-
Name
Ethyl cyanoacetate
- EINECS 203-309-0
- CAS No. 105-56-6
- Article Data109
- CAS DataBase
- Density 1.047 g/cm3
- Solubility 20 g/L (20 °C) in water
- Melting Point -22 °C
- Formula C5H7NO2
- Boiling Point 203.6 °C at 760 mmHg
- Molecular Weight 113.116
- Flash Point 84.1 °C
- Transport Information UN 3276
- Appearance Clear to very yellow liquid
- Safety 36/37/39-26
- Risk Codes 20/21/22-36/38
-
Molecular Structure
-
Hazard Symbols
 Xn,
Xn, Xi
Xi
- Synonyms Ethyl cyanoacetate [UN2666] [Keep away from food];Ethylester kyseliny kyanoctove [Czech];Cyanoacetic ester;Cyanoacetic acid, ethyl ester;Stearate Methyl;Cyanoacetic acid ethyl ester;Cyanacetate ethyle [German];Usaf kf-25;Malonic acid ethyl ester nitrile;Malonic acid, ethyl ester nitrile;Ethyl cyanacetate;Estere cianoacetico [Italian];ethyl 2-cyanoacetate;Ethyl cyanoethanoate;Acetic acid, cyano-, ethyl ester;Cyanacetate ethyle;
- PSA 50.09000
- LogP 0.46318
Synthetic route

| Conditions | Yield |
|---|---|
| With N-hydroxyphthalimide; cobalt(II) acetate In acetonitrile at 190℃; under 6750.68 Torr; for 7h; |

-
-
105-56-6
ethyl 2-cyanoacetate

| Conditions | Yield |
|---|---|
| With hydroxylamine hydrochloride In ethanol at 90℃; for 4h; pH=2; pH-value; Reagent/catalyst; Solvent; Temperature; Inert atmosphere; | 81% |

| Conditions | Yield |
|---|---|
| With sulfuric acid In dichloromethane for 18h; Heating; | 95% |
| With sulfuric acid In dichloromethane for 18h; Reflux; Dean-Stark; | 95% |
| With sulfuric acid; sulfur trioxide at 20 - 70℃; for 4h; Temperature; Reflux; | 93.6% |

| Conditions | Yield |
|---|---|
| With triethylamine at 15℃; for 4.83333h; Concentration; Reagent/catalyst; Solvent; Temperature; Time; | 94.2% |
-
-
773837-37-9
sodium cyanide

-
-
105-39-5
chloroacetic acid ethyl ester

-
A
-
10359-15-6
diethyl 2-cyanosuccinate

-
B
-
20822-61-1
triethyl 2-cyano-1,2,3-propanetricarboxylate

-
C
-
105-56-6
ethyl 2-cyanoacetate

| Conditions | Yield |
|---|---|
| With tetrabutylammomium bromide In dichloromethane; water at 20℃; for 4h; | A 10.4% B 5.4% C 44% |


| Conditions | Yield |
|---|---|
| With sulfuric acid for 3h; Time; Reflux; | 97% |
| With sulfuric acid for 3h; Reflux; | 97% |
-
-
773837-37-9
sodium cyanide

-
-
74-90-8
hydrogen cyanide

-
-
105-39-5
chloroacetic acid ethyl ester

-
-
105-56-6
ethyl 2-cyanoacetate

| Conditions | Yield |
|---|---|
| With tetrabutylammomium bromide In water; acetonitrile at 55℃; for 2h; Product distribution / selectivity; | 93.2% |

-
-
64-17-5
ethanol

-
-
372-09-8
cyanoacetic acid

-
A
-
105-56-6
ethyl 2-cyanoacetate

-
B
-
105-53-3
diethyl malonate

| Conditions | Yield |
|---|---|
| With sulfuric acid In hexane; ethyl acetate at 30 - 62℃; for 5h; Temperature; Solvent; Dean-Stark; | A 105.2 g B 1.8 g |

-
-
21860-86-6
aspartic acid 4-ethyl ester

-
-
105-56-6
ethyl 2-cyanoacetate

| Conditions | Yield |
|---|---|
| With trichloroisocyanuric acid; sodium hydroxide In water at 20℃; for 6h; Temperature; | 85% |

| Conditions | Yield |
|---|---|
| With sodium ethanolate; sodium amide; Diethyl carbonate at 140 - 160℃; | |
| With sodium ethanolate; sodium amide; Diethyl carbonate at 140 - 160℃; | |
| With sodium ethanolate; sodium amide; Diethyl carbonate at 140 - 160℃; |

| Conditions | Yield |
|---|---|
| With sodium hydroxide; tetrabutylammomium bromide In water; chlorobenzene |

| Conditions | Yield |
|---|---|
| With caesium carbonate In acetonitrile for 1.5h; Heating; | 98% |
| With cesium fluoride In N,N-dimethyl-formamide at 10 - 15℃; for 3h; | 79 % Chromat. |

| Conditions | Yield |
|---|---|
| With boron trifluoride diethyl etherate for 24h; Heating; | A 90% B 4% |
| With sulfuric acid at 130 - 135℃; |

-
-
78514-93-9
C11H16N4O2S

-
A
-
99496-77-2
1-isopropenyl-3-methylthio-1H-1,2,4-triazole

-
B
-
105-56-6
ethyl 2-cyanoacetate

| Conditions | Yield |
|---|---|
| In acetic acid at 70℃; for 2h; | A 66% B n/a |
-
-
205651-05-4
ethyl pentacyanocyclopropanecarboxylate

-
-
105-56-6
ethyl 2-cyanoacetate

| Conditions | Yield |
|---|---|
| With N,N-Dimethylaniline hydroiodide In ethyl acetate at 50℃; for 0.0833333h; Decomposition; |
-
-
151-50-8
potassium cyanide

-
-
105-39-5
chloroacetic acid ethyl ester

-
A
-
105-56-6
ethyl 2-cyanoacetate

-
B
-
105-34-0
methyl 2-cyanoacetate

| Conditions | Yield |
|---|---|
| With methanol |


| Conditions | Yield |
|---|---|
| With diethyl ether; sodium amide |
-
-
122604-89-1
benzaldehyde 2-cyano-2-ethoxycarbonylvinylamino(dimethylamino)methylenehydrazone

-
A
-
2025-40-3
ethyl benzylidenecyanoacetate

-
B
-
122604-96-0
α,α-bis(3-dimethylamino-1,2,4-triazol-1-yl)toluene

-
C
-
105-56-6
ethyl 2-cyanoacetate

| Conditions | Yield |
|---|---|
| With acetic acid at 80℃; for 0.0833333h; Yield given; | A n/a B 30% C n/a |
| With acetic acid at 80℃; for 0.0833333h; Yields of byproduct given; | A n/a B 30% C n/a |

-
-
68897-45-0
N,N,N',N',N'',N''-Hexamethylguanidiniumcyanid

-
-
105-36-2
ethyl bromoacetate

-
A
-
6926-43-8
1,1,2,2,3,3-Hexamethylguanidiniumbromid

-
B
-
105-56-6
ethyl 2-cyanoacetate

| Conditions | Yield |
|---|---|
| In dichloromethane at 20℃; for 2h; | A n/a B 53% |


| Conditions | Yield |
|---|---|
| With iron(III) chloride for 1h; Heating; | 76% |
-
-
623-47-2
propynoic acid ethyl ester

-
A
-
116270-19-0
ethyl (Z)-3-azido-2-propenoate

-
B
-
116270-20-3
E-ethyl 3-azidoacrylate

-
C
-
105-56-6
ethyl 2-cyanoacetate

-
D
-
116270-21-4
ethyl (Z)-3-(4-ethoxycarbonyl-1H-1,2,3-triazol-1-yl)propenoate

| Conditions | Yield |
|---|---|
| With borax; potassium dihydrogenphosphate; sodium azide In water at 50℃; for 0.5h; ultrasound irradiation; Yield given. Yields of byproduct given; |

-
-
7357-70-2
Cyanothioacetamide

-
-
431-35-6
3-bromo-1,1,1-trifluoroacetone

-
-
64-17-5
ethanol

-
A
-
329978-21-4
2-[4-(trifluoromethyl)thiazol-2-yl]acetonitrile

-
B
-
105-56-6
ethyl 2-cyanoacetate

| Conditions | Yield |
|---|---|
| for 18h; Heating / reflux; |

-
-
138614-14-9
mer-hydrido[[(ethoxycarbonyl)methyl]cyano](ethyl cyanoacetate)tris(triphenylphosphine)ruthenium(II)

-
-
1663-45-2
1,2-bis-(diphenylphosphino)ethane

-
B
-
105-56-6
ethyl 2-cyanoacetate

| Conditions | Yield |
|---|---|
| In benzene-d6 room temp., 3 h; NMR monitoring; | A 82% B 60% |

-
-
1722-12-9
2-chloropyrimidine

-
-
54535-14-7
ethyl 5-oxo-2,5-dihydroisoxazole-4-carboxylate

-
A
-
100422-78-4
ethyl 5-oxo-2-(pyrimidin-2-yl)-2,5-dihydroisoxazole-4-carboxylate

-
B
-
105-56-6
ethyl 2-cyanoacetate

| Conditions | Yield |
|---|---|
| at 130℃; for 0.25h; Yields of byproduct given; | A 62% B n/a |

-
-
174902-47-7
Ethyl 2-acetyl-5-oxo-2,5-dihydroisoxazole-4-carboxylate

-
A
-
76284-27-0
ethyl 2-methyloxazole-4-carboxylate

-
B
-
105-56-6
ethyl 2-cyanoacetate

| Conditions | Yield |
|---|---|
| at 540℃; under 0.05 Torr; for 1h; | A 10% B 90% |

| Conditions | Yield |
|---|---|
| With palladium 10% on activated carbon In methanol retro-Michael reaction; |
-
-
135885-92-6
3-(4-Bromo-benzoyl)-4-(4-bromo-phenyl)-1-cyano-4-hydroxy-2,6-diphenyl-cyclohexanecarboxylic acid ethyl ester

-
A
-
72758-69-1, 2403-27-2, 22966-23-0
(E)-1-(4-bromophenyl)-3-phenylprop-2-en-1-one

-
B
-
105-56-6
ethyl 2-cyanoacetate

| Conditions | Yield |
|---|---|
| With sodium ethanolate In ethanol Heating; | A 86% B n/a |
| With sodium ethanolate In ethanol at 50℃; Kinetics; Further Variations:; Temperatures; |
-
-
83624-73-1
3-Benzoyl-2,6-bis-(4-chloro-phenyl)-1-cyano-4-hydroxy-4-phenyl-cyclohexanecarboxylic acid ethyl ester

-
A
-
24721-26-4, 956-04-7, 22252-16-0
4-chlorochalcone

-
B
-
105-56-6
ethyl 2-cyanoacetate

| Conditions | Yield |
|---|---|
| With sodium ethanolate In ethanol Heating; | A 87% B n/a |
| With sodium ethanolate In ethanol at 50℃; Kinetics; Further Variations:; Temperatures; |
-
-
135885-91-5
3-Benzoyl-2,6-bis-(4-bromo-phenyl)-1-cyano-4-hydroxy-4-phenyl-cyclohexanecarboxylic acid ethyl ester

-
A
-
22966-09-2
(2E)-3-(4-bromophenyl)-1-phenylprop-2-en-1-one

-
B
-
105-56-6
ethyl 2-cyanoacetate

| Conditions | Yield |
|---|---|
| With sodium ethanolate In ethanol Heating; | A 85% B n/a |
| With sodium ethanolate In ethanol at 50℃; Kinetics; Further Variations:; Temperatures; |

| Conditions | Yield |
|---|---|
| In neat (no solvent) at 20℃; Inert atmosphere; | 100% |
| With 1,8-diazabicyclo[5.4.0]undec-7-ene In 2-methyltetrahydrofuran at 40℃; for 4h; | 87% |
| at 130℃; for 4h; | 75% |
-
-
98-01-1
furfural

-
-
105-56-6
ethyl 2-cyanoacetate

-
-
67449-75-6, 23973-22-0
ethyl (E)-2-cyano-3-(2-furyl)-2-propenoate

| Conditions | Yield |
|---|---|
| With L-proline for 0.0333333h; Knoevenagel condensation; microwave irradiation; | 100% |
| ytterbium(III) perfluorooctanesulfonate In toluene at 80℃; for 3h; Knoevenagel condensation; | 99% |
| With polyacrylonitrile fiber modified with triethylenetetramine In water at 50℃; for 1.5h; Knoevenagel condensation; | 99% |
-
-
3612-20-2
1-phenylmethyl-4-piperidone

-
-
105-56-6
ethyl 2-cyanoacetate

-
-
1463-52-1
ethyl 2-(1-benzylpiperidin-4-ylidene)-2-cyanoacetate

| Conditions | Yield |
|---|---|
| With acetic acid In toluene at 20℃; for 4h; Heating / reflux; | 100% |
| With ammonium acetate; acetic acid; benzene Abtrennen des entstehenden H2O; | |
| With triethylamine In dichloromethane at 20℃; |
-
-
108-94-1
cyclohexanone

-
-
105-56-6
ethyl 2-cyanoacetate

-
-
6802-76-2
ethyl 2-cyano-2-cyclohexylideneacetate

| Conditions | Yield |
|---|---|
| With 1,4-diaza-bicyclo[2.2.2]octane In neat liquid at 20℃; for 0.133333h; Knoevenagel Condensation; Green chemistry; | 100% |
| With third generation polystyrene supported poly(amidoamine) dendrimer In ethanol at 50℃; for 0.333333h; Knoevenagel condensation; | 98% |
| With ammonium acetate; acetic acid In toluene Knoevenagel Condensation; Reflux; | 97% |
-
-
89-98-5
2-chloro-benzaldehyde

-
-
105-56-6
ethyl 2-cyanoacetate

-
-
24393-43-9
(E)-ethyl 3-(2-chlorophenyl)-2-cyanoacrylate

| Conditions | Yield |
|---|---|
| With IRA-96 anion-exchange resin In ethanol at 25 - 30℃; for 1h; Reagent/catalyst; Knoevenagel Condensation; Sonication; Green chemistry; stereoselective reaction; | 100% |
| With polyacrylonitrile fiber functionalized with N,N-dimethyl-1,3-propanediamine In ethanol for 1.5h; Knoevenagel condensation; Reflux; | 99% |
| With polystyrene-supported DABCO In methanol at 45℃; for 1.5h; Knoevenagel Condensation; Green chemistry; stereoselective reaction; | 99% |
-
-
99-61-6
3-nitro-benzaldehyde

-
-
105-56-6
ethyl 2-cyanoacetate

-
-
18925-00-3, 14394-75-3
(E)-2-cyano-3-(3-nitrophenyl)-2-propenoic acid ethyl ester

| Conditions | Yield |
|---|---|
| With L-proline for 0.1h; Knoevenagel condensation; microwave irradiation; | 100% |
| With third generation polystyrene supported poly(amidoamine) dendrimer In ethanol at 50℃; for 0.333333h; Knoevenagel condensation; | 100% |
| With polyacrylonitrile fiber functionalized with N,N-dimethyl-1,3-propanediamine In ethanol for 1.5h; Knoevenagel condensation; Reflux; | 99% |
-
-
100-52-7
benzaldehyde

-
-
105-56-6
ethyl 2-cyanoacetate

-
-
2169-69-9
ethyl (E)-2-cyano-3-phenyl-2-propenoate

| Conditions | Yield |
|---|---|
| With tetramethyl ammoniumhydroxide at 60℃; for 0.5h; Condensation; Knoevenagel condensation; | 100% |
| With L-proline for 0.0833333h; Knoevenagel condensation; microwave irradiation; | 100% |
| ytterbium(III) perfluorooctanesulfonate In toluene at 80℃; for 8h; Knoevenagel condensation; | 100% |

| Conditions | Yield |
|---|---|
| With polymer supported poly(propylene imine)dendrimer In ethanol at 20℃; for 0.0833333h; Knoevenagel Condensation; Green chemistry; | 100% |
| With N,N-dimethyl-cyclohexanamine In water at 20℃; Knoevenagel Condensation; | 100% |
| With mesoporous hybrid catalyst HYB-75P-25B In ethanol at 59.84℃; for 3.5h; Knoevenagel condensation; Inert atmosphere; | 99% |
-
-
123-08-0
4-hydroxy-benzaldehyde

-
-
105-56-6
ethyl 2-cyanoacetate

-
-
6935-44-0, 42205-38-9
ethyl (E)-2-cyano-3-(4-hydroxyphenyl)-2-propenoate

| Conditions | Yield |
|---|---|
| With third generation polystyrene supported poly(amidoamine) dendrimer In ethanol at 50℃; for 0.25h; Knoevenagel condensation; | 100% |
| Ru(+)Cp(NCCHCO2Et)(-)*(PPh3)2 In tetrahydrofuran at 25℃; for 5h; Condensation; Aldol reaction; | 99% |
| With polyacrylonitrile fiber functionalized with N,N-dimethyl-1,3-propanediamine In ethanol for 1.5h; Knoevenagel condensation; Reflux; | 99% |
-
-
123-11-5
4-methoxy-benzaldehyde

-
-
105-56-6
ethyl 2-cyanoacetate

-
-
2017-87-0
ethyl (2E)-2-cyano-3-(4-methoxyphenyl)acrylate

| Conditions | Yield |
|---|---|
| With diazabicyclo[5.4.0]undec-7-ene-water complex at 20℃; for 0.833333h; Knoevenagel condensation; | 100% |
| With ethylenediamine-modified poly(vinyl chloride) at 20℃; for 0.0666667h; Solvent; Knoevenagel Condensation; Green chemistry; | 100% |
| With third generation polystyrene supported poly(amidoamine) dendrimer In ethanol at 50℃; for 0.25h; Knoevenagel condensation; | 99% |
-
-
123-11-5
4-methoxy-benzaldehyde

-
-
105-56-6
ethyl 2-cyanoacetate

-
-
2286-29-5
ethyl 2-cyano-3-(4-methoxyphenyl)prop-2-enoate

| Conditions | Yield |
|---|---|
| With polymer supported poly(propylene imine)dendrimer In ethanol at 20℃; for 0.0833333h; Knoevenagel Condensation; Green chemistry; | 100% |
| In N,N-dimethyl-formamide for 0.166667h; Knoevenagel Condensation; | 99.4% |
| With N-aminoethylpiperazine functionalized C60 fullerene In ethanol at 60℃; for 1h; Knoevenagel Condensation; | 99% |
-
-
121-33-5
vanillin

-
-
105-56-6
ethyl 2-cyanoacetate

-
-
132464-93-8, 13373-29-0
2-cyano-3-(4-hydroxy-3-methoxyphenyl)acrylic acid ethyl ester

| Conditions | Yield |
|---|---|
| With polymer supported poly(propylene imine)dendrimer In ethanol at 20℃; for 0.166667h; Knoevenagel Condensation; Green chemistry; | 100% |
| With ammonium sulphamate for 0.00416667h; Knoevenagel condensation; Microwave irradiation; neat (no solvent); | 96% |
| aluminum oxide In ethanol for 1.66667h; Heating; | 95% |
-
-
120-14-9
3,4-dimethoxy-benzaldehyde

-
-
105-56-6
ethyl 2-cyanoacetate

-
-
2286-55-7
ethyl α-cyano-β-(3,4-dimethoxyphenyl)acrylate

| Conditions | Yield |
|---|---|
| With piperidine; acetic acid In benzene at 120 - 130℃; for 12h; Heating / reflux; | 100% |
| With polymer supported poly(propylene imine)dendrimer In ethanol at 20℃; for 0.166667h; Knoevenagel Condensation; Green chemistry; | 99% |
| With ammonium sulphamate for 0.00416667h; Knoevenagel condensation; Microwave irradiation; neat (no solvent); | 97% |

| Conditions | Yield |
|---|---|
| With triphenylphosphine In acetonitrile at 23℃; for 0.133333h; | 100% |
| With N-ethyl-N,N-diisopropylamine; 2,6-(NMe2)2PhPd(1+)*BF4(1-) In dichloromethane at 20℃; Product distribution; Kinetics; Further Variations:; Catalysts; double Michael reaction; | 100% |
| With [2,6-bis(4',4'-dimethyl-2'-oxazolinyl)phenyl](acetonitrile)nickel(II) tetrafluoroborate; N-ethyl-N,N-diisopropylamine In dichloromethane at 0℃; for 1h; Michael addition; | 100% |
-
-
120-92-3
cyclopentanone

-
-
105-56-6
ethyl 2-cyanoacetate

-
-
5407-83-0
ethyl 2-cyano-2-cyclopentylideneacetate

| Conditions | Yield |
|---|---|
| piperidine at 23℃; under 750.06 Torr; for 2h; Knoevenagel condensation; | 100% |
| With morpholine; bis(acetylacetonate)oxovanadium at 40℃; for 1h; Reagent/catalyst; Temperature; Time; Knoevenagel Condensation; | 99% |
| With ammonium acetate; acetic acid In toluene Knoevenagel Condensation; Reflux; | 93% |
-
-
105-56-6
ethyl 2-cyanoacetate

-
-
135-02-4
ortho-anisaldehyde

-
-
14533-89-2
(E)-2-cyano-3-(2-methoxyphenyl)-2-propenoic acid ethyl ester

| Conditions | Yield |
|---|---|
| With L-proline for 0.0666667h; Knoevenagel condensation; microwave irradiation; | 100% |
| With polyacrylonitrile fiber modified with triethylenetetramine In water at 50℃; for 1.5h; Knoevenagel condensation; | 99% |
| calcined Mg-Al-layered double hydroxide-supported iPr2-amide In N,N-dimethyl-formamide at 20℃; for 0.25h; Knoevenagel condensation; | 98% |
-
-
105-56-6
ethyl 2-cyanoacetate

-
-
100-10-7
4-dimethylamino-benzaldehyde

-
-
1886-52-8, 74897-86-2, 14394-77-5
ethyl (E)-2-cyano-3-[4-(dimethylamino)phenyl]prop-2-enoate

| Conditions | Yield |
|---|---|
| With third generation polystyrene supported poly(amidoamine) dendrimer In ethanol at 50℃; for 0.25h; Knoevenagel condensation; | 100% |
| With diazabicyclo[5.4.0]undec-7-ene-water complex at 20℃; for 1h; Knoevenagel condensation; | 100% |
| With polyacrylonitrile fiber functionalized with N,N-dimethyl-1,3-propanediamine In ethanol for 1.5h; Knoevenagel condensation; Reflux; | 99% |

| Conditions | Yield |
|---|---|
| With ammonia | 100% |
| With ammonia In tetrahydrofuran; water at 20℃; for 2h; | 93% |
| With ammonium hydroxide In tetrahydrofuran at 20℃; for 24h; | 91% |
-
-
104-87-0
4-methyl-benzaldehyde

-
-
105-56-6
ethyl 2-cyanoacetate

-
-
2017-88-1
ethyl (E)-2-cyano-3-p-tolylacrylate

| Conditions | Yield |
|---|---|
| With L-proline for 0.0833333h; Knoevenagel condensation; microwave irradiation; | 100% |
| With 1,4-diaza-bicyclo[2.2.2]octane In neat liquid at 20℃; for 0.0166667h; Knoevenagel Condensation; Green chemistry; | 100% |
| With ASCPEI In ethanol at 43℃; for 3h; Knoevenagel condensation; | 99% |
-
-
121-33-5
vanillin

-
-
105-56-6
ethyl 2-cyanoacetate

-
-
132464-93-8
ethyl (E)-2-cyano-3-(3-methoxy 4-hydroxyphenyl)-2-propenoate

| Conditions | Yield |
|---|---|
| With third generation polystyrene supported poly(amidoamine) dendrimer In ethanol at 50℃; for 0.333333h; Knoevenagel condensation; | 100% |
| With aluminum oxide; potassium oxide at 23℃; for 3h; Knoevenagel condensation; | 97% |
| With dimethyl 3-methyl-9-oxo-7-(3,3,4,4,5,5,6,6,7,7,8,8,9,9,10,10,10-heptadecafluorodecyl)-2,4-di(pyridin-2-yl)-3,7-diazabicyclo-[3.3.1]nonane-1,5-dicarboxylate In water at 40℃; for 2h; Knoevenagel Condensation; | 96% |
-
-
120-14-9
3,4-dimethoxy-benzaldehyde

-
-
105-56-6
ethyl 2-cyanoacetate

-
-
24393-47-3
ethyl (E)-2-cyano-3-(3,4-dimethoxyphenyl)-2-propenoate

| Conditions | Yield |
|---|---|
| With third generation polystyrene supported poly(amidoamine) dendrimer In ethanol at 50℃; for 0.333333h; Knoevenagel condensation; | 100% |
| With ASCPEI In ethanol at 43℃; for 2h; Knoevenagel condensation; | 98% |
| With ammonium acetate for 0.25h; Knoevenagel condensation; microwave irradiation; | 94% |

| Conditions | Yield |
|---|---|
| for 18h; | 100% |
| With sodium hydroxide |
-
-
14371-10-9
(E)-3-phenylpropenal

-
-
105-56-6
ethyl 2-cyanoacetate

-
-
41109-95-9
(2E,4E)-2-cyano-5-phenyl-2,4-pentadienoic acid ethyl ester

| Conditions | Yield |
|---|---|
| With 1,4-diaza-bicyclo[2.2.2]octane In neat liquid at 20℃; for 0.25h; Knoevenagel Condensation; Green chemistry; | 100% |
| With zinc(II) chloride at 100℃; for 1.5h; | 92% |
| With 1-methyl-piperazine at 25 - 30℃; for 0.166667h; Knoevenagel condensation; | 91% |

| Conditions | Yield |
|---|---|
| at 45℃; for 1.5h; | 100% |
| at 38℃; for 2.33333h; | 91% |
| In ethanol at 20℃; | 82% |
-
-
459-57-4
4-fluorobenzaldehyde

-
-
105-56-6
ethyl 2-cyanoacetate

-
-
18861-57-9, 50737-52-5
ethyl (E)-2-cyano-3-(4-fluorophenyl)-2-propenoate

| Conditions | Yield |
|---|---|
| With 1-(3-silica-supported propyl)-3-[(3-{[1-(3-silica-supported propyl)-4,5-dihydro-1H-imidazol-3-ium-3-yl]methyl}-2,4,6-trimethylphenyl)methyl]-4,5-dihydro-1H-imidazol-3-ium chloride at 130℃; for 2h; Knoevenagel condensation; | 100% |
| With polyacrylonitrile fiber functionalized with N,N-dimethyl-1,3-propanediamine In ethanol for 1.5h; Knoevenagel condensation; Reflux; | 99% |
| With N-(propylcarbamoyl)sulfamic acid bonded on silica at 100℃; for 0.25h; | 97% |
-
-
104-88-1
4-chlorobenzaldehyde

-
-
105-56-6
ethyl 2-cyanoacetate

-
-
2169-68-8
ethyl (E)-3-(4-chlorophenyl)-2-cyanoacrylate

| Conditions | Yield |
|---|---|
| With L-proline for 0.0333333h; Knoevenagel condensation; microwave irradiation; | 100% |
| With IRA-96 anion-exchange resin In ethanol at 25 - 30℃; for 1h; Reagent/catalyst; Knoevenagel Condensation; Sonication; Green chemistry; stereoselective reaction; | 100% |
| With ammonium acetate; acetic acid for 0.0111111h; Knoevenagel condensation; Irradiation; | 99% |
-
-
551-93-9
2-aminoacetophenone

-
-
105-56-6
ethyl 2-cyanoacetate

-
-
28448-12-6
1,2-dihydro-2-oxo-4-methylquinoline-3-carbonitrile

| Conditions | Yield |
|---|---|
| With ammonium acetate for 1h; Heating; | 100% |
| cerium(III) chloride for 0.1h; microwave irradiation; | 86% |
| With ammonium acetate at 220℃; for 0.25h; | 78% |
-
-
555-16-8
4-nitrobenzaldehdye

-
-
105-56-6
ethyl 2-cyanoacetate

-
-
2017-89-2
(E)-ethyl 2-cyano-3-(4-nitrophenyl)acrylate

| Conditions | Yield |
|---|---|
| With L-proline for 0.1h; Knoevenagel condensation; microwave irradiation; | 100% |
| With third generation polystyrene supported poly(amidoamine) dendrimer In ethanol at 50℃; for 0.333333h; Knoevenagel condensation; | 100% |
| With 1,4-diaza-bicyclo[2.2.2]octane In neat liquid at 20℃; for 0.0333333h; Knoevenagel Condensation; Green chemistry; | 100% |
-
-
555-16-8
4-nitrobenzaldehdye

-
-
105-56-6
ethyl 2-cyanoacetate

-
-
96238-12-9
ethyl (Z)-2-cyano-3-(4-nitrophenyl)propenoate

| Conditions | Yield |
|---|---|
| hydrotalcite structure integrating fluoride ions In acetonitrile at 60℃; for 1.5h; Conversion of starting material; Knoevenagel Condensation; | 100% |
| With PEG-600 at 20℃; for 0.25h; Knoevenagel condensation; | 95% |
| With lipase lipoprotein from Aspergillus niger In dimethyl sulfoxide at 35℃; for 48h; Knoevenagel Condensation; Green chemistry; Enzymatic reaction; | 94% |
-
-
86-81-7
3,4,5-trimethoxy-benzaldehyde

-
-
105-56-6
ethyl 2-cyanoacetate

-
-
2601-03-8
2-cyano-3-(3,4,5-trimethoxyphenyl)acrylic acid ethyl ester

| Conditions | Yield |
|---|---|
| With ytterbium(III) triflate at 80 - 85℃; Knoevenagel condensation; neat (no solvent); | 100% |
| With recovered palladium catalyst In ethanol at 20℃; for 18h; Knoevenagel Condensation; Green chemistry; | 97% |
| With montmorillonitesilylpropylidenediamine In toluene Ambient temperature; | 96% |
| With piperidine In ethanol at 20℃; | |
| In ethanol at 20℃; Knoevenagel Condensation; Sonication; |
Ethyl cyanoacetate Analytical Methods
Ethyl cyanoacetate (CAS No.105-56-6) is both a nitrile and an ester.
As esters , it can react with acids to liberate heat along with alcohols and acids. Strong oxidizing acids may cause a vigorous reaction that is sufficiently exothermic to ignite the reaction products. Heat is also generated by the interaction of esters with caustic solutions. Flammable hydrogen is generated by mixing esters with alkali metals and hydrides. Nitriles may polymerize in the presence of metals and some metal compounds. They are incompatible with acids; mixing nitriles with strong oxidizing acids can lead to extremely violent reactions.
As nitriles Ethyl cyanoacetate (CAS No.105-56-6) is generally incompatible with other oxidizing agents such as peroxides and epoxides. The combination of bases and nitriles can produce hydrogen cyanide. Nitriles are hydrolyzed in both aqueous acid and base to give carboxylic acids (or salts of carboxylic acids). These reactions generate heat. Peroxides convert nitriles to amides. Nitriles can react vigorously with reducing agents. Acetonitrile and propionitrile are soluble in water, but nitriles higher than propionitrile have low aqueous solubility. They are also insoluble in aqueous acids.
Ethyl cyanoacetate Specification
The CAS registry number of Ethyl cyanoacetate is 105-56-6. Its EINECS registry number is 203-309-0. The IUPAC name is ethyl 2-cyanoacetate. In addition, the molecular formula is C5H7NO2 and the molecular weight is 113.11. What's more, it belongs to the classes of Pharmaceutical Intermediates; EQ - EZ Analytical Standards; Esters Analytical Standards; Nitriles; Volatiles/ Semivolatiles. It can be used as intermediates in the preparation of vitamin B6 and caffeine. Besides, it also can be used as raw materials for organic synthesis.
Physical properties about this chemical are: (1)ACD/LogP: 0.06; (2)ACD/BCF (pH 5.5): 1; (3)ACD/BCF (pH 7.4): 1; (4)ACD/KOC (pH 5.5): 1; (5)ACD/KOC (pH 7.4): 1; (6)#H bond acceptors: 3; (7)#Freely Rotating Bonds: 3; (8)Polar Surface Area: 50.09 Å2; (9)Index of Refraction: 1.412; (10)Molar Refractivity: 26.9 cm3; (11)Molar Volume: 107.9 cm3; (12)Polarizability: 10.66 ×10-24cm3; (13)Surface Tension: 35.8 dyne/cm; (14)Density: 1.047 g/cm3; (15)Flash Point: 84.1 °C; (16)Enthalpy of Vaporization: 43.99 kJ/mol; (17)Boiling Point: 203.6 °C at 760 mmHg; (18)Vapour Pressure: 0.275 mmHg at 25°C .
Preparation of Ethyl cyanoacetate: it can be prepared by ethyl chloroacetate and sodium cyanide. The ethyl chloroacetate can be obtained by chloroacetic acid and ethanol through esterification reaction. The equation is as follows:
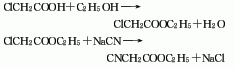
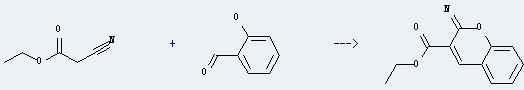
Uses of Ethyl cyanoacetate: it can react with 2-hydroxy-benzaldehyde to get 2-imino-2H-chromene-3-carboxylic acid ethyl ester. This reaction will need catalyst AlPO4-Al2O3. The reaction time is 1 hour with ambient temperature. The yield is about 58%.
When you are using this chemical, please be cautious about it as the following:
This chemical is harmful by inhalation, in contact with skin and if swallowed. It is irritating to eyes, respiratory system and skin. When you are using it, wear suitable protective clothing, gloves and eye/face protection. In case of contact with eyes, rinse immediately with plenty of water and seek medical advice.
You can still convert the following datas into molecular structure:
(1)SMILES: N#CCC(=O)OCC
(2)InChI: InChI=1/C5H7NO2/c1-2-8-5(7)3-4-6/h2-3H2,1H3
(3)InChIKey: ZIUSEGSNTOUIPT-UHFFFAOYAI
The toxicity data is as follows:
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| frog | LDLo | subcutaneous | 4200mg/kg (4200mg/kg) | PERIPHERAL NERVE AND SENSATION: SPASTIC PARALYSIS WITH OR WITHOUT SENSORY CHANGE BEHAVIORAL: TREMOR LUNGS, THORAX, OR RESPIRATION: DYSPNEA | Archives Internationales de Pharmacodynamie et de Therapie. Vol. 5, Pg. 161, 1899. |
| guinea pig | LD50 | subcutaneous | 1115mg/kg (1115mg/kg) | BEHAVIORAL: TREMOR BEHAVIORAL: MUSCLE CONTRACTION OR SPASTICITY) GASTROINTESTINAL: NAUSEA OR VOMITING | Medicina del Lavoro. Industrial Medicine. Vol. 47, Pg. 192, 1956. |
| mouse | LD50 | intraperitoneal | 500mg/kg (500mg/kg) | National Technical Information Service. Vol. AD277-689, | |
| rabbit | LDLo | subcutaneous | 1410mg/kg (1410mg/kg) | PERIPHERAL NERVE AND SENSATION: SPASTIC PARALYSIS WITH OR WITHOUT SENSORY CHANGE LUNGS, THORAX, OR RESPIRATION: RESPIRATORY STIMULATION | Archives Internationales de Pharmacodynamie et de Therapie. Vol. 5, Pg. 161, 1899. |
| rat | LDLo | oral | 400mg/kg (400mg/kg) | "Prehled Prumyslove Toxikologie; Organicke Latky," Marhold, J., Prague, Czechoslovakia, Avicenum, 1986Vol. -, Pg. 921, 1986. |
Related Products
- Ethyl (13-cis)-9-(4-methoxy-2,3,6-trimethylphenyl)-3,7-dimethyl-2,4,6,8-nonatetraenoate
- ethyl (1R,2R)-1-phenyl-2-(trideuteriomethylamino)cyclohex-3-ene-1-carboxylate,hydrochloride
- Ethyl (1S,2R)-2-(dimethylamino)-1-phenylcyclohex-3-ene-1-carboxylate hydrochloride
- Ethyl (2,4,6-trimethylbenzoyl) phenylphosphinate
- Ethyl (2-amino-4-hydroxy-6-methyl-5-pyrimidinyl)acetate
- Ethyl (2-bromopropionamido)acetate
- Ethyl (2-cyanoimino-5,6-dichloro-1,2,3,4-tetrahydroquinazolin-3-yl)acetate
- ETHYL (2E,4Z)-DECADIENOATE
- Ethyl (2-hydroxyethyl)dimethyl-ammonium benzilate chloride
- Ethyl (2-mercaptoethyl) carbamate S-ester with O,O-dimethyl phosphorodithioate
- 10557-21-8
- 10557-44-5
- 105-57-7
- 10557-74-1
- 10557-85-4
- 105580-47-0
- 10558-25-5
- 10558-44-8
- 105-58-8
- 105590-97-4
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View