-
Name
2-Chloro-1,1,1-trifluoroethane
- EINECS
- CAS No. 75-88-7
- Article Data68
- CAS DataBase
- Density 1.389
- Solubility
- Melting Point -105°C
- Formula C2H2 Cl F3
- Boiling Point 7°C
- Molecular Weight 118.486
- Flash Point
- Transport Information
- Appearance
- Safety A poison by inhalation. Experimental reproductive effects. Questionable carcinogen with experimental carcinogenic data. When heated to decomposition it emits toxic fumes of F−.
- Risk Codes R59
-
Molecular Structure
-
Hazard Symbols
 Xi
Xi
- Synonyms (Chloromethyl)trifluoromethane;1,1,1-Trifluoro-2-chloroethane; 1,1,1-Trifluorochloroethane;1,1,1-Trifluoroethyl chloride; 1-Chloro-2,2,2-trifluoroethane;2,2,2-Trifluoro-1-chloroethane; 2,2,2-Trifluorochloroethane;2,2,2-Trifluoroethyl chloride; 2-Chloro-1,1,1-trifluoroethane; F 133a; FC 133a;Forane 133a; Freon 133a; Genetron 133a; HCFC 133a; R 133a
- PSA 0.00000
- LogP 1.78750
Synthetic route

| Conditions | Yield |
|---|---|
| With hydrogen fluoride; antimony pentafluoride at 100℃; for 1h; Temperature; Autoclave; | 97% |
| With hydrogen fluoride; tantalum pentafluoride at 0 - 140℃; under 25858.1 Torr; for 10.5h; Product distribution / selectivity; Neat (no solvent); | 90% |
| With hydrogen fluoride; tantalum pentafluoride at 100 - 140℃; under 25858.1 Torr; for 6h; | 80% |

| Conditions | Yield |
|---|---|
| With hydrogen fluoride; antimonypentachloride at 80 - 90℃; Autoclave; | 92.3% |
| With hydrogen fluoride; antimonypentachloride; chlorine at 120℃; |

| Conditions | Yield |
|---|---|
| With 1,1,2-trichloro-1-fluoroethane; hydrogen fluoride; tantalum pentachloride at 0 - 140℃; for 2h; Product distribution / selectivity; Neat (no solvent); | 87.5% |
| With hydrogen fluoride; tantalum pentachloride at 0 - 140℃; for 2.5h; Product distribution / selectivity; Neat (no solvent); | 22% |
| With antimony dichloride trifluoride; hydrogen fluoride at 144℃; under 33097.9 Torr; |
-
-
75-89-8
2,2,2-trifluoroethanol

-
-
421-83-0
trifluoromethane sulfonyl chloride

-
A
-
75-88-7
1,1,1-trifluoro-2-chloroethane

-
B
-
333-36-8
1,1,1-trifluoro-2-(2,2,2-trifluoroethoxy)ethane

-
C
-
6226-25-1
2,2,2-trifluoroethyl trifluoromethanesulphonate

| Conditions | Yield |
|---|---|
| With potassium carbonate; tetra-n-propylammonium bromide In water at 10.2 - 23.3℃; for 4 - 7h; Product distribution / selectivity; | A 0.2 %Chromat. B 0.13 - 0.28 %Chromat. C 82% |


| Conditions | Yield |
|---|---|
| With ammonium persulfate; ammonium formate In N,N-dimethyl-formamide at 30 - 40℃; | 77% |
-
-
811-95-0
1,1,2-trichloro-1-fluoroethane

-
A
-
75-88-7
1,1,1-trifluoro-2-chloroethane

-
B
-
1649-08-7
1,2-dichloro-1,1-difluoroethane

| Conditions | Yield |
|---|---|
| With hydrogen fluoride; tantalum pentachloride at 110℃; for 1.5h; | A 22% B 74% |
-
-
306-83-2
1,1,1-trifluoro-2,2-dichloroethane

-
A
-
75-88-7
1,1,1-trifluoro-2-chloroethane

-
B
-
359-10-4
1-Chloro-2,2-difluoroethene

| Conditions | Yield |
|---|---|
| With water; zinc unter Zusatz eines Netzmittels; |

| Conditions | Yield |
|---|---|
| With aluminum(III) fluoride; hydrogen fluoride at 300℃; | |
| With hydrogen fluoride; boron trifluoride at 25℃; |
-
-
79-01-6
Trichloroethylene

-
A
-
75-88-7
1,1,1-trifluoro-2-chloroethane

-
B
-
354-15-4
1,2,2-trichloro-1,2-difluoroethane

-
C
-
76-13-1
1,1,2-Trichloro-1,2,2-trifluoroethane

-
D
-
76-12-0
CFC-112a

| Conditions | Yield |
|---|---|
| With cobalt (III) fluoride at 120℃; Further byproducts given; |

-
-
79-11-8
chloroacetic acid

-
A
-
75-88-7
1,1,1-trifluoro-2-chloroethane

-
B
-
38217-12-8
bis-(2-chloro-1,1-difluoro-ethyl) ether

| Conditions | Yield |
|---|---|
| With sulfur tetrafluoride at 65℃; for 3h; |

| Conditions | Yield |
|---|---|
| Ambient temperature; Irradiation; Hg2I2, Hg; |
-
-
75-38-7
Vinylidene fluoride

-
-
22082-78-6
trifluoromethyl hypochlorite

-
A
-
75-88-7
1,1,1-trifluoro-2-chloroethane

-
B
-
25957-33-9
Trifluormethyl-1,1-difluor-2-chloraethylaether

| Conditions | Yield |
|---|---|
| In trichlorofluoromethane at -111 - 22℃; for 20h; Product distribution; various temperatures, olefins, solvents and times; | A 50 % Spectr. B 50 % Spectr. |
| In trichlorofluoromethane at -111 - 22℃; for 20h; Yield given. Title compound not separated from byproducts; |

-
-
75-38-7
Vinylidene fluoride

-
A
-
75-88-7
1,1,1-trifluoro-2-chloroethane

-
B
-
25957-33-9
Trifluormethyl-1,1-difluor-2-chloraethylaether

| Conditions | Yield |
|---|---|
| With trifluoromethyl hypochlorite In trichlorofluoromethane at -111 - 22℃; for 20h; Yield given. Yields of byproduct given. Title compound not separated from byproducts; |
-
-
79-01-6
Trichloroethylene

-
A
-
811-97-2
1,1,1,2-tetrafluoroethane

-
B
-
75-88-7
1,1,1-trifluoro-2-chloroethane

| Conditions | Yield |
|---|---|
| With hydrogen fluoride; chromium(III) oxide at 250℃; Mechanism; Product distribution; hydrofluorination, chlorine/fluorine exchange mechanism; effect of DF substitution for HF; | A 10 % Chromat. B 90 % Chromat. |
| With chromium(III) oxide; hydrogen fluoride at 653℃; for 18h; Product distribution; Further Variations:; Temperatures; Fluorination; Addition; Substitution; | |
| With chromium fluoride; magnesium fluoride; hydrogen fluoride at 200℃; under 7600 Torr; Product distribution; Further Variations:; Pressures; |
-
-
151-67-7
1,1,1-trifluoro-2-bromo-2-chloroethane

-
A
-
75-88-7
1,1,1-trifluoro-2-chloroethane

-
B
-
359-10-4
1-Chloro-2,2-difluoroethene

| Conditions | Yield |
|---|---|
| With perchloric acid; CoW7-; sodium perchlorate In water; acetonitrile at 20℃; Rate constant; Product distribution; pH 7; reaction with Co(II)sepuchrate2- at 50 deg C; electron transfer reactions of polyhaloalkanes with Co(II)W12O407- and Co(II)sepulchrate2+, order of reactivity toward Co(II)W12O407-, trapping experiments with N-t-butyl-α-phenylnitrone; | |
| With liver microsomes Enzyme kinetics; Enzymatic reaction; |
-
-
28760-99-8
1,1,1-trifluoro-2-chloroethyl radical

-
-
75-88-7
1,1,1-trifluoro-2-chloroethane

| Conditions | Yield |
|---|---|
| With isopropyl alcohol |
-
-
58911-30-1
3-chloro-3-(trifluoromethyl)diazirine

-
A
-
75-88-7
1,1,1-trifluoro-2-chloroethane

-
B
-
79-38-9
Chlorotrifluoroethylene

| Conditions | Yield |
|---|---|
| Irradiation; | |
| Irradiation; radiation > 300 nm; |
-
-
354-58-5
1,1,1-Trichloro-2,2,2-trifluoroethane

-
A
-
75-88-7
1,1,1-trifluoro-2-chloroethane

-
B
-
306-83-2
1,1,1-trifluoro-2,2-dichloroethane

| Conditions | Yield |
|---|---|
| With hydrogen; RhCl(PPh3)3 In tetrahydrofuran at 100℃; under 6080 Torr; for 5h; Product distribution; further catalysts and times; effect of bases; | A 3.5 % Chromat. B 94.6 % Chromat. |
| With hydrogen; RhCl(PPh3)3 In tetrahydrofuran at 100℃; under 6080 Torr; for 5h; | A 3.5 % Chromat. B 94.6 % Chromat. |
-
-
306-83-2
1,1,1-trifluoro-2,2-dichloroethane

-
-
100-52-7
benzaldehyde

-
A
-
75-88-7
1,1,1-trifluoro-2-chloroethane

-
B
-
359-10-4
1-Chloro-2,2-difluoroethene

-
C
-
67230-92-6
2-chloro-3,3-difluoro-1-phenyl-2-propenol

-
D
-
103654-96-2, 121591-61-5, 121591-67-1
1-(α,α-dichloro-β,β,β-trifluoroethyl)benzylalcohol

-
E
-
138950-21-7
2-chloro-1,1,1-trifluoro-3-phenylpropan-3-ol

| Conditions | Yield |
|---|---|
| With zinc; copper(l) chloride In N,N-dimethyl-formamide Product distribution; Mechanism; Ambient temperature; various concentrations; without catalyst; further aldehydes; | A 24 % Spectr. B 20 % Spectr. C 37 % Spectr. D 3 % Spectr. E 12 % Spectr. |

-
-
306-83-2
1,1,1-trifluoro-2,2-dichloroethane

-
-
100-52-7
benzaldehyde

-
A
-
75-88-7
1,1,1-trifluoro-2-chloroethane

-
B
-
67230-92-6
2-chloro-3,3-difluoro-1-phenyl-2-propenol

-
C
-
103654-96-2, 121591-61-5, 121591-67-1
1-(α,α-dichloro-β,β,β-trifluoroethyl)benzylalcohol

-
D
-
138950-21-7
2-chloro-1,1,1-trifluoro-3-phenylpropan-3-ol

| Conditions | Yield |
|---|---|
| With zinc; copper(l) chloride 1.) DMF, 24 h; 2.) DMF; Yield given. Multistep reaction. Yields of byproduct given; |

-
-
370-69-4
tris(2,2,2-trifluoroethyl)phosphite

-
-
75-87-6
chloral

-
A
-
75-88-7
1,1,1-trifluoro-2-chloroethane

| Conditions | Yield |
|---|---|
| Heating; |

-
-
75-88-7
1,1,1-trifluoro-2-chloroethane

-
-
75-89-8
2,2,2-trifluoroethanol

-
-
333-36-8
1,1,1-trifluoro-2-(2,2,2-trifluoroethoxy)ethane

| Conditions | Yield |
|---|---|
| With potassium hydroxide In 1,2-dimethoxyethane at 70℃; for 4h; Temperature; | 97.4% |

| Conditions | Yield |
|---|---|
| With potassium γ-hydroxybutyrate In 4-butanolide at 200℃; under 11251.1 Torr; for 6h; | 96% |
| With 4-butanolide; potassium hydroxide at 200℃; under 11251.1 Torr; for 6h; | 68% |
| With potassium acetate; ethylene glycol; acetic acid |

| Conditions | Yield |
|---|---|
| With dmap; ammonia at 110℃; Temperature; | 94% |
| With ammonia; water at 190℃; | |
| With ammonia; water at 185℃; |
-
-
75-88-7
1,1,1-trifluoro-2-chloroethane

-
-
68716-49-4
p-bromophenylboronic acid pinacol ester

-
-
1310949-87-1
4,4,5,5-tetramethyl-2-(4-(2,2,2-trifluoroethyl)phenyl)-1,3,2-dioxaborolane

| Conditions | Yield |
|---|---|
| With 4,4'-di-tert-butyl-2,2'-bipyridine; magnesium chloride; nickel dibromide; zinc In N,N-dimethyl acetamide at 80℃; for 12h; Inert atmosphere; Schlenk technique; | 94% |
-
-
75-88-7
1,1,1-trifluoro-2-chloroethane

-
-
17878-47-6
(3-bromophenyl)trimethylsilane

| Conditions | Yield |
|---|---|
| With 4,4'-di-tert-butyl-2,2'-bipyridine; magnesium chloride; nickel dibromide; zinc In N,N-dimethyl acetamide at 80℃; for 12h; Inert atmosphere; Schlenk technique; | 93% |

| Conditions | Yield |
|---|---|
| With BuLi In tetrahydrofuran; diethyl ether (N2); BuLi slowly added to stirred soln. of ligand (1 equiv.) in Et2O at-80°C; react. temp. was maintained between -55°C and -80. degree.C for 2 h; HgCl2 (1 equiv.) in THF added at -110°C; warmedto -35°C for few h; warmed to room temp.; solvent vol. reduced in vac. to half; hexane added; mixt. was allowed to settle; filtered under N2; solvent removed in vac.; washed (hexane); elem. anal.; | 87% |
-
-
75-88-7
1,1,1-trifluoro-2-chloroethane

-
-
32316-92-0
naphthalene-2-boronic acid

-
-
1204295-99-7
2-(2,2,2-trifluoroethyl)naphthalene

| Conditions | Yield |
|---|---|
| With [2,2]bipyridinyl; nickel(II) bromide dimethoxyethane; potassium phosphate In dimethyl sulfoxide at 80℃; for 30h; Inert atmosphere; Sealed tube; | 87% |
-
-
75-88-7
1,1,1-trifluoro-2-chloroethane

-
-
127-08-2
potassium acetate

-
-
406-93-9
4,4,4-trifluorobutyric acid

| Conditions | Yield |
|---|---|
| Stage #1: 1,1,1-trifluoro-2-chloroethane; potassium acetate In 1-methyl-pyrrolidin-2-one at 180℃; for 23h; Autoclave; Sealed tube; Stage #2: With acetic anhydride In 1-methyl-pyrrolidin-2-one at 35 - 40℃; for 3h; Reagent/catalyst; Solvent; | 86.5% |

| Conditions | Yield |
|---|---|
| Stage #1: 1,1,1-trifluoro-2-chloroethane With n-butyllithium; diisopropylamine; zinc(II) chloride In tetrahydrofuran at 15 - 20℃; for 2h; Stage #2: 1-Bromo-3-iodobenzene With tetrakis(triphenylphosphine) palladium(0) at 20℃; for 18h; | 85% |

| Conditions | Yield |
|---|---|
| Stage #1: 1,1,1-trifluoro-2-chloroethane With zinc(II) chloride; lithium diisopropyl amide In tetrahydrofuran at 20℃; for 2h; Stage #2: 1-Bromo-3-iodobenzene; tetrakis(triphenylphosphine) palladium(0) In tetrahydrofuran at 20℃; for 18h; | A 85% B 1% |

-
-
75-88-7
1,1,1-trifluoro-2-chloroethane

-
-
5720-07-0
4-methoxyphenylboronic acid

-
-
157928-44-4
1-methoxy-4-(2,2,2-trifluoroethyl)benzene

| Conditions | Yield |
|---|---|
| With [2,2]bipyridinyl; nickel(II) bromide dimethoxyethane; potassium phosphate In dimethyl sulfoxide at 80℃; for 30h; Inert atmosphere; Sealed tube; | 85% |
-
-
75-88-7
1,1,1-trifluoro-2-chloroethane

-
-
49562-28-9
fenofibrate

| Conditions | Yield |
|---|---|
| With 4,4'-di-tert-butyl-2,2'-bipyridine; magnesium chloride; nickel dibromide; zinc In N,N-dimethyl acetamide at 80℃; for 12h; Inert atmosphere; Schlenk technique; | 85% |
-
-
75-88-7
1,1,1-trifluoro-2-chloroethane

-
-
1461-22-9
tributyltin chloride

-
-
214422-68-1
tributyl(chlorodifluorovinyl)tin

| Conditions | Yield |
|---|---|
| With BuLi In diethyl ether BuLi added to soln. of ligand in Et2O at -80°C; after 2 h soln. of Bu3SnCl in Et2O (dissolved at -80°C) added slowly at -110°C; warmed to room temp. overnight; hexane added; filtered; solvent removed on rotary evaporator; distd. in vac. (242°C, 0.2 mmHg); elem. anal.; | 84% |
-
-
75-88-7
1,1,1-trifluoro-2-chloroethane

-
-
2767-54-6
triethyltin bromide

-
-
329911-50-4
triethyl(chlorodifluorovinyl)tin

| Conditions | Yield |
|---|---|
| With BuLi In diethyl ether (N2); BuLi added to soln. of ligand in Et2O at -80°C; after 2 h soln. of Et3SnBr in Et2O (dissolved at -80°C) added slowly at -110°C; warmed to room temp. overnight; hexane added; filtered; solvent removed on rotary evaporator; distd. in vac. (182°C, 0.2 mmHg); elem. anal.; | 84% |

| Conditions | Yield |
|---|---|
| Stage #1: 1,1,1-trifluoro-2-chloroethane With n-butyllithium; diisopropylamine; zinc(II) chloride In tetrahydrofuran at 15 - 20℃; for 2h; Stage #2: 4-tolyl iodide With tetrakis(triphenylphosphine) palladium(0) at 20℃; for 12h; | 83% |
| Stage #1: 1,1,1-trifluoro-2-chloroethane With zinc(II) chloride; lithium diisopropyl amide In tetrahydrofuran at 20℃; for 2h; Stage #2: 4-tolyl iodide; tetrakis(triphenylphosphine) palladium(0) In tetrahydrofuran at 20℃; for 12h; | 83% |

| Conditions | Yield |
|---|---|
| With caesium carbonate In N,N-dimethyl-formamide for 16h; Inert atmosphere; | 83% |
-
-
75-88-7
1,1,1-trifluoro-2-chloroethane

-
-
3972-65-4
1-bromo-4-tert-butylbenzene

-
-
1099597-89-3
1-(tert-butyl)-4-(2,2,2-trifluoroethyl)benzene

| Conditions | Yield |
|---|---|
| With 4,4'-di-tert-butyl-2,2'-bipyridine; magnesium chloride; nickel dibromide; zinc In N,N-dimethyl acetamide at 80℃; for 12h; Inert atmosphere; Schlenk technique; | 83% |
-
-
193694-04-1
N-TBS-4-bromoindole

-
-
75-88-7
1,1,1-trifluoro-2-chloroethane

| Conditions | Yield |
|---|---|
| With 4,4'-di-tert-butyl-2,2'-bipyridine; magnesium chloride; nickel dibromide; zinc In N,N-dimethyl acetamide at 80℃; for 20h; Inert atmosphere; Schlenk technique; | 83% |
-
-
75-88-7
1,1,1-trifluoro-2-chloroethane

-
-
17282-40-5
N-(ethoxycarbonylmethyl)pyridinium bromide

| Conditions | Yield |
|---|---|
| With potassium carbonate; triethylamine In N,N-dimethyl-formamide at 80℃; for 24h; | 82% |
1,1,1-Trifluoro-2-chloroethane Chemical Properties
Molecular Structure of 1,1,1-Trifluoro-2-chloroethane (CAS NO.75-88-7):
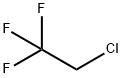
EINECS: 200-912-0
IUPAC Name: 2-Chloro-1,1,1-trifluoroethane
Molecular Formula: C2H2ClF3
Molecular Weight: 118.485490 g/mol
XLogP3-AA: 1.9
H-Bond Donor: 0
H-Bond Acceptor: 3
Canonical SMILES: C(C(F)(F)F)Cl
InChI: InChI=1S/C2H2ClF3/c3-1-2(4,5)6/h1H2
InChIKey: CYXIKYKBLDZZNW-UHFFFAOYSA-N
Index of Refraction: 1.298
Molar Refractivity: 16.62 cm3
Molar Volume: 89.3 cm3
Surface Tension: 14.3 dyne/cm
Density: 1.326 g/cm3
Enthalpy of Vaporization: 24.48 kJ/mol
Boiling Point: 6.9 °C at 760 mmHg
Vapour Pressure: 1430 mmHg at 25 °C
Melting Point: -105 °C
Water Solubility: 365.4 mg/L at 25 °C
BRN: 1733200
1,1,1-Trifluoro-2-chloroethane Toxicity Data With Reference
| 1. | orl-rat TDLo:78 g/kg/1Y-I:CAR,REP | TXAPA9 Toxicology and Applied Pharmacology. 72 (1984),15. | ||
| 2. | ihl-mus LC50:15 pph/1H | BJANAD British Journal of Anesthesia. 37 (1965),716. |
1,1,1-Trifluoro-2-chloroethane Consensus Reports
IARC Cancer Review: Group 3 IMEMDT IARC Monographs on the Evaluation of Carcinogenic Risk of Chemicals to Man . 7 ,1987,p. 56.(World Health Organization, Internation Agency for Research on Cancer,Lyon, France.: ) (Single copies can be ordered from WHO Publications Centre U.S.A., 49 Sheridan Avenue, Albany, NY 12210) ; Animal Limited Evidence IMEMDT IARC Monographs on the Evaluation of Carcinogenic Risk of Chemicals to Man . 41 ,1986,p. 253.(World Health Organization, Internation Agency for Research on Cancer,Lyon, France.: ) (Single copies can be ordered from WHO Publications Centre U.S.A., 49 Sheridan Avenue, Albany, NY 12210) . Reported in EPA TSCA Inventory.
1,1,1-Trifluoro-2-chloroethane Safety Profile
Safety Information of 1,1,1-Trifluoro-2-chloroethane (CAS NO.75-88-7):
Hazard Codes: Xi 
Hazard Note: Irritant
Risk Statements: 59
R59:Dangerous to the ozone layer
Safety Statements: 9-23-59
S9:Keep container in a well-ventilated place
S23:Do not breathe vapour
S59:Refer to manufacturer / supplier for information on recovery / recycling
RIDADR: 1983
TSCA: T
HazardClass: 2.2
A poison by inhalation. Experimental reproductive effects. Questionable carcinogen with experimental carcinogenic data. When heated to decomposition it emits toxic fumes of F−.
1,1,1-Trifluoro-2-chloroethane Specification
1,1,1-Trifluoro-2-chloroethane with CAS Registry Number of 75-88-7 is a colorless, odorless gas. It is also known as 1,1,1-Trifluoroethyl chloride ; 1-Chloro-2,2,2-trifluoroethane ; 2,2,2-Trifluorochloroethane ; 2-Chloro-1,1,1-trifluoroethane ; 4-01-00-00128 (Beilstein Handbook Reference) ; CCRIS 154 ; CFC 133a ; FC 133a ; Forane 133a ; Freon 133a ; Genetron 133a ; HCFC 133a ; HCFC-133a ; HSDB 6949 ; R 133a ; Chloro-1,1,1-trifluoroethane ; Ethane, 2-chloro-1,1,1-trifluoro- ; 1-Chloro-2,2,2-trifluoroethane or Refrigerant gas R 133a [UN1983] [Non-flammable gas] ; Chlorotrifluoroethane ; HCFC-133a ; UN1983 . It can react violently with strong reducing agents and undergo oxidation with strong oxidizing agents and under extremes of temperature.
Related Products
- 10,10'-Bis([1,1'-biphenyl]-4-yl)-9,9'-bianthracene
- 10,10'-Dibromo-9,9'-bianthryl
- 10,10-Dimethylanthrone
- 10,10-Oxybisphenoxarsine
- 10-[1,1'-Biphenyl]-4-yl-2-(1-methylethyl)-9-oxo-9H-thioxanthenium hexafluorophosphate
- 10,11-Dehydroimipramine
- 10,11-Dihydro-11-oxodibenzo[b,f][1,4]thiazepine
- 10,11-Dihydro-2-(4-methyl-1-piperazinyl)-11-(2-athiazolyl)-pyridazino(3,4-b)(1,4)benzoxazepine
- 10,11-DIHYDRO-2-(4-METHYL-1-PIPERAZINYL)-11-(3,4-XYLYL)PYRIDAZINO(3,4b)(1,4)-BENZOXAZEPINE MALEATE
- 10,11-Dihydro-5-(3-dimethylamino-2-methylpropyl)-5h-dibenz (b,f)azepine
- 75887-54-6
- 75888-21-0
- 75890-68-5
- 7589-27-7
- 7589-35-7
- 75893-82-2
- 758-96-3
- 75896-68-3
- 75-89-8
- 75898-26-9
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View