Synthetic route
-
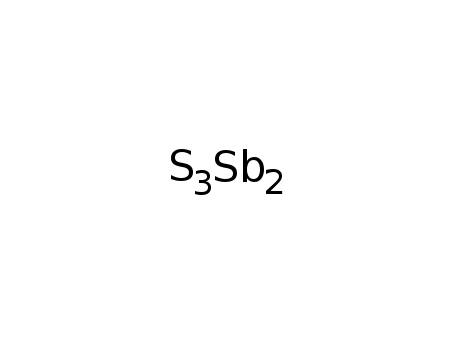
-
antimony(III) sulfide
Conditions
| Conditions | Yield |
|---|
| With hydrogen In neat (no solvent) red heat;; | 100% |
| With H2 In neat (no solvent) red heat;; | 100% |
| With potassium ferrocyanide In melt under KCN;; impured with up to 3% Fe;; | 72% |
-
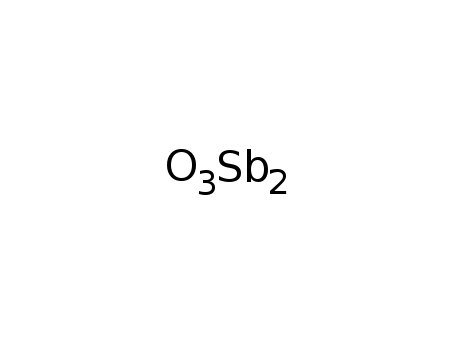
-
antimony(III) trioxide
Conditions
| Conditions | Yield |
|---|
| With urea byproducts: H2O, CO2, N2; react. in a crucible over a low Meker flame, heating gently for 10 min; metal was extd. manually with a spatula; | 98% |
| With P3N5 In neat (no solvent) heating Sb2O3 with P3N5;; | |
| Electrolysis; in melted mixt. of 2B2O3*Na2O and NaF at 800°C; | |
-

-
antimony(III) sulfide
Conditions
| Conditions | Yield |
|---|
| In water in an aq. soln. of potassium sulfide, 200-240°C, 60-85 MPa; | 97% |
Conditions
| Conditions | Yield |
|---|
| With hydrogen In neat (no solvent) in a reversed chlorine detonating gas flame;; | A 93%
B 7% |
Conditions
| Conditions | Yield |
|---|
| With sodium hydroxide; isopropyl alcohol In water other Radiation; NaOH soln. dropping into Sb-salt soln. (hydrolysis prevention, PVA or SDS as surfactant, i-PrOH as scavenger of OH radicals ), bubbling with N2 for 1 h, irradiation in a field of 70000 Ci (60)Co γ-ray source, 8.82E+4 Gy radiation dose, pptn.; ppt. filtration off, washing (water, alcohol), vac. drying at 60°C; optical microscopy; | 70% |
| With sodium hydroxide; isopropyl alcohol In water other Radiation; NaOH soln. dropping into Sb-salt soln. (hydrolysis prevention, PVA or SDS as surfactant, i-PrOH as scavenger of OH radicals ), bubbling with N2 for 1 h, irradiation in a field of 70000 Ci (60)Co γ-ray source, 6.24E+4 Gy radiation dose, pptn.; ppt. filtration off, washing (water, alcohol), vac. drying at 60°C; optical microscopy; | 62% |
| With sodium hydroxide; isopropyl alcohol In water other Radiation; NaOH soln. dropping into Sb-salt soln. (hydrolysis prevention, PVA or SDS as surfactant, i-PrOH as scavenger of OH radicals ), bubbling with N2 for 1 h, irradiation in a field of 70000 Ci (60)Co γ-ray source, 5.14E+4 Gy radiation dose, pptn.; ppt. filtration off, washing (water, alcohol), vac. drying at 60°C; optical microscopy; | 45% |
-

-
antimony(III) trioxide
-
B

-
cobalt antimonide
Conditions
| Conditions | Yield |
|---|
| With Na2CO3 In melt Electrolysis; (N2); at 650-700°C; voltages 3-5 V; current 1 A; graphite electrodes; 95 mol.% metaantimonate in the melt; | A 70%
B 29% |
-
B
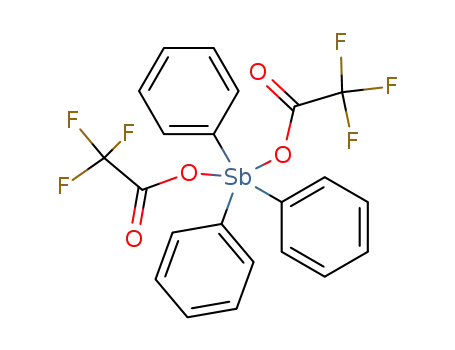
-
36971-66-1
triphenylantimony bis(trifluoroacetate)
Conditions
| Conditions | Yield |
|---|
| In benzene reflux for 2 h; | A n/a
B 62% |
| In benzene reflux for 2 h; | A n/a
B 62% |
-

-
antimony(III) trioxide
-

-
antimony(III) sulfide
Conditions
| Conditions | Yield |
|---|
| by heating in inert gas flow; | A n/a
B 50% |
| by heating in inert gas flow; | A n/a
B 50% |
-
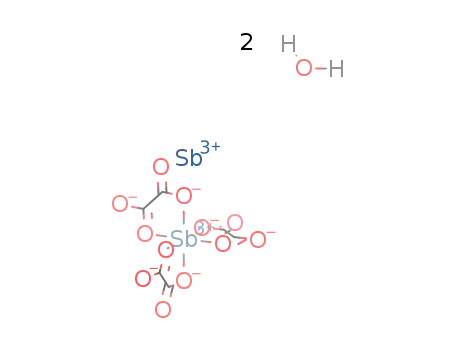
-
antimony(III)tris(oxalato)antimonate(III) dihydrate
-
A

-
antimony(III) trioxide
-
C

-
antimony suboxide
Conditions
| Conditions | Yield |
|---|
| In neat (no solvent) Kinetics; byproducts: H2O, CO, CO2; sample heating in DSC apparatus in dynamic N2 at 10 K/min up to 670°C; DSC, XRD; | A 1%
B n/a
C n/a |
-

-
(99)Ge0965(00)Sb0035
Conditions
| Conditions | Yield |
|---|
| In neat (no solvent) Kinetics; decompn. on heating to 670-770K;; X-ray microspectral and microstructural investigation; solid mixture obtained;; | |
| In melt Kinetics; decompn. on heating to 860-870K;; X-ray microspectral and microstructural investigation; liquid mixture obtained;; | |
-
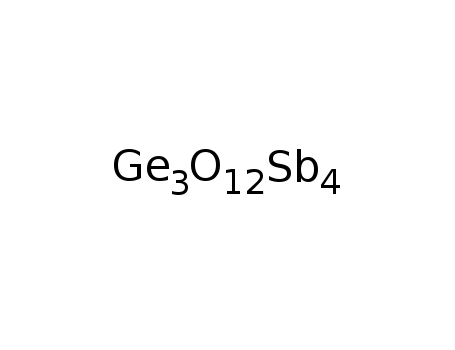
-
Sb4Ge3O12
-
B

-
germanium dioxide
Conditions
| Conditions | Yield |
|---|
| With isopropyl alcohol In isopropyl alcohol High Pressure; interaction in autoclave with supercritical isopropanol; identified by X-ray diffraction; | |
-

-
cobalt antimonide
Conditions
| Conditions | Yield |
|---|
| In neat (no solvent) below 800 °C very slow reactn., above 1000 °C very lively reactn.;; | |
| In neat (no solvent) below 800 °C very slow reactn., above 1000 °C very lively reactn.;; | |
-
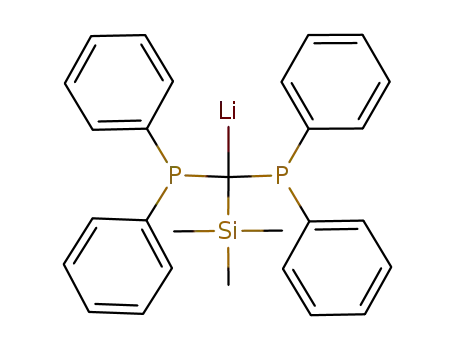
-
LiC(P(C6H5)2)2(Si(CH3)3)
Conditions
| Conditions | Yield |
|---|
| In tetrahydrofuran byproducts: Ph2P(Cl)=C(PPh2)(SiMe3); N2 atm.; cooling (-78°C to room temp.), stirring (12 h); | |
-
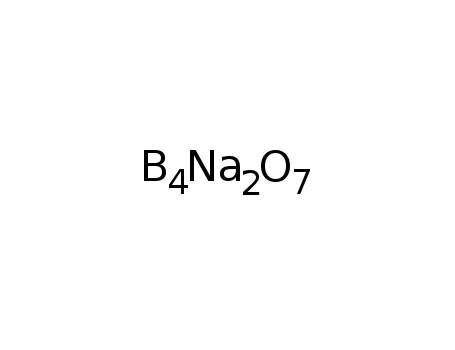
-
Sodium borate
-

-
antimony(III) sulfide
-
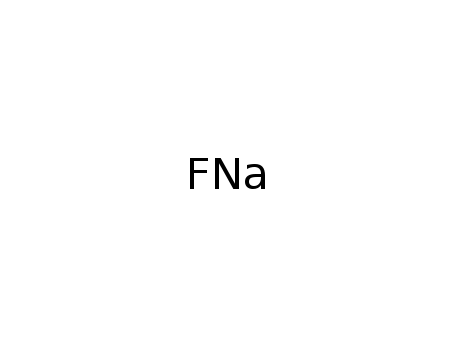
-
sodium fluoride
Conditions
| Conditions | Yield |
|---|
| In melt Electrolysis; 800 °C;; contains only traces of S;; | |
| In melt Electrolysis; 800 °C;; contains only traces of S;; | |
-

-
galium antimonide
-
B
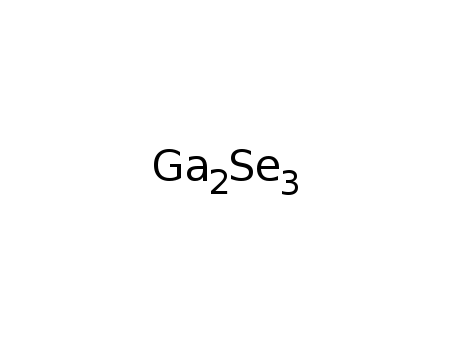
-
gallium selenide
Conditions
| Conditions | Yield |
|---|
| equilibrium; | |
| equilibrium; | |
-

-
galium antimonide
-
B

-
gallium(I) selenide
Conditions
| Conditions | Yield |
|---|
| equilibrium; | |
| equilibrium; | |
-

-
galium antimonide
-
B

-
gallium(II) selenide
Conditions
| Conditions | Yield |
|---|
| equilibrium; | |
| equilibrium; | |
-

-
antimony(III) sulfide
-
-
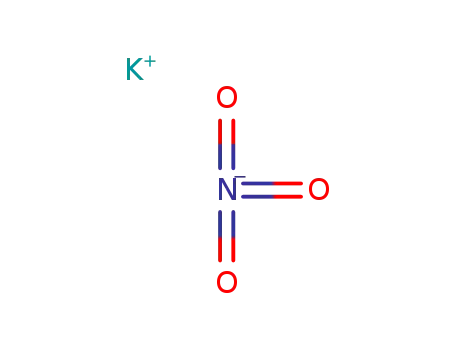
potassium nitrate
-
B
-
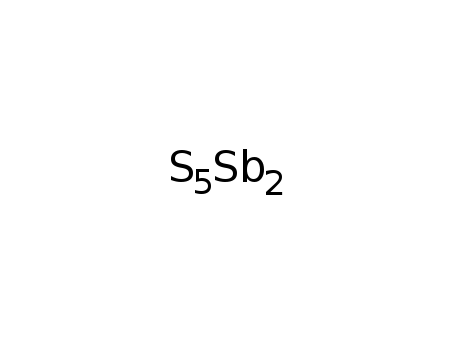
antimony(V) sulfide
-

-
lead(II) sulfide
-

-
antimony(III) sulfide
-
C

-
antimony subsulfide
-

-
lead(II) sulfide
-

-
antimony(III) sulfide
-
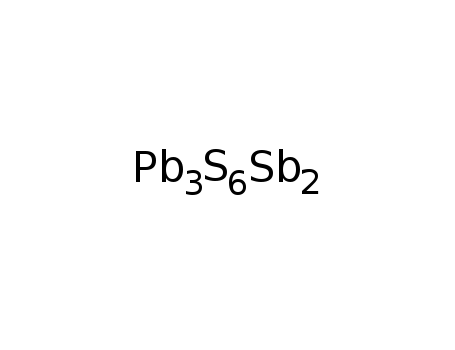
-
falkmanite
Conditions
| Conditions | Yield |
|---|
| With hydrogen sulfide in H2-H2S atm. at 400°C PbS:Sb2S3 mol. ratio 1-6:1; H2S in 22.0%; equilibrium; | |
| With H2S in H2-H2S atm. at 400°C PbS:Sb2S3 mol. ratio 1-6:1; H2S in 22.0%; equilibrium; | |
-

-
lead(II) sulfide
-

-
antimony(III) sulfide
-
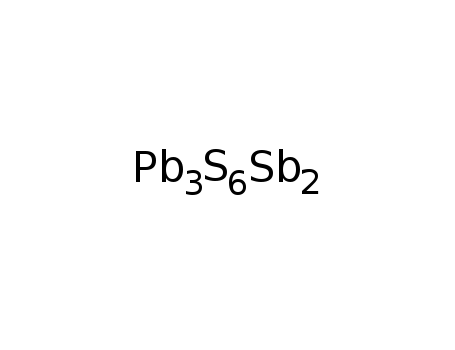
-
falkmanite
-

-
boulangerite
Conditions
| Conditions | Yield |
|---|
| With hydrogen sulfide in H2-H2S atm. at 400°C PbS:Sb2S3 mol. ratio 1-6:1; H2S in 24.7%; equilibrium; | |
| With H2S in H2-H2S atm. at 400°C PbS:Sb2S3 mol. ratio 1-6:1; H2S in 24.7%; equilibrium; | |
-

-
lead(II) sulfide
-

-
antimony(III) sulfide
-
B

-
5PbS*3Sb2S3=Pb5Sb6S14
-
C

-
7PbS*5Sb2S3=Pb7Sb10S22
Conditions
| Conditions | Yield |
|---|
| With hydrogen sulfide in H2-H2S atm. at 400°C PbS:Sb2S3 mol. ratio 1-6:1; H2S in 33.6%; equilibrium; | |
| With H2S in H2-H2S atm. at 400°C PbS:Sb2S3 mol. ratio 1-6:1; H2S in 33.6%; equilibrium; | |
-

-
lead(II) sulfide
-

-
antimony(III) sulfide
-
B

-
5PbS*3Sb2S3=Pb5Sb6S14
-

-
boulangerite
Conditions
| Conditions | Yield |
|---|
| With hydrogen sulfide in H2-H2S atm. at 400°C PbS:Sb2S3 mol. ratio 1-6:1; H2S in 30.1%; equilibrium; | |
| With H2S in H2-H2S atm. at 400°C PbS:Sb2S3 mol. ratio 1-6:1; H2S in 30.1%; equilibrium; | |
-

-
lead(II) sulfide
-

-
antimony(III) sulfide
-
B

-
5PbS*4Sb2S3=Pb5Sb8S17
-
C

-
7PbS*5Sb2S3=Pb7Sb10S22
Conditions
| Conditions | Yield |
|---|
| With hydrogen sulfide in H2-H2S atm. at 400°C PbS:Sb2S3 mol. ratio 1-6:1; H2S in 35.9%; equilibrium; | |
| With H2S in H2-H2S atm. at 400°C PbS:Sb2S3 mol. ratio 1-6:1; H2S in 35.9%; equilibrium; | |
-

-
lead(II) sulfide
-

-
antimony(III) sulfide
-
B

-
5PbS*4Sb2S3=Pb5Sb8S17
-
C

-
Pb(SbS2)2
Conditions
| Conditions | Yield |
|---|
| With hydrogen sulfide in H2-H2S atm. at 400°C PbS:Sb2S3 mol. ratio 1-6:1; H2S in 37.0%; equilibrium; | |
| With H2S in H2-H2S atm. at 400°C PbS:Sb2S3 mol. ratio 1-6:1; H2S in 37.0%; equilibrium; | |
-

-
lead(II) sulfide
-

-
antimony(III) sulfide
-
B

-
Pb(SbS2)2
Conditions
| Conditions | Yield |
|---|
| With hydrogen sulfide in H2-H2S atm. at 400°C PbS:Sb2S3 mol. ratio 1-6:1; H2S in 39.8%; equilibrium; | |
| With H2S in H2-H2S atm. at 400°C PbS:Sb2S3 mol. ratio 1-6:1; H2S in 39.8%; equilibrium; | |
-
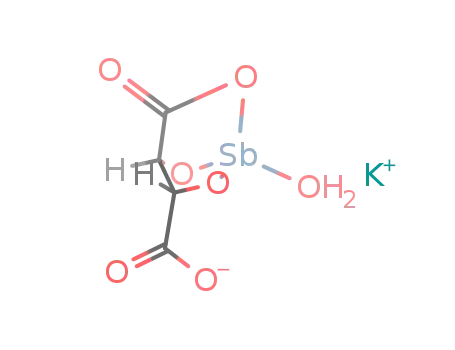
-
K(1+)*{C4H2O6SbH2O}(1-)=K{C4H2O6Sb(H2O)}
Conditions
| Conditions | Yield |
|---|
| In not given Electrolysis; electrodeposition of Sb (ionic strength = 0.54 (KNO3)); | |
-
B

-
potassium chloride
Conditions
| Conditions | Yield |
|---|
| In toluene reactn. in boiling toluene soln.;; | |
| In toluene reactn. in boiling toluene soln.;; | |
-

-
osmium
-

-
OsSb2
Conditions
| Conditions | Yield |
|---|
| at 1150℃; Sealed tube; | 100% |
| melting in an evacuated quartz tube, annealing between 800 and 1000°C; | |
| melting in an evacuated quartz tube, annealing between 800 and 1000°C; | |
| in evacuated quartz tube; | |
| at 999.84℃; Inert atmosphere; | |
-
Ge#dotGa#dotSb
-
Ge#dotGa#dotSb
Conditions
| Conditions | Yield |
|---|
| In neat (no solvent) Ge, Ga and Sb placed in evacuated (1E-2 Pa), sealed quartz ampoules, annealed (800°C, 1400 h); single phase (microstructural analysis), samples prepared with total of 0.25 at.% dopant (Ge = 99.75 at.%) and others with total dopant content of 5 E19 cm-3, also Ge:Sb = 3:1, 1:1 and 1:3; | 100% |
-

-
indium(III) antimonide
Conditions
| Conditions | Yield |
|---|
| In neat (no solvent) In, Sb evacuated, closed in an outgassed quartz ampoule at .apprx.1E-4 Pa, heated at 500.+-.20°C, 70h or heated at 400.+-.20°C, 110h; | 100% |
| In melt crystn. from the melt with nearly stoichiometric composition;; single crystals obtained;; | |
| In neat (no solvent) High Pressure; 0.7 GPa, laser heating; | |
-

-
antimony(III) sulfide
Conditions
| Conditions | Yield |
|---|
| In melt melting of Sb and S at 450-500 °C gives complete reaction; slow cooling;; | 100% |
| heating; | |
| mixt. fusing (evac. quartz ampoule); vac. sublimation; | |
-

-
antimony(III) trioxide
Conditions
| Conditions | Yield |
|---|
| With air at 800°C from melted metal; filtering; | 100% |
| With carbon dioxide byproducts: CO; at 830-1100°C; | |
| Electrolysis; at Sb anode; | |
-

-
europium
-
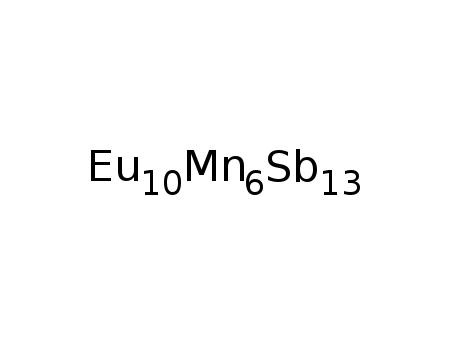
-
Eu10Mn6Sb13
Conditions
| Conditions | Yield |
|---|
| With tin at 999.84℃; for 36h; Glovebox; Inert atmosphere; | 100% |
| In further solvent(s) Sn, Eu, Mn, and Sb layered into an alumina crucible, placed into a fusedsilica tube with a 2-nd crucible filled with SiO2 wood placed on top, s ealed under Ar, heated slowly to 500°C, allowed to dwell for 1 h,heated slowly to 1100°C,; the react. vessel slowly cooled to 600°C, removed, inverted, placed into a centrifuge, spun for 1 min at 6500 rpm; | |
| In further solvent(s) Sn, Eu, Mn, and Sb placed in a graphite tube, vac.-sealed in a fused silica tube, heated to 1000°C slowly, held at 1000°C for 24 h, cooled to 550°C slowly, held at 550°C for 4 d, cooled toroom temp. slowly; | |
-

-
rubidium
-
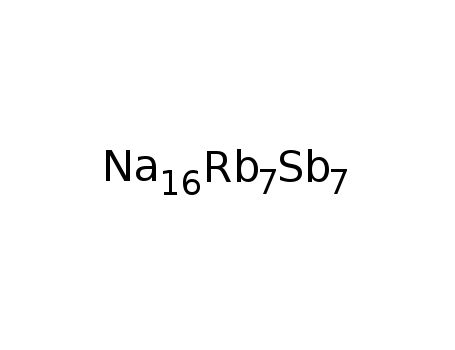
-
Na16(RbRb6)Sb7
Conditions
| Conditions | Yield |
|---|
| In neat (no solvent) byproducts: RbSb; molar ratio Na:Rb:Sb stoichiometric to 2:1:1; heating (12 h to 873 K; 120 h, 873 K), cooling (48 h, room temp.); XRD; | 100% |
-
germanium doped with antimony
-
germanium doped with antimony
Conditions
| Conditions | Yield |
|---|
| In neat (no solvent) Ge and Sb placed in evacuated (1E-2 Pa), sealed quartz ampoules, annealed (800°C, 1400 h); single phase (microstructural analysis), dopant content of 5 E19 cm-3; | 100% |
| In neat (no solvent) evapg. Ge with Sb;; coating;; | |
| In neat (no solvent) Ge single crystals homogenized with Sb at 970 K for 1450 h in sealed quartz ampule evacuated (1.3E-3 Pa); | |
-

-
galium antimonide
Conditions
| Conditions | Yield |
|---|
| In neat (no solvent) Ga, Sb evacuated, closed in an outgassed quartz ampoule at .apprx.1E-4 Pa, heated at 580.+-.20°C, 140h; | 100% |
| melting in a quartz crucible in flowing hydrogen (purified over Pd, flow rate 70 ml/min); for compensation of evapn. of Sb, 0.1% excess Sb was applied;; | |
| In neat (no solvent) Sb/Ga flux ratio was approx. 8.5, GaAs(001) as substrate, mol. beam epitaxy; | |
-

-
ZrNiSb
Conditions
| Conditions | Yield |
|---|
| In neat (no solvent) Electric Arc; heating (650°C, 4 d; dynamic Ar atm., arc melting), annealing (5 h, 1050-1200°C); | 100% |
| In neat (no solvent) vac. (5E-3 mbar); stoichiometric ratio, heating (1 week, 1100°C); | |
| In melt Electric Arc; arc melted under Ar gettered with Ti; 5 wt.-% of Sb required to compensate evaporative losses during arc-melting; ingots sealed in evacuated fused-silica tubes and annealed at 870 K for 720 h; quenched in cold water; XRD; EDX; | |
-
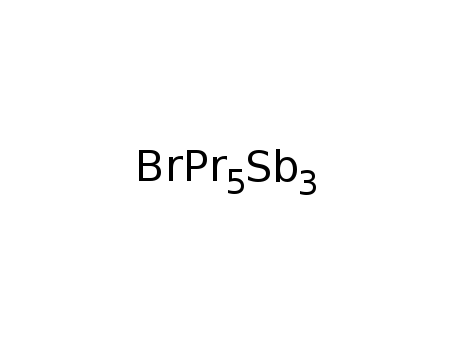
-
Pr5Sb3Br
Conditions
| Conditions | Yield |
|---|
| In neat (no solvent) (N2), heating (950°C, 2 weeks); | 100% |
-

-
hafnium
-

-
HfNiSb
Conditions
| Conditions | Yield |
|---|
| In neat (no solvent) Electric Arc; heating (650°C, 4 d; dynamic Ar atm., arc melting); | 100% |
| In neat (no solvent) Electric Arc; repeated remelting, annealing in evac. quartz tube for 250 h at 800°C, quenching (cold water); | |
-

-
tellurium
-

-
Ge0.4Sn0.4Sb0.13Te
Conditions
| Conditions | Yield |
|---|
| at 590 - 900℃; for 120h; Inert atmosphere; | 100% |
-

-
tellurium
-
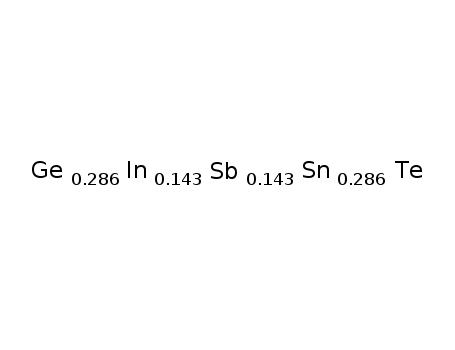
-
Ge0286Sn0286In0143Sb0143Te
Conditions
| Conditions | Yield |
|---|
| at 590 - 900℃; for 120h; Inert atmosphere; | 100% |
-

-
tellurium
-

-
Ge0.72Sn0.08In0067Sb0067Te
Conditions
| Conditions | Yield |
|---|
| at 590 - 900℃; for 120h; Inert atmosphere; | 100% |
-

-
tellurium
-
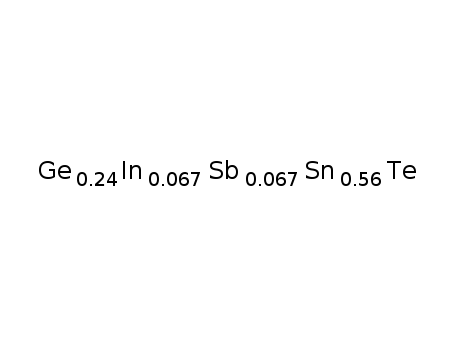
-
Ge0.24Sn0.56In0067Sb0067Te
Conditions
| Conditions | Yield |
|---|
| at 590 - 900℃; for 120h; Inert atmosphere; | 100% |
-

-
tellurium
-

-
Ge0.08Sn0.72In0067Sb0067Te
Conditions
| Conditions | Yield |
|---|
| at 590 - 900℃; for 120h; Inert atmosphere; | 100% |
-

-
tellurium
-
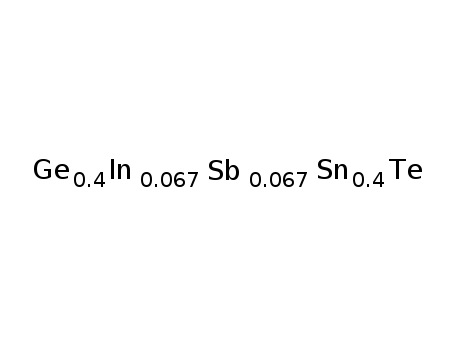
-
Ge0.4Sn0.4In0067Sb0067Te
Conditions
| Conditions | Yield |
|---|
| at 590 - 900℃; for 120h; Inert atmosphere; | 100% |
-

-
tellurium
-
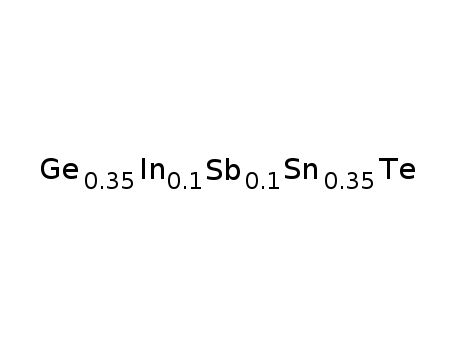
-
Ge0.35Sn0.35In0.1Sb0.1Te
Conditions
| Conditions | Yield |
|---|
| at 590 - 900℃; for 120h; Inert atmosphere; | 100% |
-

-
tellurium
-
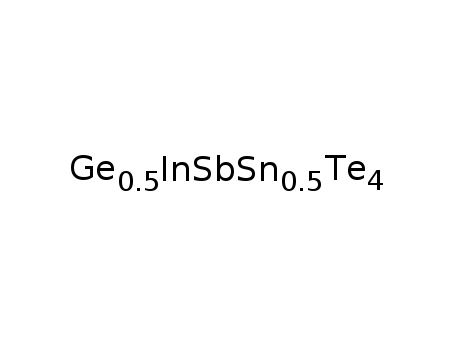
-
Ge0.5Sn0.5InSbTe4
Conditions
| Conditions | Yield |
|---|
| at 550 - 950℃; for 240h; Inert atmosphere; | 100% |
-

-
tellurium
-

-
GeSnInSbTe5
Conditions
| Conditions | Yield |
|---|
| at 350 - 950℃; for 240h; Inert atmosphere; | 100% |
-

-
tellurium
-
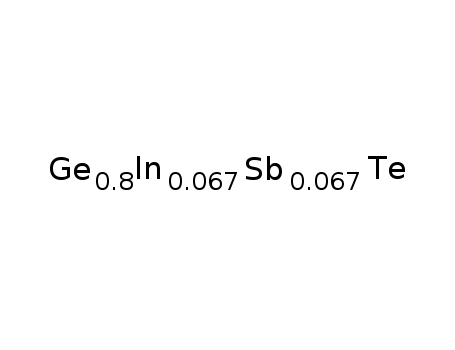
-
Ge0.8In0067Sb0067Te
Conditions
| Conditions | Yield |
|---|
| at 590 - 900℃; for 120h; Inert atmosphere; | 100% |
-

-
tellurium
-
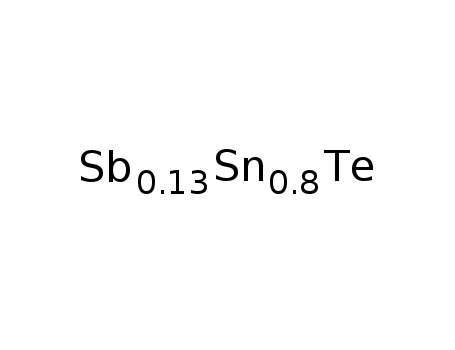
-
Sn0.8Sb0.13Te
Conditions
| Conditions | Yield |
|---|
| at 590 - 900℃; for 120h; Inert atmosphere; | 100% |
-

-
tellurium
-
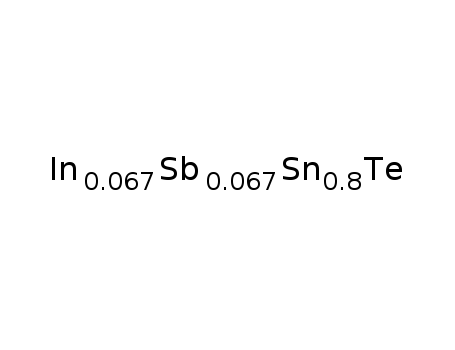
-
Sn0.8In0067Sb0067Te
Conditions
| Conditions | Yield |
|---|
| at 590 - 900℃; for 120h; Inert atmosphere; | 100% |
-
-
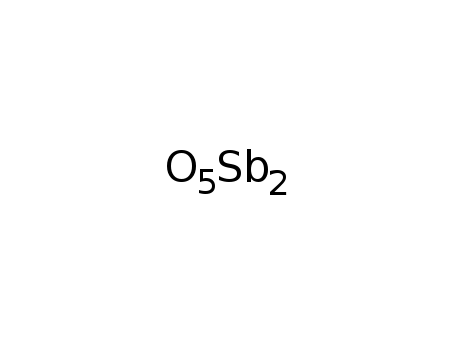
antimony pentoxide
Conditions
| Conditions | Yield |
|---|
| In water byproducts: NO2; Sb powder was covered with concd. nitric acid, heated under an open hood and over a bunsen burner flame, solid was filtered, washed with water,left to dry, heated in open beaker over a bunsen burner flame; | 99.729% |
-
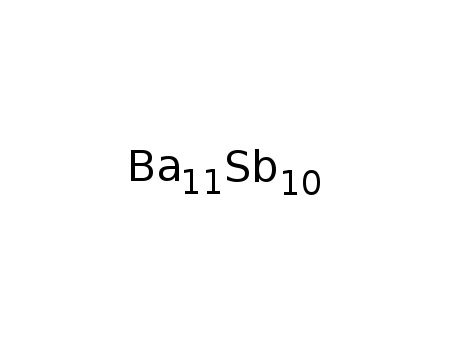
-
Ba11Sb10
Conditions
| Conditions | Yield |
|---|
| In melt Electric Arc; 1:1-mixt. of Sr and Sb; | 99% |
| In melt under Ar; mixt. of Ba (12.43 mmol) and Sb (8.276) heated to 850°Cwith 200°C/h, cooled to room temp. with 100°C/h; | |
| In melt 5:4-mixt. of Ba and Sb heated in alumina or Ta-crucible to 1100°C, kept at this temp. for ca. 1 h, cooled with 50-100°C/h; | |
-

-
mercury
-
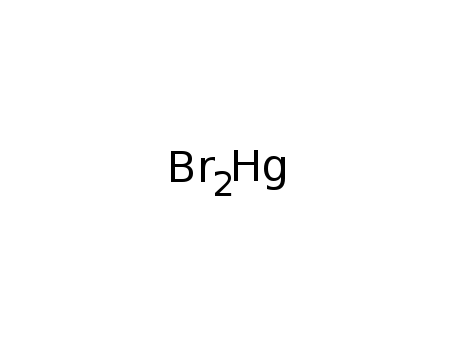
-
mercury dibromide
-
-
Hg6Sb4(4+)*BiBr6(3-)*Br(1-)=Hg6Sb4BiBr7
Conditions
| Conditions | Yield |
|---|
| In neat (no solvent) mixt. of HgBr2, Hg, Sb, Bi (molar ratio 7/5/8/2) was heated in sealed evacuated glass ampoule at temp. 270-290°C, after seven days ppt. was deposited on colder part of ampoule; XRD; | 99% |
-
-
ytterbium
-

-
Yb8Ge3Sb5
Conditions
| Conditions | Yield |
|---|
| In neat (no solvent, solid phase) Electric Arc; mixt. of 1 mmol of Yb, 0.38 mmol of Ge and 0.63 mmol of Sb cold pressed into pellet, arc melting, annealed at 750°C for 1 week; elem. anal.; | 99% |
| In neat (no solvent, solid phase) Yb, Ge and Sb combined, pressed into pellet, loaded in carbon-coated quartz tube, heated to 850°C in 10 h, held at 850°C for 72 h,cooled to 50°C in 12 h; | |
-

-
thallium
-

-
mercury
-
-
mercury dibromide
-
-
Hg3Sb2TlBr3
Conditions
| Conditions | Yield |
|---|
| In neat (no solvent, solid phase) Hg, HgBr2, Sb and Tl in molar ratio 3:3:4:2 sealed in glass ampoule, heated at 300°C for 48 h; cooled to 260°C; | 99% |
Antimony Chemical Properties
Molecular Structure:
.png)
Molecular Formula: Sb
Molecular Weight: 121.76
IUPAC Name: Antimony
Synonyms of Antimony (CAS NO.7440-36-0): Antimony Black ; Antimony, metallic ; Antimony, regulus ; Antymon ; Antymon [Polish] ; C.I. 77050 ; CI 77050 ; EINECS 231-146-5 ; HSDB 508 ; Regulus of antimony ; Stibium ; Stibium metallicum ; UNII-9IT35J3UV3 ; Antimony and compounds ; Antimony powder ; Antimony powder [UN2871] [Poison] ; Antimony, elemental ; UN2871
CAS NO: 7440-36-0
Classification Code: Tumor data
Melting point: 630 °C
Boiling Point: 1950 °C
Density: 6.69 g/mL at 25 °C
Flash Point: 1380°C
Solubility H2O: soluble
Form: powder
Water Solubility: INSOLUBLE
Merck: 13,698
Antimony Uses
Antimony (CAS NO.7440-36-0) can be used in the manufacture Ⅲ ~ V family compound semiconductor, high purity alloys, antimony sulfide and used for cooling components and silicon, germanium crystals doped with agents.
Additionally,it is used as analytical reagent, but also for the preparation of alloys and salt ,but also for high-purity reagents and raw materials for the semiconductor
Antimony Production
Industrial production of Antimony (CAS NO.7440-36-0) has distillation and zone melting method.
Distillation of antimony block by adding the reactor, which leads to chlorine chlorination reaction of antimony trichloride. Crude antimony trichloride will join the distiller, by adding a small amount of scrap metal antimony by vacuum distillation to remove its impurities antimony pentachloride, and then distilled under atmospheric pressure. To take 221 ~ 225 ℃ of the fractions by cooling crystallization obtained antimony trichloride, after reduction, the obtained purity metal antimony ≥ 99.999%. And then this metal antimony as raw material, purified by double distillation, the purity of obtained high-purity antimony ≥ 99.9999%.
Antimony Toxicity Data With Reference
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
| guinea pig | LD50 | intraperitoneal | 150mg/kg (150mg/kg) | | Environmental Quality and Safety, Supplement. Vol. 1, Pg. 1, 1975. |
| mouse | LD50 | intraperitoneal | 90mg/kg (90mg/kg) | | "Toxicometric Parameters of Industrial Toxic Chemicals Under Single Exposure," Izmerov, N.F., et al., Moscow, Centre of International Projects, GKNT, 1982Vol. -, Pg. 22, 1982. |
| rat | LD50 | intraperitoneal | 100mg/kg (100mg/kg) | | "Toxicometric Parameters of Industrial Toxic Chemicals Under Single Exposure," Izmerov, N.F., et al., Moscow, Centre of International Projects, GKNT, 1982Vol. -, Pg. 22, 1982. |
| rat | LD50 | oral | 7gm/kg (7000mg/kg) | | Environmental Quality and Safety, Supplement. Vol. 1, Pg. 1, 1975. |
Antimony Consensus Reports
Antimony (CAS NO.7440-36-0) and its compounds are on the Community Right-To-Know List. Reported in EPA TSCA Inventory.
Antimony Safety Profile
Hazard Codes of Antimony (CAS NO.7440-36-0):  N,
N, Xn,Xi
Xn,Xi
Risk Statements: 34-51/53-20/22-36/37/38
R34: Causes burns.
R51/53: Toxic to aquatic organisms, may cause long-term adverse effects in the aquatic environment.
R20/22: Harmful by inhalation and if swallowed.
R36/37/38: Irritating to eyes, respiratory system and skin.
Safety Statements: 60-61-36/37/39-26
S60: This material and its container must be disposed of as hazardous waste.
S61: Avoid release to the environment. Refer to special instructions / safety data sheets.
S36/37/39: Wear suitable protective clothing, gloves and eye/face protection.
S26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice.
RIDADR: UN 3264 8/PG 2
WGK Germany: 2
RTECS: CC4025000
HazardClass: 6.1
PackingGroup: III
An experimental poison by intraperitoneal route. Questionable carcinogen with experimental carcinogenic data. Moderate fire and explosion hazard in the forms of dust and vapor when exposed to heat or flame. See also POWDERED METALS. When heated or on contact with acid it emits toxic fumes of SbH3. Electrolysis of acid sulfides and stirred Sb halide yields explosive Sb. It can react violently with NH4NO3, halogens, BrN3, BrF3, HClO3, ClO, ClF3, HNO3, KNO3, KMnO4, K2O2, NaNO3, oxidants.
Antimony Standards and Recommendations
OSHA PEL: TWA 0.5 mg(Sb)/m3
ACGIH TLV: TWA 0.5 mg(Sb)/m3
DFG MAK: 0.5 mg(Sb)/m3
NIOSH REL: TWA 0.5 mg(Sb)/m3
DOT Classification: 6.1; Label: KEEP AWAY FROM FOOD
Antimony Analytical Methods
For occupational chemical analysis use OSHA: #ID-125 G or NIOSH: Elements in Blood or Tissue, 8005.
Antimony Specification
Storage Features of Antimony (CAS NO.7440-36-0).
Treasury ventilated low-temperature drying; with acids, food additives and kept separate
 N,
N, Xn
Xn








































































































![]()
 N,
N, Xn,Xi
Xn,Xi