-
Name
3-Cyclohexene-1-carboxaldehyde
- EINECS 202-858-3
- CAS No. 100-50-5
- Article Data62
- CAS DataBase
- Density 0.94 g/cm3
- Solubility 11.5g/L at 20℃
- Melting Point 1-3 °C
- Formula C7H10O
- Boiling Point 105 °C at 760 mmHg
- Molecular Weight 110.156
- Flash Point 57.2 °C
- Transport Information UN 2498 3/PG 3
- Appearance slightly yellowish-green liquid
- Safety 26-36-45
- Risk Codes 21-36/37/38
-
Molecular Structure
-
Hazard Symbols
 Xn
Xn
- Synonyms 1,2,3, 6-Tetrahydrobenzaldehyde;1,2,5,6-Tetrahydrobenzaldehyde;cyclohex-3-ene-1-carbaldehyde;1-Formyl-3-cyclohexene;Cyclohexene-4-carboxaldehyde;3-cyclohexeneformaldehyde;3-Cyclohexene-1-caboxaldehyde;3-cyclohexene-1-carboxaldehyde;1,2,3,6-tetrahydroBenzaldehyde; 4-formyl-1-cyclohexene;3-cyclohe;supply 3-cyclohexene-1-carboxaldehyde;3-Cyclohexen-1-aldehyde;(1R)-cyclohex-3-ene-1-carbaldehyde;(1S)-cyclohex-3-ene-1-carbaldehyde;3-Cyclohexen-1-Carboxaldehyde;
- PSA 17.07000
- LogP 1.54160
Synthetic route

| Conditions | Yield |
|---|---|
| With erbium(III) triflate at 20℃; for 2h; Diels-Alder reaction; Ionic liquid; regioselective reaction; | 99% |
| In 1,4 dimethylcyclohexane at 150℃; under 30003 Torr; for 3h; Solvent; Temperature; Inert atmosphere; Autoclave; | 90% |
| K10-Fe(III) In dichloromethane at 20℃; for 6h; | 83% |
-
-
53178-49-7
2-(3-cyclohexenyl)-1,3-dithiane

-
-
100-50-5
3-Cyclohexene-1-carboxaldehyde

| Conditions | Yield |
|---|---|
| With silica gel; copper(II) nitrate In tetrachloromethane for 0.416667h; Ambient temperature; | 97% |
| With p-benzoquinone; sodium iodide In water; acetonitrile at 100℃; for 72h; | 74 %Chromat. |
-
-
72581-32-9, 1679-51-2
cyclohex-3-enylmethanol

-
-
100-50-5
3-Cyclohexene-1-carboxaldehyde

| Conditions | Yield |
|---|---|
| With titanium(IV) oxide; oxygen at 29.84℃; under 760.051 Torr; for 24h; Sealed tube; Irradiation; | 96% |
| With 2,2,6,6-tetramethyl-piperidine-N-oxyl; periodic acid In dichloromethane at 20℃; for 2h; | 78% |
| With tetrabutylammomium bromide; dihydrogen peroxide; [Ru3O(OAc)6(MeOH)3](1+)*AcO(1-) In dichloromethane at 20℃; for 24h; | 45% |
| In dichloromethane |

-
-
100-50-5
3-Cyclohexene-1-carboxaldehyde

| Conditions | Yield |
|---|---|
| With silica gel; copper(II) nitrate In tetrachloromethane for 1h; Ambient temperature; | 96% |
-
-
99-49-0, 22327-39-5
Carvone

-
-
100-50-5
3-Cyclohexene-1-carboxaldehyde

| Conditions | Yield |
|---|---|
| With LaNi5 hydride In tetrahydrofuran; methanol for 8h; Mechanism; Ambient temperature; other alkynes, alkenes, aldehydes, ketones; other rare earth alloys; var. reaction time and temp.; | 91% |

| Conditions | Yield |
|---|---|
| With 4Na(1+)*6H(1+)*NiMo6O24(10-)=Na4H6NiMo6O24; oxygen In water; acetonitrile at 20℃; under 760.051 Torr; for 12h; Irradiation; | 87% |
-
-
151259-43-7
2-Allyl-hex-5-enal

-
-
100-50-5
3-Cyclohexene-1-carboxaldehyde

| Conditions | Yield |
|---|---|
| Cl2(PCy3)(N,N'-(Mes)2-imidazolidin-2-yl)Ru=CHC6H5 In benzene at 20℃; for 1h; | 82% |

| Conditions | Yield |
|---|---|
| With hypoiodous acid; acetone at -40 - 20℃; | A 72% B n/a |

| Conditions | Yield |
|---|---|
| With diethylaluminium chloride; triphenylphosphine In dichloromethane at 25℃; for 1h; | 61% |
| With diethylaluminium chloride; triphenylphosphine In dichloromethane at 25℃; for 1h; | 61% |
| In hexane at 410℃; |
-
-
498-66-8
norborn-2-ene

-
A
-
497-38-1
Norbornan-2-on

-
B
-
100-50-5
3-Cyclohexene-1-carboxaldehyde

-
C
-
278-74-0, 3146-39-2, 57378-36-6
exo-2,3-epoxynorbornane

-
D
-
844874-18-6
exo-2,3-epoxynorbornane

| Conditions | Yield |
|---|---|
| With iodosylbenzene; TFTEP*FeCl In dichloromethane at 0℃; Yields of byproduct given; | A 3.9% B 0.3% C n/a D n/a |
| With iodosylbenzene; OEP*FeCl In dichloromethane at 0℃; Yield given. Yields of byproduct given; | A 1.6% B 0.2% C n/a D n/a |
| With iodosylbenzene; TFTEP*MnCl In dichloromethane at 0℃; Yield given. Yields of byproduct given; | A 0.8% B 0.2% C n/a D n/a |


| Conditions | Yield |
|---|---|
| With oxygen; bis(ethylene)rhodium acetylacetonate In toluene at 80℃; under 37503 Torr; for 1h; | A 1% B 0.3% |
| With oxygen; bis(ethylene)rhodium acetylacetonate In toluene at 80℃; under 37503 Torr; for 1h; | A 1% B 0.3% |

| Conditions | Yield |
|---|---|
| beim kurzen Kochen des Reaktionsprodukts mit verd. Schwefelsaeure; |

-
-
50552-13-1
Cyclohex-3-enylmethylnitrit

-
-
100-50-5
3-Cyclohexene-1-carboxaldehyde

| Conditions | Yield |
|---|---|
| In benzene for 10h; Irradiation; |
-
-
498-66-8
norborn-2-ene

-
A
-
100-50-5
3-Cyclohexene-1-carboxaldehyde

-
B
-
278-74-0, 3146-39-2, 57378-36-6
exo-2,3-epoxynorbornane

-
C
-
844874-18-6
exo-2,3-epoxynorbornane

| Conditions | Yield |
|---|---|
| With iodosylbenzene; Mn(III)(SAP)NCS In acetonitrile Product distribution; other alkenes, var. additives (surfactants), var. pyridine concentrations; | |
| With iodosylbenzene; Mn(T4PyP)OAc In acetonitrile at 30℃; var. olefins; other catalyst; |

-
-
498-66-8
norborn-2-ene

-
A
-
497-38-1
Norbornan-2-on

-
B
-
100-50-5
3-Cyclohexene-1-carboxaldehyde

-
C
-
844874-18-6
exo-2,3-epoxynorbornane

| Conditions | Yield |
|---|---|
| With pyrimido<5.4-g>pteridine 10-oxide In acetonitrile Product distribution; Mechanism; Ambient temperature; Irradiation; | A 4 % Chromat. B n/a C 42 % Chromat. |

-
-
111292-74-1
Bicyclo<3.1.1>hept-2-en-exo-6-ol

-
-
100-50-5
3-Cyclohexene-1-carboxaldehyde

| Conditions | Yield |
|---|---|
| With sodium hydroxide |
-
-
201230-82-2
carbon monoxide

-
-
1165952-92-0
cyclohexa-1,4-diene

-
-
100-50-5
3-Cyclohexene-1-carboxaldehyde

| Conditions | Yield |
|---|---|
| With rhodium (triphenylphosphine)acetylacetonatecarbonyl; hydrogen; triphenylphosphine at 80℃; under 7600 Torr; for 3h; | 71 % Spectr. |
| With hydrogen; rhodium 1.) from -196 deg C to RT, 2.) toluene, 50 deg C, 48 h, 70 atm; Yield given. Multistep reaction; |
-
-
201230-82-2
carbon monoxide

-
-
1165952-91-9
cyclohexa-1,3-diene

-
A
-
1192-88-7
1-cyclohexene-1-carboxaldehyde

-
B
-
100-50-5
3-Cyclohexene-1-carboxaldehyde

| Conditions | Yield |
|---|---|
| With triphenyl phosphite; acetylacetonatobis(triphenylphosphite)rhodium; hydrogen at 80℃; under 7600 Torr; for 3h; Title compound not separated from byproducts; | A 31 % Spectr. B 42 % Spectr. |
| With rhodium (triphenylphosphine)acetylacetonatecarbonyl; hydrogen; triphenylphosphine at 80℃; under 7600 Torr; for 3h; Title compound not separated from byproducts; | A 43 % Spectr. B 30 % Spectr. |


| Conditions | Yield |
|---|---|
| With acetic acid at 250℃; |

| Conditions | Yield |
|---|---|
| With toluene Behandeln des Reaktionsproduktes mit verd. Schwefelsaeure; |
-
-
201230-82-2
carbon monoxide

-
-
1165952-91-9
cyclohexa-1,3-diene

-
A
-
33424-83-8
1,4-cyclohexanedicarboxaldehyde

-
B
-
100-50-5
3-Cyclohexene-1-carboxaldehyde

-
C
-
55309-54-1
cis/trans-1,3-cyclohexanedicarboxaldehyde

-
D
-
42540-33-0
cyclohex-2-enecarbaldehyde

| Conditions | Yield |
|---|---|
| With Rh4(CO)14; hydrogen In hexane at 19.85℃; Product distribution; Hydroformylation; reaction was studied by in-situ high-pressure IR spectroscopy; |

-
-
3872-23-9
Copper(I) trifluoromethanethiolate

-
-
844874-18-6
exo-2,3-epoxynorbornane

-
A
-
497-38-1
Norbornan-2-on

-
B
-
100-50-5
3-Cyclohexene-1-carboxaldehyde

| Conditions | Yield |
|---|---|
| In toluene at 100 - 110℃; Title compound not separated from byproducts; |


| Conditions | Yield |
|---|---|
| Stage #1: 4-ethenylcyclohexene With cis-[Ru(2,9-Me2phen)2(OH2)2](PF6)2 In acetonitrile at 55℃; for 0.5h; Stage #2: With dihydrogen peroxide In acetonitrile for 8h; |
-
-
72581-32-9, 1679-51-2
cyclohex-3-enylmethanol

-
A
-
4771-80-6
1,2,5,6-tetrahydrobenzoic acid

-
B
-
100-50-5
3-Cyclohexene-1-carboxaldehyde

| Conditions | Yield |
|---|---|
| With peracetic acid; dihydrogen peroxide; palladium on activated charcoal In dichloromethane; water at 59.84℃; for 1h; | |
| With peracetic acid; dihydrogen peroxide; Mn(III)AlPO-5 In dichloromethane; water at 89.84℃; for 1h; |
-
-
72581-32-9, 1679-51-2
cyclohex-3-enylmethanol

-
B
-
100-50-5
3-Cyclohexene-1-carboxaldehyde

| Conditions | Yield |
|---|---|
| With peracetic acid; dihydrogen peroxide; palladium on activated charcoal In water; acetonitrile at 59.84℃; for 1h; |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 1) NaH, DMSO / 1) 25 deg C, 15 min., DMSO; 2) 25 deg C, 1 h 2: 61 percent / Et2AlCl, PPh3 / CH2Cl2 / 1 h / 25 °C View Scheme |
-
-
72581-32-9, 1679-51-2
cyclohex-3-enylmethanol

-
A
-
100-50-5
3-Cyclohexene-1-carboxaldehyde

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: NaNO2, H2SO4 / H2O / -10 °C 2: benzene / 10 h / Irradiation View Scheme |
-
-
15111-56-5
ethyl-3-cyclohexene-1-carboxylate

-
A
-
100-50-5
3-Cyclohexene-1-carboxaldehyde

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: LiAlH4 / diethyl ether / Heating 2: NaNO2, H2SO4 / H2O / -10 °C 3: benzene / 10 h / Irradiation View Scheme |

| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid In benzene for 2h; Heating; | 100% |

| Conditions | Yield |
|---|---|
| With C4H11FeMo6NO24(3-)*3C16H36N(1+); water; oxygen; sodium carbonate at 50℃; under 760.051 Torr; for 8h; Green chemistry; | 99% |
| With 4H3N*4H(1+)*CuMo6O18(OH)6(4-); water; oxygen; sodium carbonate at 50℃; under 760.051 Torr; for 12h; | 99% |
| With 2-mesityl-6,7-dihydro-5H-pyrrolo[2,1-c][1,2,4]triazol-2-ium tetrafluoroborate; 1,3,4,6,7,8-hexahydro-2H-pyrimido[1,2-a]pyrimidine In acetonitrile at 20℃; | 96% |
-
-
100-50-5
3-Cyclohexene-1-carboxaldehyde

-
-
72581-32-9, 1679-51-2
cyclohex-3-enylmethanol

| Conditions | Yield |
|---|---|
| Stage #1: 3-Cyclohexene-1-carboxaldehyde With lithium aluminium tetrahydride In tetrahydrofuran at 0 - 20℃; for 3h; Inert atmosphere; Stage #2: With sodium hydroxide In water at 0℃; for 12h; Inert atmosphere; | 99% |
| With 1-Methylpyrrolidine; C13H12MnN3O5; hydrogen In tetrahydrofuran at 100℃; under 37503.8 Torr; for 24h; Autoclave; Glovebox; | 98% |
| With dmap; formic acid; hexarhodium hexadecacarbonyl; carbon monoxide In tetrahydrofuran at 30℃; under 3800 Torr; for 5h; | 93% |
-
-
100-50-5
3-Cyclohexene-1-carboxaldehyde

-
-
2611-00-9
3-cyclohexene-1-carboxylic acid 3-cyclohexen-1-ylmethyl ester

| Conditions | Yield |
|---|---|
| With trimethylaluminum; isopropyl alcohol; 2,7-dimethyl-1,8-biphenylenediol In hexane; toluene at 21℃; for 0.25h; Tishchenko reaction; | 99% |
| With (2,7-dimethyl-1,8-biphenylenedioxy)-bis(diisopropoxyaluminum) In toluene at 21℃; for 0.25h; Dimerization; Tishchenko reaction; | 98% |
| i-PrOAl 1,1'-biphenyl-2-oxy-2'-perfluorooctanesulfonamide In toluene at 20℃; for 0.5h; Tishchenko reaction; | 95% |
-
-
589-09-3
N-allyl-N-phenylamine

-
-
100-50-5
3-Cyclohexene-1-carboxaldehyde

-
-
100747-84-0
N-<(1-allyl-3-cyclohexenyl)methylene>aniline

| Conditions | Yield |
|---|---|
| With Pd(PPh3); trifluoroacetic acid In benzene at 80℃; for 20h; | 99% |
-
-
100-50-5
3-Cyclohexene-1-carboxaldehyde

-
-
25015-63-8
4,4,5,5-tetramethyl-[1,3,2]-dioxaboralane

| Conditions | Yield |
|---|---|
| With FeBr2*2THF; C30H46N4Si2(2+) In acetonitrile at 20℃; for 12h; Glovebox; Inert atmosphere; Sealed tube; | 99% |
| With C14H24N4Si(1+)*I(1-) In benzene-d6 at 24℃; for 0.166667h; Catalytic behavior; Inert atmosphere; Schlenk technique; regioselective reaction; | 86% |
| With [((2,6-(i-Pr)2C6H3)NC(=CH2)CH=C(Me)N(2,6-(i-Pr)2C6H3))Ge] In benzene-d6 at 60℃; for 24h; Inert atmosphere; Glovebox; Sealed tube; chemoselective reaction; | 98 %Spectr. |
| With C18H21La In tetrahydrofuran at 20℃; for 0.166667h; Inert atmosphere; | |
| With tris(η5-methylcyclopentadienyl)lanthanum(III) In tetrahydrofuran at 20℃; for 0.166667h; Catalytic behavior; Schlenk technique; Glovebox; Inert atmosphere; chemoselective reaction; | 99 %Spectr. |

| Conditions | Yield |
|---|---|
| With tin(IV) chloride In dichloromethane for 0.166667h; Ambient temperature; | 98% |

| Conditions | Yield |
|---|---|
| With magnesium sulfate In dichloromethane for 0.5h; Ambient temperature; | 98% |
| In toluene at 110 - 115℃; Dean-Stark; |

| Conditions | Yield |
|---|---|
| With (triphenylphosphine)copper(I) hydride hexamer In toluene at 20℃; for 2h; | 98% |

| Conditions | Yield |
|---|---|
| With bismuth(lll) trifluoromethanesulfonate; acetylhydroxamic acid In acetonitrile for 16h; Reflux; | 97% |
| With ammonium acetate; acetic acid; 4-acetylamino-2,2,6,6-tetramethylpiperidine-1-oxoammonium tetrafluoroborate at 70℃; for 12h; Inert atmosphere; | 94% |
| Stage #1: 3-Cyclohexene-1-carboxaldehyde With hydroxylamine hydrochloride; triethylamine In acetonitrile at 80℃; Stage #2: With phthalic anhydride In acetonitrile at 25 - 80℃; for 18h; | 64% |
| Multi-step reaction with 2 steps 1: 94 percent / hydroxylamine hydrochloride, sodium acetate / H2O / 2 h / 45 °C 2: 77 percent / acetic acid anhydride / 1 h / Ambient temperature View Scheme | |
| Multi-step reaction with 2 steps 1: acetylhydroxamic acid; boron trifluoride diethyl etherate / methanol / Reflux 2: bismuth(lll) trifluoromethanesulfonate / acetonitrile / 8 h / Reflux View Scheme |

| Conditions | Yield |
|---|---|
| With palladium/alumina; hydrogen In hexane at 325℃; Flow reactor; Green chemistry; | A 3.6% B 96.4% |
| With hydrogen In hexane at 325℃; Flow reactor; Green chemistry; | A 83.4% B 16.4% |
| In octane at 300℃; for 1h; Reagent/catalyst; Temperature; | A 82% B 10% |
| In octane at 200℃; for 1h; | A 25% B 44% |
-
-
107-21-1
ethylene glycol

-
-
100-50-5
3-Cyclohexene-1-carboxaldehyde

-
-
28193-08-0
2-(3-cyclohexenyl)-1,3-dioxolane

| Conditions | Yield |
|---|---|
| With Envirocat EPZ10 supported reagent for 0.0333333h; microwave irradiation; | 96% |
| With KU-2 cation exchanger H(1+) In toluene at 130℃; | 87% |
| With ammonium chloride; benzene | |
| With ammonium chloride; benzene |
-
-
106-95-6
allyl bromide

-
-
100-50-5
3-Cyclohexene-1-carboxaldehyde

-
-
91055-81-1
α-(2-propenyl)-3-cyclohexen-1-methanol

| Conditions | Yield |
|---|---|
| With tin; hydrogen bromide In diethyl ether; water Ambient temperature; | 96% |
| With manganese; cyclopentadienyl titanium(IV) trichloride; trimethylsilyl bromide; triethylamine hydrobromide In tetrahydrofuran Inert atmosphere; | 90% |
| (i) Mg, (ii) /BRN= 774001/; Multistep reaction; | |
| With magnesium In diethyl ether |
-
-
16695-14-0
Malonic acid monomethyl ester

-
-
100-50-5
3-Cyclohexene-1-carboxaldehyde

-
-
124939-60-2
(propene-2 oate de methyle) yl-4 cyclohexene trans

| Conditions | Yield |
|---|---|
| With piperidine In pyridine RT, overnight, reflux, 2 h; | 96% |
-
-
100-50-5
3-Cyclohexene-1-carboxaldehyde

| Conditions | Yield |
|---|---|
| With chloro-trimethyl-silane; zinc; bis(cyclopentadienyl)vanadium dichloride In tetrahydrofuran at 20℃; for 13h; Reduction; | 96% |
| With samarium; trimethylsilyl bromide In tetrahydrofuran at 20℃; for 0.5h; Inert atmosphere; | 64% |
-
-
33729-92-9
tert-butyldiphenylsilane

-
-
100-50-5
3-Cyclohexene-1-carboxaldehyde

| Conditions | Yield |
|---|---|
| With (triphenylphosphine)copper(I) hydride hexamer In toluene at 40℃; for 6h; | 96% |
-
-
557-93-7
2-bromoprop-1-ene

-
-
100-50-5
3-Cyclohexene-1-carboxaldehyde

-
-
139021-69-5
4-(1'-hydroxy-2'-methylprop-2-enyl)cyclohexene

| Conditions | Yield |
|---|---|
| Stage #1: 2-bromoprop-1-ene With lithium In diethyl ether at -30 - 20℃; for 1.75h; Inert atmosphere; Stage #2: 3-Cyclohexene-1-carboxaldehyde In diethyl ether at -30 - 20℃; for 1.75h; Inert atmosphere; Stage #3: With water; ammonium chloride In diethyl ether Cooling with ice; | 96% |

| Conditions | Yield |
|---|---|
| With indium(III) tris[bis(trifluoromethanesulfonyl)amide]; methyldiphenylsilane In 1,4-dioxane at 50℃; for 24h; Inert atmosphere; Schlenk technique; | 96% |

| Conditions | Yield |
|---|---|
| With sodium hydroxide In ethanol at 25℃; for 3h; | 96% |
-
-
122-51-0
orthoformic acid triethyl ester

-
-
100-50-5
3-Cyclohexene-1-carboxaldehyde

-
-
6314-88-1
1,2,5,6-tetrahydrobenzaldehyde diethyl acetal

| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid In ethanol Heating; | 95% |
| With Amberlyst 15 at 4℃; for 1h; | 93% |
| With ethanol; ammonium chloride |
-
-
7677-24-9
trimethylsilyl cyanide

-
-
100-50-5
3-Cyclohexene-1-carboxaldehyde

-
-
85906-00-9
cyclohex-3-enyl(trimethylsilyloxy)acetonitrile

| Conditions | Yield |
|---|---|
| niobium pentafluoride at 20℃; for 0.166667h; | 95% |
| With N-methyl-2-indolinone In dichloromethane at 20℃; for 0.166667h; | 90% |
| With zinc(II) iodide |
-
-
17640-15-2
methyl cyanoformate

-
-
100-50-5
3-Cyclohexene-1-carboxaldehyde

| Conditions | Yield |
|---|---|
| With diisopropylamine In tetrahydrofuran Addition; | 95% |

| Conditions | Yield |
|---|---|
| With (triphenylphosphine)copper(I) hydride hexamer In toluene at 20℃; for 2h; | 95% |

| Conditions | Yield |
|---|---|
| Stage #1: malononitrile; 3-Cyclohexene-1-carboxaldehyde With morpholine In ethanol for 0.25h; Stage #2: recorcinol In ethanol at 20℃; for 72h; | 95% |

-
-
1539-32-8
4-isopropyl-2,6-dimethyl-1,4-dihydro-pyridine-3,5-dicarboxylic acid diethyl ester

-
-
106-99-0
buta-1,3-diene

-
-
100-50-5
3-Cyclohexene-1-carboxaldehyde

| Conditions | Yield |
|---|---|
| With chromium dichloride; (4s,6s)-2,4,5,6-tetra(9H-carbazol-9-yl)isophthalonitrile In tetrahydrofuran at 20℃; for 24h; Irradiation; diastereoselective reaction; | 95% |

-
-
100-50-5
3-Cyclohexene-1-carboxaldehyde

-
-
57606-87-8, 57606-88-9, 84049-09-2, 112710-09-5, 112710-10-8, 4736-13-4
3-cyclohexene-1-carboxaldehyde oxime

| Conditions | Yield |
|---|---|
| With hydroxylamine hydrochloride; sodium acetate In water at 45℃; for 2h; | 94% |
| With hydroxylamine hydrochloride; sodium acetate In methanol Ambient temperature; | 92% |
| With hydroxylamine hydrochloride; sodium carbonate In ethanol; water for 3h; Ambient temperature; | 90% |
3-Cyclohexene-1-carboxaldehyde Chemical Properties
IUPAC Name: Cyclohex-3-ene-1-carbaldehyde
Synonyms of 3-Cyclohexene-1-carboxaldehyde (CAS NO.100-50-5): 1,2,3,6-Tetrahydrobenzaldehyde ; 4-Formyl-1-cyclohexene
CAS NO: 100-50-5
Molecular Formula: C7H10O
Molecular Weight: 110.15
Molecular Structure: 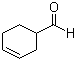
EINECS: 202-858-3
H bond acceptors: 1
H bond donors: 0
Freely Rotating Bonds: 1
Polar Surface Area: 17.07 Å2
Index of Refraction: 1.55
Molar Refractivity: 33.88 cm3
Molar Volume: 106.3 cm3
Surface Tension: 39.4 dyne/cm
Density: 1.035 g/cm3
Flash Point: 57.2 °C
Enthalpy of Vaporization: 34.4 kJ/mol
Boiling Point: 105 °C at 760 mmHg
Vapour Pressure: 30.1 mmHg at 25°C
Stability: Stable. Incompatible with strong oxidizing agents, strong bases, strong reducing agents.
Appearance: 3-Cyclohexene-1-carboxaldehyde (CAS NO.100-50-5) is slightly yellowish-green liquid.
Sensitive: Air Sensitive
3-Cyclohexene-1-carboxaldehyde Toxicity Data With Reference
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| rabbit | LD50 | skin | 1300uL/kg (1.3mL/kg) | LUNGS, THORAX, OR RESPIRATION: OTHER CHANGES BLOOD: HEMORRHAGE SKIN AND APPENDAGES (SKIN): "DERMATITIS, OTHER: AFTER SYSTEMIC EXPOSURE" | National Technical Information Service. Vol. OTS0535072, |
| rat | LC50 | inhalation | 2000ppm/4H (2000ppm) | "Prehled Prumyslove Toxikologie; Organicke Latky," Marhold, J., Prague, Czechoslovakia, Avicenum, 1986Vol. -, Pg. 276, 1986. | |
| rat | LD50 | oral | 2460uL/kg (2.46mL/kg) | American Industrial Hygiene Association Journal. Vol. 23, Pg. 95, 1962. |
3-Cyclohexene-1-carboxaldehyde Consensus Reports
Reported in EPA TSCA Inventory.
3-Cyclohexene-1-carboxaldehyde Safety Profile
Safety Information of 3-Cyclohexene-1-carboxaldehyde (CAS NO.100-50-5):
Hazard Codes:  Xn
Xn
Risk Statements: 21-36/37/38
R21: Harmful in contact with skin.
R36/37/38: Irritating to eyes, respiratory system and skin.
Safety Statements: 26-36-45
S26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice.
S36: Wear suitable protective clothing.
S45: In case of accident or if you feel unwell, seek medical advice immediately (show the label whenever possible.)
RIDADR: UN 2498 3/PG 3
WGK Germany: 1
RTECS: GW2800000
F: 10
HazardClass: 3
PackingGroup: III
Moderately toxic by ingestion, inhalation, and skin contact. Corrosive. An eye, skin, and mucous membrane irritant. Flammable liquid. When heated to decomposition it emits acrid smoke and irritating fumes. See also ALDEHYDES.
3-Cyclohexene-1-carboxaldehyde Standards and Recommendations
DOT Classification: 3; Label: Flammable Liquid
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View