-
Name
2,3-Dichlorophenol
- EINECS 209-399-8
- CAS No. 576-24-9
- Article Data28
- CAS DataBase
- Density 1.458 g/cm3
- Solubility <0.1 g/100 mL at 20℃
- Melting Point 56 °C
- Formula C6H4Cl2O
- Boiling Point 206 °C at 760 mmHg
- Molecular Weight 163.003
- Flash Point 101.6 °C
- Transport Information UN 2020 6.1/PG 3
- Appearance Brown crystals (from ligroin, benzene).
- Safety 26-28-61-45-36/37/39
- Risk Codes 22-36/38-51/53-34
-
Molecular Structure
-
Hazard Symbols
 Xn;
Xn;  Xi;
Xi;  N;
N;  C
C
- Synonyms NSC 60646;
- PSA 20.23000
- LogP 2.69900
Synthetic route

| Conditions | Yield |
|---|---|
| With wild-type cytochrome P450cam In ethanol at 30℃; for 0.0333333h; pH=7.4; Enzyme kinetics; Further Variations:; Reagents; Oxidation; | A 10% B 90% |
| With oxygen; titanium(IV) oxide In perchloric acid at 23℃; for 0.25h; pH=1; Kinetics; Further Variations:; Catalysts; reaction time; UV-irradiation; | |
| With (difluoroboryl)dimethylglyoximatocobalt(II) bis(acetonitrile); water; 3-cyano-1-methylquinolinium perchlorate In acetonitrile at 20℃; for 5h; Inert atmosphere; Irradiation; | |
| Stage #1: 1,2-dichloro-benzene With formic acid; CoO40W12(5-)*16H2O*5K(1+); lithium formate at 25℃; for 3h; Electrochemical reaction; Stage #2: With perchloric acid In diethyl ether; water at 20℃; for 0.166667h; Reagent/catalyst; |
-
-
151169-74-3
2,3-dichlorobenzeneboronic acid

-
-
576-24-9
2,3-dichlorophenol

| Conditions | Yield |
|---|---|
| With 10-methylacridine-3(10H)-one; oxygen; N-ethyl-N,N-diisopropylamine In water at 20℃; for 42h; Irradiation; Green chemistry; | 88% |

| Conditions | Yield |
|---|---|
| With sodium methylate In N,N,N,N,N,N-hexamethylphosphoric triamide at 120℃; | 50% |
-
-
108-43-0
3-monochlorophenol

-
A
-
583-78-8
2,5-dichlorophenol

-
B
-
95-77-2
3,4-dichlorophenol

-
C
-
576-24-9
2,3-dichlorophenol

| Conditions | Yield |
|---|---|
| With oxygen; silica gel; copper dichloride at 350℃; Formation of xenobiotics; Further byproducts.; | A 0.227% B 0.027% C 0.051% D 0.115% |
| With oxygen; silica gel; copper dichloride at 375℃; Formation of xenobiotics; Further byproducts.; | A 0.139% B 0.019% C 0.032% D 0.2629% |

-
-
108-43-0
3-monochlorophenol

-
A
-
583-78-8
2,5-dichlorophenol

-
B
-
95-77-2
3,4-dichlorophenol

-
C
-
576-24-9
2,3-dichlorophenol

-
D
-
95-95-4
2,4,5-trichlorophenol

| Conditions | Yield |
|---|---|
| With oxygen; silica gel; copper dichloride at 300℃; Product distribution; Further Variations:; Temperatures; | A 0.262% B 0.044% C 0.061% D 0.021% |

-
-
56962-00-6
2-amino-3-chlorophenol

-
-
576-24-9
2,3-dichlorophenol

| Conditions | Yield |
|---|---|
| With hydrogenchloride Diazotization.Behandlung der Diazoniumsalz-Loesung mit CuCl2 und konz. wss. HCl; |
-
-
56962-01-7
2-chloro-3-aminophenol

-
-
576-24-9
2,3-dichlorophenol

| Conditions | Yield |
|---|---|
| With hydrogenchloride Diazotization.Behandlung der Diazoniumsalz-Loesung mit Kupfer-Pulver und wss. HCl; |
-
-
67-56-1
methanol

-
-
124-41-4
sodium methylate

-
-
87-61-6
1,2,3-trichlorobenzene

-
A
-
87-65-0
2,6-Dichlorophenol

-
B
-
1984-59-4
2,3-dichloroanisole

-
C
-
576-24-9
2,3-dichlorophenol

| Conditions | Yield |
|---|---|
| at 180℃; im Rohr; |


| Conditions | Yield |
|---|---|
| With methanol; sodium methylate at 180℃; |

| Conditions | Yield |
|---|---|
| With N-chloro-3-methyl-2,6-diphenylpiperidin-4-one; hydrogen cation In ethanol; water at 30℃; Product distribution; Thermodynamic data; ΔH(excit.), ΔS(excit.); | |
| With N-chloro-3-methyl-2,6-diphenylpiperidin-4-one; hydrogen cation In ethanol; water at 30℃; |
-
-
79435-99-7
5,6-Dichlorcyclohexan-3,5-dien-1,2-diol

-
A
-
591-35-5
3,5-dichlorophenol

-
B
-
95-77-2
3,4-dichlorophenol

-
C
-
576-24-9
2,3-dichlorophenol

| Conditions | Yield |
|---|---|
| With hydrogenchloride at 23℃; for 24h; different reagent; |

-
-
95-50-1
1,2-dichloro-benzene

-
A
-
87-65-0
2,6-Dichlorophenol

-
B
-
95-77-2
3,4-dichlorophenol

-
C
-
576-24-9
2,3-dichlorophenol

| Conditions | Yield |
|---|---|
| With Pseudomonas putida 50802 at 30℃; for 100h; Mechanism; different reagents; |


-
-
576-24-9
2,3-dichlorophenol

| Conditions | Yield |
|---|---|
| With sulfuric acid Durchleiten von Wasserdampf; |

-
-
576-24-9
2,3-dichlorophenol

| Conditions | Yield |
|---|---|
| With sulfuric acid |

| Conditions | Yield |
|---|---|
| Diazotierung des gebildeten Amins und Behandlung der Diazoniumsalz-Loesung mit Kupfer-Pulver und wss. HCl; |

-
-
288-32-4
1H-imidazole

-
-
61925-85-7
2,3-dichlorophenyl acetate

-
A
-
2466-76-4
N-Acetylimidazole

-
B
-
576-24-9
2,3-dichlorophenol

| Conditions | Yield |
|---|---|
| With sodium dodecyl-sulfate In water; acetonitrile at 25℃; pH=8.92; Kinetics; Acetylation; |

-
-
108-43-0
3-monochlorophenol

-
A
-
583-78-8
2,5-dichlorophenol

-
B
-
95-77-2
3,4-dichlorophenol

-
C
-
576-24-9
2,3-dichlorophenol

| Conditions | Yield |
|---|---|
| With sodium hydroxide; sodium hypochlorite; sodium nitrate at 24.85℃; Kinetics; | |
| With sodium hydroxide; tert-butylhypochlorite; sodium chloride at 24.85℃; Kinetics; Further Variations:; pH-values; conc.; | |
| With 1,3-dichloro-5,5-dimethylhydantoin; diisopropylamine hydrochloride In toluene at 0℃; for 4h; Darkness; Overall yield = 59 %; regioselective reaction; |

-
-
935-95-5
2,3,5,6-tetrachlorophenol

-
A
-
87-65-0
2,6-Dichlorophenol

-
B
-
933-75-5
2,3,6-trichlorophenol

-
C
-
95-57-8
2-monochlorophenol

-
D
-
576-24-9
2,3-dichlorophenol

| Conditions | Yield |
|---|---|
| With tetraethylammonium bromide In methanol Electrolysis; Further byproducts given. Title compound not separated from byproducts; |


-
-
576-24-9
2,3-dichlorophenol

| Conditions | Yield |
|---|---|
| With hydrogenchloride; air; sewage sludge ash at 400℃; Formation of xenobiotics; |

| Conditions | Yield |
|---|---|
| With potassium chloride; water at 25℃; pH=12.7; Kinetics; |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: tetraethylmmonium bromide / methanol / Electrolysis 2: tetraethylmmonium bromide / methanol / Electrolysis View Scheme |
-
-
92-59-1
N-benzyl-N-ethylaniline

-
-
1527472-72-5
2,3-dichlorophenyl picolinate

-
A
-
576-24-9
2,3-dichlorophenol

-
B
-
1199382-83-6
N-ethyl-N-phenylpicolinamide

-
D
-
100-52-7
benzaldehyde

| Conditions | Yield |
|---|---|
| In chlorobenzene at 115℃; for 24h; Green chemistry; |

-
-
576-24-9
2,3-dichlorophenol

-
-
107-30-2
chloromethyl methyl ether

-
-
118166-34-0
2,3-dichlorophenyl methoxymethyl ether

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetonitrile at 50 - 60℃; for 0.666667h; | 100% |

| Conditions | Yield |
|---|---|
| With potassium carbonate In 1,4-dioxane at 60℃; for 48h; Green chemistry; | 100% |

| Conditions | Yield |
|---|---|
| Stage #1: 2,3-dichlorophenol With potassium carbonate In acetone for 0.5h; Stage #2: dimethyl sulfate In acetone at 60℃; for 3h; | 99.1% |
| Stage #1: 2,3-dichlorophenol With potassium hydroxide In toluene for 0.5h; Stage #2: dimethyl sulfate In toluene at 100℃; for 5h; Stage #3: With potassium hydroxide In toluene for 1h; Reflux; | 96.6% |
| With alkali |

| Conditions | Yield |
|---|---|
| With 9,10-dihydroanthracene; water at 356.85℃; for 3h; Kinetics; | A 99% B 0.3% |

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetonitrile at 25℃; for 18h; | 99% |

| Conditions | Yield |
|---|---|
| With potassium methanolate In N,N-dimethyl-formamide at 155℃; for 0.5h; Temperature; Reagent/catalyst; Microwave irradiation; | 98.5% |
-
-
576-24-9
2,3-dichlorophenol

-
-
105-36-2
ethyl bromoacetate

-
-
37536-92-8
(dichloro-2,3 phenoxy)acetate d'ethyle

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetone at 50℃; for 3h; | 98% |
| With sodium ethanolate In ethanol for 2.5h; Heating; | 86% |
| Stage #1: 2,3-dichlorophenol With water; potassium carbonate for 0.2h; Metallation; Irradiation; Stage #2: ethyl bromoacetate In benzene at 50℃; for 0.25h; Substitution; Irradiation; | 82% |

| Conditions | Yield |
|---|---|
| With potassium carbonate In butanone for 24h; Heating; | 98% |
-
-
576-24-9
2,3-dichlorophenol

-
-
541-41-3
chloroformic acid ethyl ester

-
-
207238-21-9
Carbonic acid 2,3-dichloro-phenyl ester ethyl ester

| Conditions | Yield |
|---|---|
| With sodium hydroxide In water for 1.5h; Ambient temperature; | 98% |
-
-
576-24-9
2,3-dichlorophenol

-
-
115314-14-2
(2s)-(+)-glycidyl 3-nitrobenzenesulfonate

-
-
134598-06-4
(2S)-2-([(2,3-dichlorophenyl)oxy]methyl)oxirane

| Conditions | Yield |
|---|---|
| Stage #1: 2,3-dichlorophenol With potassium carbonate In acetone for 0.5h; Reflux; Stage #2: (2s)-(+)-glycidyl 3-nitrobenzenesulfonate Cooling; Reflux; | 96% |

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetone for 7h; Heating; | 95% |
-
-
576-24-9
2,3-dichlorophenol

-
-
350-46-9
4-Fluoronitrobenzene

-
-
82239-20-1
1,2-dichloro-3-(4-nitrophenoxy)benzene

| Conditions | Yield |
|---|---|
| With potassium carbonate In water; N,N-dimethyl-formamide at 110℃; for 5h; | 95% |

| Conditions | Yield |
|---|---|
| With sodium hydroxide In neat (no solvent) at 90℃; for 6h; Green chemistry; | 95% |

| Conditions | Yield |
|---|---|
| With N,N-dimethyl acetamide; triethylamine at 100℃; under 15001.5 Torr; for 4h; Autoclave; | 94% |


| Conditions | Yield |
|---|---|
| With caesium carbonate In dimethyl sulfoxide at 95℃; for 12h; | 93% |
| Stage #1: 2,3-dichlorophenol With potassium carbonate In N,N-dimethyl-formamide at 20℃; for 0.5h; Stage #2: Bromoacetaldehyde diethyl acetal In N,N-dimethyl-formamide for 72h; Reflux; |
-
-
576-24-9
2,3-dichlorophenol

-
-
115314-14-2, 115314-17-5
(R)-glycidyl nosylate

-
-
198226-59-4
(2R)-2-([(2,3-dichlorophenyl)oxy]methyl)oxirane

| Conditions | Yield |
|---|---|
| Stage #1: 2,3-dichlorophenol With potassium carbonate In acetone for 0.5h; Reflux; Stage #2: (R)-glycidyl nosylate Cooling; Reflux; | 92% |
-
-
576-24-9
2,3-dichlorophenol

-
-
105-39-5
chloroacetic acid ethyl ester

-
-
2976-74-1
2,3-dichlorophenoxyacetic acid

| Conditions | Yield |
|---|---|
| Stage #1: 2,3-dichlorophenol; chloroacetic acid ethyl ester With potassium carbonate; potassium iodide In water; N,N-dimethyl-formamide at 100℃; for 0.0666667h; Microwave irradiation; Stage #2: With sodium hydroxide for 0.0833333h; Microwave irradiation; | 91% |
-
-
5271-67-0
2-Thiophenecarbonyl chloride

-
-
576-24-9
2,3-dichlorophenol

-
-
55901-84-3
Thiophen-2-carbonsaeure-(2,3-dichlor)-phenylester

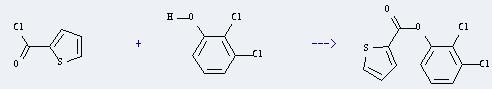
| Conditions | Yield |
|---|---|
| With pyridine In tetrahydrofuran for 1h; Heating; | 90% |
-
-
16801-19-7
2-[2-(vinyloxy)ethoxymethyl]oxirane

-
-
576-24-9
2,3-dichlorophenol

-
-
98934-98-6
2-{2-[1-(2,3-Dichloro-phenoxy)-ethoxy]-ethoxymethyl}-oxirane

| Conditions | Yield |
|---|---|
| With heptafluorobutyric Acid at 50 - 80℃; | 90% |

| Conditions | Yield |
|---|---|
| With sodium In methanol for 7h; Heating; | 90% |
-
-
576-24-9
2,3-dichlorophenol

-
-
10352-63-3
chloromethyl sodium sulfonate

| Conditions | Yield |
|---|---|
| With sodium hydroxide; water for 0.00833333h; Condensation; Irradiation; | 90% |

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide at 70℃; for 3h; Inert atmosphere; | 90% |
| With potassium carbonate In N,N-dimethyl-formamide at 20℃; for 5h; Inert atmosphere; |

| Conditions | Yield |
|---|---|
| With borane-ammonia complex In water; isopropyl alcohol at 50℃; for 5h; Sealed tube; | 90% |
| In cyclohexane for 24h; UV-irradiation; | 9 %Chromat. |
-
-
576-24-9
2,3-dichlorophenol

-
-
79-07-2
Chloroacetamide

-
-
57245-31-5
8-chloro-3,4-dihydro-2H-1,4-benzoxazin-3-one

| Conditions | Yield |
|---|---|
| With caesium carbonate at 130℃; Microwave irradiation; | 89% |
-
-
576-24-9
2,3-dichlorophenol

-
-
14301-31-6
2-chloro-N-(2-(3,4-dimethoxyphenyl)ethyl)acetamide

-
-
223686-69-9
2-(2,3-dichloro-phenoxy)-N-[2-(3,4-dimethoxy-phenyl)-ethyl]-acetamide

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetonitrile for 6h; Heating; | 88% |
-
-
576-24-9
2,3-dichlorophenol

-
-
88-10-8
N,N-diethylcarbamyl chloride

-
-
1225288-00-5
2,3-dichlorophenyl N,N-diethyl O-carbamate

| Conditions | Yield |
|---|---|
| Stage #1: 2,3-dichlorophenol With potassium carbonate In acetonitrile for 0.5h; Stage #2: N,N-diethylcarbamyl chloride In acetonitrile for 5h; Reflux; | 87% |
-
-
7462-74-0
2-bromo-2-methylpropanamide

-
-
576-24-9
2,3-dichlorophenol

-
-
1226261-49-9
2-(2,3-dichlorophenoxy)-2-methylpropanamide

| Conditions | Yield |
|---|---|
| Stage #1: 2,3-dichlorophenol With sodium hydroxide In N,N-dimethyl-formamide at 20℃; for 1h; Inert atmosphere; Stage #2: 2-bromo-2-methylpropanamide In N,N-dimethyl-formamide at 20℃; for 2h; Inert atmosphere; | 87% |
-
-
350-30-1
3-chloro-4-fluoronitrobenzene

-
-
576-24-9
2,3-dichlorophenol

-
-
317336-87-1
3.chloro-4-(2,3-dichlorophenoxy)phenylamine

| Conditions | Yield |
|---|---|
| Stage #1: 3-chloro-4-fluoronitrobenzene; 2,3-dichlorophenol With potassium hydrogencarbonate In N,N-dimethyl-formamide at 100℃; for 1.5h; Stage #2: With hydrogenchloride; iron In ethanol; water at 110℃; for 3h; Stage #3: With sodium hydrogencarbonate In water | 86% |
2,3-Dichlorophenol Consensus Reports
2,3-Dichlorophenol Specification
The IUPAC name of this chemical is 2,3-Dichlorophenol. With the CAS registry number 576-24-9 and EINECS registry number 209-399-8, it is also named as Phenol,2,3-dichloro-. In addition, the molecular formula is C6H4Cl2O and the molecular weight is 163.001. It is a kind of light brown crystalline solid and belongs to the classes of Aromatic Phenols; Phenol Thiophenol Mercaptan; Chlorine Compounds; Phenols; Alphabetic; D; DIA - DIC; Organic Building Blocks; Oxygen Compounds. What's more, it is stable and incompatible with strong oxidizing agents, acid chlorides and acid anhydrides.
Physical properties about this chemical are: (1)ACD/LogP: 2.83; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): 2.83; (4)ACD/LogD (pH 7.4): 2.59; (5)ACD/BCF (pH 5.5): 82.59; (6)ACD/BCF (pH 7.4): 47.9; (7)ACD/KOC (pH 5.5): 817.7; (8)ACD/KOC (pH 7.4): 474.31; (9)#H bond acceptors: 1; (10)#H bond donors: 1; (11)#Freely Rotating Bonds: 1; (12)Polar Surface Area: 9.23 Å2; (13)Index of Refraction: 1.593; (14)Molar Refractivity: 37.92 cm3; (15)Molar Volume: 111.7 cm3; (16)Polarizability: 15.03 ×10-24cm3; (17)Surface Tension: 47.8 dyne/cm; (18)Density: 1.458 g/cm3; (19)Flash Point: 101.6 °C; (20)Enthalpy of Vaporization: 46.03 kJ/mol; (21)Boiling Point: 206 °C at 760 mmHg; (22)Vapour Pressure: 0.169 mmHg at 25°C.
Preparation of 2,3-Dichlorophenol: it can be prepared by 1,2,3-trichlorobenzene. The 1,2,3-trichlorobenzene will become into salt by sulphonate. Then by means of high pressure hydrolysis, you can get 3,4-dichlorine-2-hydroxybenzenesulfonic acid. The sulfonateyl will be took off through sulfuric acid hydrolysis at last. In addition, it can be prepared by 2,3-dichloro-anisole. This reaction will need reagent sodium methoxide and solvent hexamethylphosphoric acid triamide. The yield is about 50% at reaction temperature of 50 °C.
.jpg)
Uses of 2,3-Dichlorophenol: it can be usd as intermediates to synthetize uric acid. In addition, it can react with thiophene-2-carbonyl chloride to get Thiophen-2-carbonsaeure-(2,3-dichlor)-phenylester. This reaction will need reagent pyridine and solvent tetrahydrofuran. The reaction time is one hour by heating. The yield is about 90%.
When you are using this chemical, please be cautious about it as the following:
This chemical is harmful if swallowed, irritating to eyes and skin. And it is toxic to aquatic organisms, may cause long-term adverse effects in the aquatic environment. In addition, it may cause burns. It is refer to special instructions/safety data sheets. During using it, wear suitable protective clothing, gloves and eye/face protection. In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. If contact it with skin, wash immediately with plenty of soap-suds. In case of accident or if you feel unwell, seek medical advice immediately (show the label whenever possible.). At last, avoid release to the environment.
You can still convert the following datas into molecular structure:
(1)SMILES: Clc1c(O)cccc1Cl
(2)InChI: InChI=1/C6H4Cl2O/c7-4-2-1-3-5(9)6(4)8/h1-3,9H
(3)InChIKey: UMPSXRYVXUPCOS-UHFFFAOYAZ
The toxicity data is as follows:
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| mouse | LD50 | oral | 2376mg/kg (2376mg/kg) | BEHAVIORAL: TREMOR BEHAVIORAL: CONVULSIONS OR EFFECT ON SEIZURE THRESHOLD LUNGS, THORAX, OR RESPIRATION: RESPIRATORY STIMULATION | Toxicology Letters. Vol. 29, Pg. 39, 1985. |
Related Products
- 20,22-DIHYDRODIGITOXIN
- 20,29,30-Trinorlupane,(17alpha)-
- 20-ETHYL-6-β,8-DIHYDROXY-1-α-METHOXY-4-METHYLHETERATISAN-14-ONE
- 20-Ethylprostaglandin F2-alpha
- 20-Isopropylcholanthrene
- 20-METHYLCHOLANTHREN-15-ONE
- 20-METHYLCHOLANTHRENE PICRATE
- 20-METHYLCHOLANTHRENE-TRINITRO-BENZENE
- 20(S)-Ginsenoside C-K
- 2,10-DIFLUOROBENZO(rst)PENTAPHENE
- 57-62-5
- 5762-56-1
- 57625-74-8
- 57625-97-5
- 576-26-1
- 5762-64-1
- 57627-75-5
- 57632-67-4
- 5763-44-0
- 57634-55-6
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View