
-
24393-52-0
ethyl (E)-3-(4-chlorophenyl)prop-2-enoate

| Conditions | Yield |
|---|---|
| With poly{styrene-co-[4-(N-methyl-N-vinylbenzyl-amino)pyridine]-co-[1-phenyl-3-(4-vinylbenzyl)piperazine]} In N,N-dimethyl-formamide at 50℃; decarboxylative Doebner-Knoevenagel condensation reaction; optical yield given as %de; stereoselective reaction; | 98% |
| With dmap |
-
-
1071-46-1
hydrogen ethyl malonate

| Conditions | Yield |
|---|---|
| With pyridine; trichlorophosphate In acetonitrile at 0 - 5℃; for 2h; | 98% |
-
-
1071-46-1
hydrogen ethyl malonate

-
-
74389-78-9
4-(p-isopropylphenyl)-3-buten-2-one

-
-
111945-86-9
3-Hydroxy-5-(4-isopropyl-phenyl)-cyclohex-2-enone

| Conditions | Yield |
|---|---|
| With sodium methylate In methanol | 97% |
Related products
Downstream Products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View
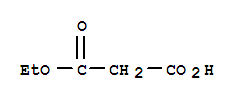








 Xi
Xi