-
Name
Carbohydrazide
- EINECS 207-837-2
- CAS No. 497-18-7
- Article Data42
- CAS DataBase
- Density 1.341 g/cm3
- Solubility soluble in water, insoluble in alcohol, ether, chloroform, benzene
- Melting Point 150-153 °C(lit.)
- Formula CH6N4O
- Boiling Point 167.26°C (rough estimate)
- Molecular Weight 90.0848
- Flash Point
- Transport Information UN 3077
- Appearance white powder
- Safety 61-36/37/39
- Risk Codes 5-51/53-36/37/38-20/21/22-43
-
Molecular Structure
-
Hazard Symbols
 Xn,
Xn, Xi,
Xi, N
N
- Synonyms 1, 3-Diaminourea;Carbonic acid, dihydrazide;Carbazic acid, hydrazide;Carbonothioic dichloride;Urea, N,N-diamino-;Carbonohydrazide;1,3-Diaminomocovina;Hydrazinecarboxamide, N-amino-;1,3-Diaminourea;Semicarbazide, 4-amino-;Urea, 1,3-diamino-;OCarbohydrazide;Hydrazine, carbonylbis-;
- PSA 93.17000
- LogP 0.21540
Synthetic route

| Conditions | Yield |
|---|---|
| With hydrazine hydrate In methanol; water at 75℃; for 0.000833333h; Solvent; | 99% |
| With hydrazine hydrate In methanol; water at 70 - 75℃; for 3h; | 98.5% |
| With hydrazine hydrate; acetic acid In ethanol for 3h; Reflux; | 89.1% |

| Conditions | Yield |
|---|---|
| With hydrazine hydrate In ethanol; water at 75 - 80℃; for 2h; | 98.8% |
| With hydrazine hydrate Substitution; Heating; | 80% |
| With hydrazine hydrate destilliert langsam den abgespaltenen Alkohol und anschliessend Wasser und nicht umgesetztes Ausgangsmaterial ab; |
-
-
2980-33-8
1H-3,5-dinitropyridine-2-one

-
A
-
2075-46-9
4-nitro-1H-pyrazole

-
B
-
497-18-7
carbonodihydrazide

| Conditions | Yield |
|---|---|
| With hydrazine hydrate at 90 - 95℃; for 7h; | A 87% B n/a |

| Conditions | Yield |
|---|---|
| With hydrazine hydrate at 90 - 95℃; for 7h; | A 74% B 75% |
| With hydrazine hydrate at 90 - 95℃; for 7h; Product distribution; also 3,5-dinitropyridone investigated; | A 74% B 75% |
-
-
616-38-6
carbonic acid dimethyl ester

-
A
-
6294-89-9
hydrazinecarboxylic acid methyl ester

-
B
-
497-18-7
carbonodihydrazide

| Conditions | Yield |
|---|---|
| With hydrazine hydrate In neat (no solvent) at 80℃; Temperature; | A 53% B 35% |

-
A
-
78831-00-2
6-phenyl-4,5-dihydro-1,2,4-triazin-3(2H)-one

-
B
-
497-18-7
carbonodihydrazide

| Conditions | Yield |
|---|---|
| With hydrazine hydrate In water; acetic acid for 6h; hydrazinolysis; Heating; | A 45% B n/a |

| Conditions | Yield |
|---|---|
| With water; hydrazine man saugt das auskrystallisierende Hydrazinphenolat ab und dampft die waessr. Loesung im Vakuum ein; |
-
-
10195-79-6
hydrazonium hydrazincarbonate

-
-
497-18-7
carbonodihydrazide

| Conditions | Yield |
|---|---|
| at 140℃; im geschlossenen Rohr; | |
| at 140℃; | |
| heating at 140°C in a bomb tube; |

| Conditions | Yield |
|---|---|
| With hydrazine hydrate |
-
-
7647-01-0
hydrogenchloride

-
-
27048-31-3
benzaldehyde (5-phenyl-[1,3,4]oxadiazol-2-yl)-hydrazone

-
-
497-18-7
carbonodihydrazide
-
-
7647-01-0
hydrogenchloride

-
-
1003-23-2
4-amino-1,2,4-triazol-5-one

-
A
-
64-18-6
formic acid

-
B
-
497-18-7
carbonodihydrazide


-
-
497-18-7
carbonodihydrazide

| Conditions | Yield |
|---|---|
| at 140℃; |

-
-
497-18-7
carbonodihydrazide

| Conditions | Yield |
|---|---|
| With oxalic acid at 180℃; Herstellung; |

-
-
497-18-7
carbonodihydrazide

| Conditions | Yield |
|---|---|
| With sebacic acid anhydride at 230℃; |
-
-
4468-90-0
1,5-dicarbamoyl carbonohydrazide

-
-
7732-18-5
water

-
A
-
21531-96-4
4-aminourazole

-
B
-
497-18-7
carbonodihydrazide

| Conditions | Yield |
|---|---|
| at 150℃; |


| Conditions | Yield |
|---|---|
| With diluted acid Nebenprod.2:Hydrazin; |
-
-
7647-01-0
hydrogenchloride

-
-
3658-33-1
benzoyl carbohydrazide

-
A
-
497-18-7
carbonodihydrazide

-
B
-
65-85-0
benzoic acid

-
-
7647-01-0
hydrogenchloride

-
-
861536-01-8
1-(4-amino-benzoyl)-carbonohydrazide

-
A
-
497-18-7
carbonodihydrazide

-
B
-
150-13-0
4-amino-benzoic acid

-
-
102598-99-2
N,N',N''-tris-benzylidenamino-guanidine

-
A
-
497-18-7
carbonodihydrazide

-
B
-
100-52-7
benzaldehyde

-
C
-
302-01-2
hydrazine


| Conditions | Yield |
|---|---|
| With hydrazine hydrate at 70℃; for 4h; |
-
-
75-44-5
phosgene

-
-
302-01-2
hydrazine

-
B
-
471-31-8
carbazic acid

-
C
-
497-18-7
carbonodihydrazide

-
D
-
1617-13-6
1,2-bis(hydrazinocarbonyl)hydrazine

| Conditions | Yield |
|---|---|
| about 5°C, vacuum apparate; | A n/a B <1 C n/a D n/a |


| Conditions | Yield |
|---|---|
| In methanol; water at 60 - 65℃; for 9h; Solvent; Temperature; | 99.4% |
| With potassium bromide In water at 20℃; for 6h; Inert atmosphere; | 10 %Chromat. |

| Conditions | Yield |
|---|---|
| at 20℃; for 0.0666667h; | 99% |

-
-
497-18-7
carbonodihydrazide

| Conditions | Yield |
|---|---|
| In water addn. of ligand soln. to CuSO4 soln. (molar ratio = 1:1; stirring, pptn.); filtration, washing (EtOH), drying; elem. anal.; | 99% |
-
-
497-18-7
carbonodihydrazide

-
-
1135076-68-4, 1254123-97-1
1,1'-ethylenebis(5-nitroimino-4,5-dihydrotetrazole)

-
-
1158828-14-8
carbohydrazinium ethylene bis(5-nitroiminotetrazolate)

| Conditions | Yield |
|---|---|
| In water at 20℃; | 99% |

| Conditions | Yield |
|---|---|
| In ethanol at 23℃; | 99% |
-
-
497-18-7
carbonodihydrazide

-
-
90-02-8
salicylaldehyde

-
-
6638-49-9
bis[[2-hydroxyphenyl]methylene]carbonic dihydrazide

| Conditions | Yield |
|---|---|
| In ethanol Reflux; | 98% |
| In ethanol Reflux; | 98% |
| In ethanol; water for 24h; Reflux; | 95% |

| Conditions | Yield |
|---|---|
| In benzene addn. of carbohydrazide to soln. of SbCl3 in dry benzene (molar ratio 1:1), mixt. stirred (40°C, 20 h), cream coloured ppt.; filtered, washed (repeatedly with warm anhydrous alcohol), dried under vac. at 40°C; elem. anal.; | 98% |

-
-
497-18-7
carbonodihydrazide

| Conditions | Yield |
|---|---|
| In water addn. of excess NaClO4 to soln. of Cu(ClO4)2 and ligand (molar ratio Cu:ligand=1:4, stirring); cooling (0-5°C, pptn.), filtration, washing (EtOH), drying; elem. anal.; | 98% |

-
-
497-18-7
carbonodihydrazide

-
-
1258291-10-9
1-methoxy-5-nitroiminotetrazole

| Conditions | Yield |
|---|---|
| In water at 20℃; for 0.166667h; Thermodynamic data; | 98% |

| Conditions | Yield |
|---|---|
| In methanol at 50℃; for 1h; | 98% |

| Conditions | Yield |
|---|---|
| With acetic acid In ethanol for 5h; Reflux; | 98% |
| With acetic acid In ethanol for 2h; Reflux; | 88% |

| Conditions | Yield |
|---|---|
| With formic acid for 2h; Reflux; | 98% |


| Conditions | Yield |
|---|---|
| at 20℃; for 0.05h; | 97% |
| With water |

| Conditions | Yield |
|---|---|
| at 20℃; for 0.0666667h; | 97% |

| Conditions | Yield |
|---|---|
| at 20℃; for 0.0666667h; | 97% |

| Conditions | Yield |
|---|---|
| at 20℃; for 0.05h; | 97% |

| Conditions | Yield |
|---|---|
| In benzene addn. of carbohydrazide to soln. of SbCl3 in dry benzene (molar ratio 2:1), mixt. stirred (40°C, 20 h), cream coloured ppt.; filtered, washed (repeatedly with warm anhydrous alcohol), dried under vac. at 40°C; elem. anal.; | 97% |
-
-
497-18-7
carbonodihydrazide

-
-
94838-88-7
2-formyl-4,5-(methylenedioxy)benzeneboronic acid

| Conditions | Yield |
|---|---|
| With formic acid In water for 2h; Reflux; | 97% |

| Conditions | Yield |
|---|---|
| at 20℃; for 0.0666667h; | 96% |
-
-
497-18-7
carbonodihydrazide

-
-
89-95-2
2-methyl-benzyl alcohol

-
-
6638-49-9
bis[[2-hydroxyphenyl]methylene]carbonic dihydrazide

| Conditions | Yield |
|---|---|
| at 20℃; for 0.0333333h; | 96% |

| Conditions | Yield |
|---|---|
| at 20℃; for 0.0833333h; | 96% |

| Conditions | Yield |
|---|---|
| at 20℃; for 0.0666667h; | 96% |
-
-
497-18-7
carbonodihydrazide

-
-
1609957-32-5
3,3’-dinitramino-4,4’-bifurazane

| Conditions | Yield |
|---|---|
| In methanol Heating; | 96% |
-
-
497-18-7
carbonodihydrazide

| Conditions | Yield |
|---|---|
| Stage #1: dipotassium bis(1-oxidotetrazolyl)furoxane With hydrogenchloride Stage #2: carbonodihydrazide In water Heating; | 96% |

| Conditions | Yield |
|---|---|
| Stage #1: carbonodihydrazide With hydrogenchloride In water at 20℃; for 0.166667h; Stage #2: 2-methylpropenal In water | 96% |
-
-
497-18-7
carbonodihydrazide

-
-
123-73-9
crotonaldehyde

-
-
6341-81-7, 26233-36-3
bis(2-butenylideneamino)urea

| Conditions | Yield |
|---|---|
| Stage #1: carbonodihydrazide With hydrogenchloride In water at 20℃; for 0.166667h; Stage #2: crotonaldehyde In water at 20℃; for 1h; | 96% |

| Conditions | Yield |
|---|---|
| In ethanol at 60℃; for 2h; | 95% |

| Conditions | Yield |
|---|---|
| at 20℃; for 0.0833333h; | 95% |
Carbohydrazide Consensus Reports
Reported in EPA TSCA Inventory.
Carbohydrazide Standards and Recommendations
DOT Classification: Forbidden
Carbohydrazide Specification
The Carbohydrazide, with the CAS registry number 497-18-7, is also known as 1,3-Diaminourea. It belongs to the product categories of Pharmaceutical Intermediates; Aromatic Hydrazides, Hydrazines, Hydrazones and Oximes; API intermediates; Water Ttreatment Chemicals; Carbonyl Compounds; Hydrazides; Organic Building Blocks. Its EINECS number is 207-837-2. This chemical's molecular formula is CH6N4O and molecular weight is 90.08. What's more, its systematic name is Carbonohydrazide. This chemical should be sealed and stored in a ventilated and dry place. Moreover, it should be protected from oxides. It is used as an oxygen scavenger in water treatment for boilers. It is an alternative to the hazardous and potentially carcinogenic hydrazine. Carbohydrazide reacts with oxygen to make water, nitrogen and urea. It is widely used in the production of drugs, herbicides, plant growth regulators, and dyestuffs.
Physical properties of Carbohydrazide are: (1)ACD/LogP: -1.657; (2)# of Rule of 5 Violations: 1; (3)ACD/LogD (pH 5.5): -1.69; (4)ACD/LogD (pH 7.4): -1.66; (5)ACD/BCF (pH 5.5): 1.00; (6)ACD/BCF (pH 7.4): 1.00; (7)ACD/KOC (pH 5.5): 2.79; (8)ACD/KOC (pH 7.4): 2.99; (9)#H bond acceptors: 5; (10)#H bond donors: 6; (11)#Freely Rotating Bonds: 2; (12)Polar Surface Area: 93.17 Å2; (13)Index of Refraction: 1.537; (14)Molar Refractivity: 20.972 cm3; (15)Molar Volume: 67.149 cm3; (16)Polarizability: 8.314×10-24cm3; (17)Surface Tension: 65.4 dyne/cm; (18)Density: 1.341 g/cm3.
Preparation: this chemical can be prepared by carbonic acid diethyl ester by heating. This reaction will need reagent N2H4·H2O. The yield is about 80%.
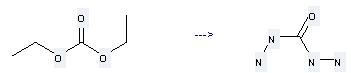
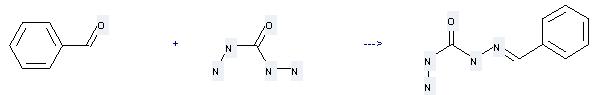
Uses of Carbohydrazide: it can be used to produce 1-benzylidene-carbonohydrazide at the temperature of 20 °C. It will need solvent methanol with the reaction time of 24 hours. The yield is about 54%.
When you are using this chemical, please be cautious about it as the following:
This chemical is harmful by inhalation, in contact with skin and if swallowed. It is irritating to eyes, respiratory system and skin. It may cause sensitisation by skin contact. This substance is toxic to aquatic organisms as it may cause long-term adverse effects in the aquatic environment. When heating, it may cause an explosion. When using it, you need to wear suitable protective clothing, gloves and eye/face protection. You should avoid releasing it to the environment, and you need to refer to special instructions/safety data sheet.
You can still convert the following datas into molecular structure:
(1)SMILES: O=C(NN)NN
(2)Std. InChI: InChI=1S/CH6N4O/c2-4-1(6)5-3/h2-3H2,(H2,4,5,6)
(3)Std. InChIKey: XEVRDFDBXJMZFG-UHFFFAOYSA-N
The toxicity data is as follows:
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| mouse | LD50 | intraperitoneal | 167mg/kg (167mg/kg) | Journal of Medicinal and Pharmaceutical Chemistry. Vol. 4, Pg. 259, 1961. | |
| mouse | LD50 | intravenous | 120mg/kg (120mg/kg) | BEHAVIORAL: CONVULSIONS OR EFFECT ON SEIZURE THRESHOLD | Journal of Pharmacology and Experimental Therapeutics. Vol. 122, Pg. 110, 1958. |
| mouse | LD50 | subcutaneous | 131mg/kg (131mg/kg) | Acta Biologica et Medica Germanica. Vol. 21, Pg. 635, 1968. |
Related Products
- Carbohydrazide
- 49719-60-0
- 497-19-8
- 49721-45-1
- 49722-76-1
- 49722-90-9
- 497-23-4
- 497-25-6
- 497259-23-1
- 497262-02-9
- 497-30-3
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View