Synthetic route

| Conditions | Yield |
|---|---|
| With sulfuric acid; nitric acid at 110℃; for 4h; Temperature; | 92.2% |
| With sulfuric acid; nitric acid at 20 - 100℃; Nitration; | 89% |
| With sulfuric acid; nitric acid 1.) 0 deg C, 10 min, 2.) 95 deg C, 6 h, then room temp., 12 h; | 69% |

| Conditions | Yield |
|---|---|
| With sulfuric acid; nitric acid at 110℃; for 4h; | 92.2% |
| With sulfuric acid; nitric acid | |
| durch Nitrierung; |

| Conditions | Yield |
|---|---|
| With sulfuric acid; nitric acid at 20 - 90℃; Nitration; | 84% |
| With nitric acid; Petroleum ether azeotropes Abdestillieren des gebildeten Wassers; | |
| With sulfuric acid; nitric acid |

| Conditions | Yield |
|---|---|
| With sulfuric acid; nitric acid 1.) 90-100 deg C, 2 h, 2.) 25 deg C, overnight; | 62% |
| Nitrierung; | |
| durch Nitrierung; |

| Conditions | Yield |
|---|---|
| With lead(IV) acetate; acetic acid |

| Conditions | Yield |
|---|---|
| With sulfuric acid; nitric acid at 60 - 70℃; |

| Conditions | Yield |
|---|---|
| With sulfuric acid; nitric acid at 93 - 103℃; | |
| With ammonium nitrate In sulfuric acid at 0 - 25℃; |
-
-
77601-83-3
2,4,6-trinitrophenylacetic acid

-
-
118-96-7
2,4,6-Trinitrotoluene

| Conditions | Yield |
|---|---|
| With water | |
| With ethanol |
-
-
99-65-0
meta-dinitrobenzene

-
-
546-67-8
lead(IV) tetraacetate

-
-
64-19-7
acetic acid

-
A
-
118-96-7
2,4,6-Trinitrotoluene

-
B
-
632-92-8
2,4-dimethyl-1,3,5-trinitrobenzene

| Conditions | Yield |
|---|---|
| Erwaermen des Reaktionsprodukts mit rauchender Salpetersaeure und konz. H2SO4 auf 75-120grad; |

-
-
99-35-4
1,3,5-trinitrobenzene

-
-
546-67-8
lead(IV) tetraacetate

-
-
64-19-7
acetic acid

-
A
-
118-96-7
2,4,6-Trinitrotoluene

-
B
-
632-92-8
2,4-dimethyl-1,3,5-trinitrobenzene


| Conditions | Yield |
|---|---|
| With sulfuric acid; nitric acid Thermodynamic data; ΔH, E(a); |
-
-
121-14-2
2,4-dinitrotoluene

-
A
-
118-96-7
2,4,6-Trinitrotoluene

-
B
-
18242-38-1
1-bromo-2-methyl-3,5-dinitrobenzene

| Conditions | Yield |
|---|---|
| With sulfuric acid; bromine; nitric acid at 90℃; for 0.166667h; Title compound not separated from byproducts; | A 0.4 % Chromat. B 94.7 % Chromat. |
-
-
121-14-2
2,4-dinitrotoluene

-
A
-
118-96-7
2,4,6-Trinitrotoluene

-
B
-
18242-38-1
1-bromo-2-methyl-3,5-dinitrobenzene

-
C
-
96-90-2
1-chloro-2-methyl-3,5-dinitrobenzene

| Conditions | Yield |
|---|---|
| With sulfuric acid; nitric acid; chlorine at 90℃; for 0.8h; Rate constant; Mechanism; Product distribution; further reaction times; oleum instead of sulphuric acid; | A 19.1 % Chromat. B 8.8 % Chromat. C 52.3 % Chromat. |


| Conditions | Yield |
|---|---|
| With sodium methylate In methanol at 25℃; Equilibrium constant; Rate constant; |

| Conditions | Yield |
|---|---|
| With sodium methylate In methanol at 25℃; Equilibrium constant; Rate constant; |

| Conditions | Yield |
|---|---|
| With sodium methylate In methanol at 25℃; Equilibrium constant; Rate constant; |

| Conditions | Yield |
|---|---|
| With sodium methylate In methanol at 25℃; Equilibrium constant; Rate constant; |

| Conditions | Yield |
|---|---|
| With sodium methylate In methanol at 25℃; Equilibrium constant; Rate constant; |

| Conditions | Yield |
|---|---|
| With Isopropylamine hydroperchlorate In dimethyl sulfoxide at 25℃; Equilibrium constant; |

| Conditions | Yield |
|---|---|
| With n-butylammonium perchlorate In dimethyl sulfoxide at 25℃; Equilibrium constant; |

-
-
118-96-7
2,4,6-Trinitrotoluene

| Conditions | Yield |
|---|---|
| With sodium sulfite In water at 25℃; Equilibrium constant; variation with solvent composition; |

| Conditions | Yield |
|---|---|
| With piperidine hydrochloride In dimethyl sulfoxide at 25℃; Equilibrium constant; |

| Conditions | Yield |
|---|---|
| With benzylamine hydrogenperchlorate In dimethyl sulfoxide at 25℃; Equilibrium constant; |

| Conditions | Yield |
|---|---|
| In acetonitrile at 25℃; Rate constant; different solvents; |

| Conditions | Yield |
|---|---|
| In methanol; dimethyl sulfoxide at 25℃; Rate constant; Equilibrium constant; |

| Conditions | Yield |
|---|---|
| unter azeotropem Abdestillieren des entstehenden Wassers; | |
| in der Waerme bei groesserer Konzentration der Salpetersaeure; | |
| unter azeotropem Abdestillieren des entstehenden Wassers; |

| Conditions | Yield |
|---|---|
| in der Waerme; |

-
-
99-87-6
4-methylisopropylbenzene

-
-
7664-93-9
sulfuric acid

-
-
7697-37-2
nitric acid

-
A
-
118-96-7
2,4,6-Trinitrotoluene

-
B
-
16533-71-4
3,5-dinitro-p-toluic acid

| Conditions | Yield |
|---|---|
| at 60 - 70℃; |


| Conditions | Yield |
|---|---|
| With piperidine In benzene for 12h; Knoevenagel Condensation; Dean-Stark; Reflux; | 95.1% |
-
-
118-96-7
2,4,6-Trinitrotoluene

-
-
35113-75-8
1-nitromethyl-2,4,6-trinitrobenzene

| Conditions | Yield |
|---|---|
| With potassium hydroxide; fluorotrinitromethane In tetrahydrofuran; methanol; water for 0.0333333h; Nitration; | 95% |
-
-
118-96-7
2,4,6-Trinitrotoluene

| Conditions | Yield |
|---|---|
| Stage #1: 2,4,6-Trinitrotoluene With hydrogen; pyrographite In methanol at 120℃; under 45004.5 Torr; for 5h; Large scale; Stage #2: With hydrogenchloride In water for 1h; Reagent/catalyst; Solvent; Pressure; Large scale; | 95% |
-
-
118-96-7
2,4,6-Trinitrotoluene

-
-
606-34-8
2,4,6-trinitrobenzaldehyde

-
-
20062-22-0
2,2’,4,4’,6,6’-hexanitrostilbene

| Conditions | Yield |
|---|---|
| With piperidine In toluene Reflux; | 94.2% |
-
-
118-96-7
2,4,6-Trinitrotoluene

| Conditions | Yield |
|---|---|
| Stage #1: 2,4,6-Trinitrotoluene With hydrogen In toluene at 120℃; under 30003 Torr; for 10h; Autoclave; Stage #2: With hydrogenchloride In water for 0.5h; Solvent; Pressure; Temperature; | 93.1% |

| Conditions | Yield |
|---|---|
| With piperidine In benzene for 14h; Knoevenagel Condensation; Dean-Stark; Reflux; | 90.3% |

| Conditions | Yield |
|---|---|
| With piperidine In benzene for 6h; Knoevenagel Condensation; Dean-Stark; Reflux; | 90.2% |
-
-
1125-80-0
3-methylisoquinoline

-
-
118-96-7
2,4,6-Trinitrotoluene

-
-
98-88-4
benzoyl chloride

-
-
94170-03-3
[3-Methyl-1-(2,4,6-trinitro-benzyl)-1H-isoquinolin-2-yl]-phenyl-methanone

| Conditions | Yield |
|---|---|
| In chloroform at 60 - 70℃; for 1h; | 90% |


| Conditions | Yield |
|---|---|
| With hydrogenchloride; palladium on activated charcoal; hydrogen In methanol at 20℃; under 18751.9 Torr; for 2h; Solvent; Pressure; Time; | 90% |
| With iron(III) chloride; pyrographite; hydrazine hydrate In methanol for 7h; Heating; | 67.8% |
-
-
118-96-7
2,4,6-Trinitrotoluene

-
-
544676-41-7
3-formyl-4,6-dinitro-1-phenyl-1H-indazole

| Conditions | Yield |
|---|---|
| With potassium carbonate In ethanol at 20℃; for 24h; | 90% |

| Conditions | Yield |
|---|---|
| 90% | |
| In toluene at 20℃; for 24h; | 70% |

| Conditions | Yield |
|---|---|
| With piperidine In benzene for 11h; Knoevenagel Condensation; Dean-Stark; Reflux; | 89.5% |

| Conditions | Yield |
|---|---|
| With sodium chlorate; nitric acid | 88% |
| With sodium dichromate; sulfuric acid at 45 - 55℃; for 2h; Oxidation; | 14% |
| With sodium chlorate; nitric acid Reinigung ueber das Natrium-Salz; |
-
-
118-96-7
2,4,6-Trinitrotoluene

-
-
100-52-7
benzaldehyde

-
-
61599-68-6
1,3,5-trinitro-2-[(E)-2-phenylvinyl]benzene

| Conditions | Yield |
|---|---|
| With piperidine; silica gel In neat (no solvent) at 120℃; for 0.333333h; Microwave irradiation; | 88% |
| With piperidine In benzene for 6h; Condensation; Heating; | 81% |
| With HTc-4-Cal In toluene for 15h; Reagent/catalyst; Time; Reflux; Dean-Stark; | 70% |
| With piperidine | |
| With piperidine; ethanol at 40℃; |
-
-
118-96-7
2,4,6-Trinitrotoluene

-
-
2365-48-2
Methyl thioglycolate

-
A
-
321596-17-2
2,4-dinitro-6-[(methoxycarbonyl)methylthio]toluene

| Conditions | Yield |
|---|---|
| With potassium carbonate In 1-methyl-pyrrolidin-2-one at 20℃; for 24h; | A 88% B n/a |

-
-
118-96-7
2,4,6-Trinitrotoluene

-
-
104-88-1
4-chlorobenzaldehyde

-
-
65200-05-7
2-[(E)-2-(4-Chloro-phenyl)-vinyl]-1,3,5-trinitro-benzene

| Conditions | Yield |
|---|---|
| With piperidine In benzene for 5.5h; Knoevenagel Condensation; Dean-Stark; Reflux; | 87.6% |

| Conditions | Yield |
|---|---|
| With piperidine In benzene for 6h; Knoevenagel Condensation; Dean-Stark; Reflux; | 87% |
-
-
19788-49-9
Ethyl 2-mercaptopropionate

-
-
118-96-7
2,4,6-Trinitrotoluene

| Conditions | Yield |
|---|---|
| With potassium carbonate In 1-methyl-pyrrolidin-2-one at 20℃; for 24h; | A 86% B n/a |

-
-
118-96-7
2,4,6-Trinitrotoluene

-
-
52886-05-2
2,4,6-trinitro(α,αα-D3)toluene

| Conditions | Yield |
|---|---|
| With tributyl-amine; water-d2 In N,N-dimethyl-formamide for 5h; | 85% |
| With [(2)H6]acetone; deuteromethanol; water-d2; triethylamine for 1.5h; Ambient temperature; | 63% |
-
-
118-96-7
2,4,6-Trinitrotoluene

-
-
540-63-6
ethane-1,2-dithiol

-
-
1608182-18-8
1,2-bis((2-methyl-3,5-dinitrophenyl)thio)ethane

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide at 20℃; for 2h; | 85% |
-
-
118-96-7
2,4,6-Trinitrotoluene

-
-
623-51-8
ethyl 2-sulfanylacetate

-
-
367925-03-9
(2-methyl-3,5-dinitro-phenylsulfanyl)-acetic acid ethyl ester

| Conditions | Yield |
|---|---|
| With alkaline resin In acetone at 50℃; for 20h; Large scale; | 85% |
-
-
118-96-7
2,4,6-Trinitrotoluene

-
-
123-11-5
4-methoxy-benzaldehyde

-
-
61599-69-7
2-[(E)-2-(4-methoxyphenyl)vinyl]-1,3,5-trinitrobenzene

| Conditions | Yield |
|---|---|
| With HTc-4-Cal In toluene for 24h; Knoevenagel Condensation; Reflux; Dean-Stark; | 83% |
| With piperidine In benzene for 6h; Condensation; Heating; | 60% |
| With piperidine |

| Conditions | Yield |
|---|---|
| With piperidine; silica gel In neat (no solvent) at 120℃; for 0.333333h; Microwave irradiation; | 83% |
| With HTc-4-Cal In toluene for 24h; Knoevenagel Condensation; Reflux; Dean-Stark; | 71% |
-
-
107-03-9
1-thiopropane

-
-
118-96-7
2,4,6-Trinitrotoluene

-
-
1608182-14-4
(2-methyl-3,5-dinitrophenyl)(propyl)sulfane

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide at 20℃; for 2h; | 83% |

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide at 20℃; | 83% |

| Conditions | Yield |
|---|---|
| With piperidine; silica gel In neat (no solvent) at 120℃; for 0.333333h; Microwave irradiation; | 82% |
| With piperazine at 80 - 90℃; |
-
-
118-96-7
2,4,6-Trinitrotoluene

-
-
104-88-1
4-chlorobenzaldehyde

-
-
61599-70-0
trans-4′-chloro-2,4,6-trinitrostilbene

| Conditions | Yield |
|---|---|
| With piperidine; silica gel In neat (no solvent) at 120℃; for 0.333333h; Microwave irradiation; | 82% |
| With HTc-4-Cal In toluene for 24h; Knoevenagel Condensation; Reflux; Dean-Stark; | 70% |
| With piperidine at 120℃; |
Related products
Downstream Products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View



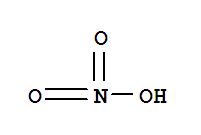










 E,
E,  T,
T,  N,
N,  Xn,
Xn,  F,
F,  B
B