-
Name
Sulfamic acid
- EINECS 226-218-8
- CAS No. 5329-14-6
- Article Data143
- CAS DataBase
- Density 1.913 g/cm3
- Solubility 146.8 g/L (20 °C) in water
- Melting Point 215-225 °C (dec.)(lit.)
- Formula H3NO3S
- Boiling Point 247oC
- Molecular Weight 97.0947
- Flash Point 205oC
- Transport Information UN 2967
- Appearance white crystalline solid
- Safety 26-28-61-28A
- Risk Codes 36/38-52/53
-
Molecular Structure
-
Hazard Symbols
 Xi
Xi
- Synonyms Stannous sulfamate;Scale Cleen;Amido Sulfonic Acid;Amidoschwefelsaeure;Aminosulfonic acid;Sulfamidsaeure;Amidosulfonic acid;Sulfamidic acid;Imidosulfonic acid;Aminosulfonic Acid (Sulfamic acid);
- PSA 88.77000
- LogP 0.52900
Synthetic route

| Conditions | Yield |
|---|---|
| In sulfuric acid byproducts: CO2; urea dild. in 100% H2SO4 (stirring/cooling); addn. of small portions of fuming H2SO4 (temp. shouldn't exceed 45°C/stirring/cooling); heating carefully to 80°C; evolution of CO2; addn. of next portion fuming H2SO4 and repeating;; left for some time; filtration, washing (concd. H2SO4); drying on clay; left overnight in air; recrystn. from water;; | 94% |
| In sulfuric acid heating with excess fuming H2SO4 (80-90°C, depending on SO3-content); vigorous reaction, evolution of heat;; |

| Conditions | Yield |
|---|---|
| With air In melt two salts was reacted in melt in air; | A n/a B n/a C 92% |


| Conditions | Yield |
|---|---|
| In water aq. acetoxim soln., containing liquid SO2, (25°C) left for 5 h in autoclave;; | 90% |
| In water aq. acetoxim soln. cooled to -80°C; excess of SO2 condensed; sealing of tube; warming up to 0°C (pressure increases); left for 5.5 h at that temp.;; opened flask releases excess of SO2; filtration; dried by washing (alc., ether);; | 77% |
| In ethanol; water acetoxim dild. in water/ethanol is satd. with SO2 gas (0°C); leftfor three days (0°C); (pptd. solid HSO3NH2 will be removed);; filtration, addn. of SO2; washing (alc., ether); dried in air;; | 76% |
| In water aq. medium; dependence of pressure (1.14-3.52 kg/cm*cm more than atm.) and temp. (5-30°C); formation of liquid SO2 should be avoided; pptn.;; | |
| In water aq. medium; dependence of pressure (1.14-3.52 kg/cm*cm more than atm.) and temp. (5-30°C); formation of liquid SO2 should be avoided; pptn.;; |

| Conditions | Yield |
|---|---|
| In water byproducts: pyridine; diln. of C5H5NSO3 in ammonia water (10% NH3) under sepn. of pyridine;; extraction of pyridine (ether); evapn. (water bath); addn. of concd. H2SO4 (cooling); filtration; drying on clay; recrystn from water;; | 80% |

| Conditions | Yield |
|---|---|
| In water byproducts: trimethylamine; diln. in ammonia water by short heating under sepn. of (CH3)N;; evapn.; removal of exceeding NH3 and (CH3)3N; addn. of concd. H2SO4; sepn. of HSO3NH2;; | 80% |

| Conditions | Yield |
|---|---|
| With sodium carbonate In water byproducts: sodium sulfate; aq. soln. of NaNO2/Na2CO3; introduction of SO2 (until acidic/litmus); addn. of a drop concd. H2SO4; formation of N(SO3)(3-); hydrolysis; short heating or left for several hours (stream of air); neutralization (Na2CO3); evapn.; left (12h/0°C);; filtration from sodium sulfate; addn. of concd. H2SO4; immediate pptn. of most HSO3NH2 (left for another day/cooling); dried on clay; washed (ice cold water); recrystn. (hot water);; | 75% |
| With Na2CO3 In water byproducts: sodium sulfate; aq. soln. of NaNO2/Na2CO3; introduction of SO2 (until acidic/litmus); addn. of a drop concd. H2SO4; formation of N(SO3)(3-); hydrolysis; short heating or left for several hours (stream of air); neutralization (Na2CO3); evapn.; left (12h/0°C);; filtration from sodium sulfate; addn. of concd. H2SO4; immediate pptn. of most HSO3NH2 (left for another day/cooling); dried on clay; washed (ice cold water); recrystn. (hot water);; | 75% |

| Conditions | Yield |
|---|---|
| In neat (no solvent) byproducts: CO2; vigorous reaction (careful heating/cooling/stirring); temp. dependence on mechanism discussed;; | 73.4% |
| In neat (no solvent) byproducts: CO2; vigorous reaction (careful heating/cooling/stirring); temp. dependence on mechanism discussed;; | 73.4% |
| In not given equimolar amounts;; |


| Conditions | Yield |
|---|---|
| In water aq. hydroxylammonium sulfate soln. cooled to -30°C; excess of SO2 condensed into vessel and left for 24 h in autoclave (at common temps.);; removal of SO2 (passing air through soln.); crystallization, filtration;washing (alc., ether); drying (air); further pptn. of HSO3NH2 by addn. of concd. H2SO4 to mother lye (at 0°C);; | 72% |
| In water aq. medium; dependence of pressure (1.14-3.52 kg/cm*cm more than atm.) and temp. (5-30°C); formation of liquid SO2 should be avoided; pptn.;; | |
| In water aq. medium; dependence of pressure (1.14-3.52 kg/cm*cm more than atm.) and temp. (5-30°C); formation of liquid SO2 should be avoided; pptn.;; |

| Conditions | Yield |
|---|---|
| In water K2S2O7 (contaminated with 40% KHSO4) dild. in ammonia water (25%/shakingfor 0.5 h/cooling with cold water); until evolution of heat stops; heating (3 min/water bath);; left cooling; filtration, washing (ammonia water); evapn.; addn. of concd. H2SO4; left for several hours (common temps.); filtration; drying on clay; recrystn. from water;; | 60% |

| Conditions | Yield |
|---|---|
| In water byproducts: dimethylaniline; diln. in ice cold ammonia water (10% NH3/) left warming up to common temps. under sepn. of (CH3)2NC6H5;; evapn. (removal of exceeding NH3 and (CH3)2NC6H5/water bath); addn. of concd. H2SO4; sepn. of HSO3NH2;; | 58% |

| Conditions | Yield |
|---|---|
| In sulfuric acid heating a soln. of 1.1g pyridinium salt of carbamidosulfonic acid in 2.5 ml 100% H2SO4 at 110 °C for 0.5 h;; | 50% |
-
-
13954-94-4
trisulfimide

-
A
-
7664-93-9
sulfuric acid

-
B
-
5329-14-6
aminosulfonic acid

-
C
-
7803-58-9
SULFAMIDE

| Conditions | Yield |
|---|---|
| With water in the cold, 48 h, 2 M HCl; | A 33% B n/a C n/a |
| With H2O in the cold, 48 h, 2 M HCl; | A 33% B n/a C n/a |
| In water decompn. in aq. soln.; 0 and 22°C discussed; presece of acid discussed;; |


| Conditions | Yield |
|---|---|
| In water introduction of SO2 into aq. hydroxylammonium salt soln.;; | A 10% B n/a |
| In water introduction of SO2 into aq. hydroxylammonium salt soln.;; | A 10% B n/a |


| Conditions | Yield |
|---|---|
| at 25℃; unter Druck; |


| Conditions | Yield |
|---|---|
| at 0℃; unter Druck; |

-
-
70475-81-9
(N-acetyl-acetimidoyl)-amidosulfuric acid

-
-
7732-18-5
water

-
A
-
625-77-4
N-acetylacetamide

-
B
-
5329-14-6
aminosulfonic acid



| Conditions | Yield |
|---|---|
| In sulfuric acid react. of SO3-vapor with urea, sulfatolysis;; | |
| In sulfuric acid aq. H2SO4; react. of SO3-vapor with urea, sulfatolysis;; |

| Conditions | Yield |
|---|---|
| With sulfuric acid In sulfuric acid at 80 - 90°C; |


| Conditions | Yield |
|---|---|
| heating on water bath; equimolar amts. of educts; forming small amt. of triuret; |

-
-
75-52-5
nitromethane

-
-
7446-11-9
sulfur trioxide

-
A
-
124-38-9
carbon dioxide

-
B
-
5329-14-6
aminosulfonic acid

-
C
-
2950-43-8
hydroxylamine-O-sulfonic acid

| Conditions | Yield |
|---|---|
| above 0°C; |

-
-
7446-09-5
sulfur dioxide

-
-
7664-41-7
ammonia

-
A
-
79796-32-0
S4N2

-
B
-
5329-14-6
aminosulfonic acid

-
C
-
7704-34-9
sulfur

-
D
-
28950-34-7
tetrasulfur tetranitride

| Conditions | Yield |
|---|---|
| With water In gas excess of SO2, 50-80°C, the solid product hydrolysed by water; |


| Conditions | Yield |
|---|---|
| In thionyl chloride amidosulfuric acid placed into a flask and suspended in thionyl dichloride under dry N2; HSO3Cl added, then the flask fitted with a reflux condenser, connected to a CaCl2 drying tube; heated at 130°C for 24 h; reaction mixture fractionally distilled under vacuum; main fraction collected at 95°C/650 Pa; | 99% |
-
-
495-69-2
Hippuric Acid

-
-
5329-14-6
aminosulfonic acid

-
-
143230-70-0
4-chloro-3-(2,2,3,3,3-pentafluoropropoxymethyl)aniline

-
-
143230-69-7
2-phenyl-4,5-oxazoledione 4-[4-chloro-3-(2,2,3,3,3,-pentafluoropropoxymethyl)-phenylhydrazone]

| Conditions | Yield |
|---|---|
| With hydrogenchloride; sodium acetate; acetic anhydride; acetic acid; sodium nitrite In water | 98.1% |


| Conditions | Yield |
|---|---|
| With chlorosulfonic acid at 130℃; for 24h; Inert atmosphere; | 98% |
-
-
495-69-2
Hippuric Acid

-
-
5329-14-6
aminosulfonic acid

-
-
143230-70-0
4-chloro-3-(2,2,3,3,3-pentafluoropropoxymethyl)aniline

| Conditions | Yield |
|---|---|
| With hydrogenchloride; sodium acetate; acetic anhydride; acetic acid; sodium nitrite In water | 97.95% |
| With hydrogenchloride; sodium acetate; acetic anhydride; acetic acid; sodium nitrite In water | 97.1% |


| Conditions | Yield |
|---|---|
| With KOH In water calcd. amts. of educts; mixing cold aq. solns. of educts under stirring; product pptd. after 2-3 d; sucking off, washing with cold H2O, drying; | 97% |

| Conditions | Yield |
|---|---|
| With NaOH In water calcd. amts. of educts; pptn. 3-4 d; washing with alc. and ether; drying in vac. over P2O5; | 97% |
| With sodium hydroxide In water 1.5 fold molar excess of NaOH; amidosulfuric acid neutralized with NaOH soln.; adding hot aq. soln. of HgCl2; pptd. after 3-4 d; washing with H2O, alc. and ether; drying in vac.; | |
| With NaOH In water 1.5 fold molar excess of NaOH; amidosulfuric acid neutralized with NaOH soln.; adding hot aq. soln. of HgCl2; pptd. after 3-4 d; washing with H2O, alc. and ether; drying in vac.; |
-
-
7790-94-5
chlorosulfonic acid

-
-
7719-09-7
thionyl chloride

-
-
5329-14-6
aminosulfonic acid

-
-
15873-42-4
bis(chlorosulfonyl)amine

| Conditions | Yield |
|---|---|
| at 70 - 130℃; for 24h; | 96% |
| at 130℃; Inert atmosphere; | Ca. 449 g |

-
-
4519-40-8
2,3-difluoroanilline

-
-
5329-14-6
aminosulfonic acid

-
-
79-10-7
acrylic acid

-
-
94977-52-3
2,4-Difluorocinnamic acid

| Conditions | Yield |
|---|---|
| With sulfuric acid; sodium nitrite; palladium diacetate In water | 95% |

-
-
7758-19-2
sodium chlorite

-
-
70991-79-6
4-iodo-α-methylbenzeneacetaldehyde

-
-
5329-14-6
aminosulfonic acid

-
-
34645-72-2
(RS)-2-(4'-iodophenyl)propanoic acid

| Conditions | Yield |
|---|---|
| With sodium hydroxide In water; 1,2-dichloro-ethane | 94% |

-
-
7758-19-2
sodium chlorite

-
-
70991-79-6
4-iodo-α-methylbenzeneacetaldehyde

-
-
5329-14-6
aminosulfonic acid

-
-
34645-72-2
(RS)-2-(4'-iodophenyl)propanoic acid

| Conditions | Yield |
|---|---|
| With sodium hydroxide; sodium hydrogen sulphite In water; 1,2-dichloro-ethane | 94% |


| Conditions | Yield |
|---|---|
| With NaOH In water calcd. amts. of educts; adding aq. suspension of freshly prepared HgO into hot neutralized soln. of amidosulfuric acid; cooling, sucking off; washing with cold H2O; drying; | 94% |
| With NaOH In water calcd. amts. of educts; adding boiling aq. soln. of neutralized amidosulfuric acid dropwise into aq. suspension of yellow HgO; product pptd. by cooling after 24 h; | 79% |

| Conditions | Yield |
|---|---|
| With KOH In water calcd. amts. of educts; mixing cold aq. solns. of educts under stirring, heating, cooling; decanting with cold H2O; sucking off; product contains Cl; | 94% |
-
-
5329-14-6
aminosulfonic acid

-
-
78919-26-3
3β,7β-dihydroxy-5β-cholan-24-oic acid

| Conditions | Yield |
|---|---|
| In DMF (N,N-dimethyl-formamide) at 45 - 90℃; for 0.5h; | 93.7% |

| Conditions | Yield |
|---|---|
| With KOH In water adding amidosulfuric acid into aq. KOH soln.; this soln. adding to aq. suspension of freshly pptd. HgO; shaking for 1 h; filtrate storing for several h; sucking off the ppt.; washing with cold H2O, drying; | 92% |
| With potassium hydroxide In not given calcd. amts. of educts; amidosulfuric acid neutralized with KOH soln., adding more KOH soln. and freshly prepared HgO; heating 1 h on water bath; product pptd. from filtrate at ambient temp.; sucking off; recrystn. from 0.5 % KOH soln.; washing with alc. and ether; drying in air; storage in vac. over P2O5; | |
| With KOH In not given calcd. amts. of educts; amidosulfuric acid neutralized with KOH soln., adding more KOH soln. and freshly prepared HgO; heating 1 h on water bath; product pptd. from filtrate at ambient temp.; sucking off; recrystn. from 0.5 % KOH soln.; washing with alc. and ether; drying in air; storage in vac. over P2O5; |
-
-
7758-19-2
sodium chlorite

-
-
5329-14-6
aminosulfonic acid

-
-
203919-39-5
2-(benzo[1,3]dioxol-5-yl)-4-chloro-6-isopropoxy-2H-chromene-3-carbaldehyde

-
-
203917-92-4
2-(benzo[1,3]dioxol-5-yl)-4-chloro-6-isopropoxy-2H-chromene-3-carboxylic acid

| Conditions | Yield |
|---|---|
| With sodium hydroxide; sodium sulfite In water; toluene | 91% |

-
-
5329-14-6
aminosulfonic acid

-
-
88-73-3
2-Chloronitrobenzene

-
B
-
97-08-5
3-nitro-4-chlorosulfonyl chloride

| Conditions | Yield |
|---|---|
| With chlorosulfonic acid | A n/a B 90.8% |

-
-
5329-14-6
aminosulfonic acid

-
-
67387-76-2
3-cyclopentyloxy-4-methoxybenzylaldehyde

-
-
144036-17-9
3-cyclopentyloxy-4-methoxy benzoic acid

| Conditions | Yield |
|---|---|
| With sodium chloride In water; acetic acid | 90% |
| With sodium In water; acetic acid |
-
-
7758-19-2
sodium chlorite

-
-
100611-61-8
(1-phenyl-cyclopentyl)-acetaldehyde

-
-
5329-14-6
aminosulfonic acid

-
-
70239-29-1
(1-phenyl-cyclopentyl)-acetic acid

| Conditions | Yield |
|---|---|
| In tert-butyl alcohol | 90% |

-
-
1310-73-2
sodium hydroxide

-
-
5329-14-6
aminosulfonic acid

-
-
10035-10-6, 12258-64-9
hydrogen bromide

-
-
134509-56-1
N-bromosulfamic acid sodium salt

| Conditions | Yield |
|---|---|
| Stage #1: hydrogen bromide With dihydrogen peroxide In water at 61.1011℃; Stage #2: aminosulfonic acid In water at 34.99℃; Stage #3: sodium hydroxide With sode de l'acide trichloroisocyanurique Product distribution / selectivity; more than 3 stages; | 88.4% |
| Stage #1: hydrogen bromide With dihydrogen peroxide In water at 61.1011℃; Stage #2: aminosulfonic acid In water at 29.4344 - 34.99℃; Stage #3: sodium hydroxide With trichloroisocyanuric acid Product distribution / selectivity; more than 3 stages; | 80.1% |
| Stage #1: hydrogen bromide With dihydrogen peroxide In water at 61.1011℃; Stage #2: aminosulfonic acid In water at 34.99℃; Stage #3: sodium hydroxide With calcium hypochlorite Product distribution / selectivity; more than 3 stages; | 70.8% |

-
-
5329-14-6
aminosulfonic acid

-
-
13244-33-2
2-amino-5-methoxybenzenesulfonic acid

-
-
40567-33-7
2-sulfo-4-methoxybenzoic acid

| Conditions | Yield |
|---|---|
| With sulfuric acid; sodium nitrite; palladium(II) chloride In water; acetonitrile | 88% |

| Conditions | Yield |
|---|---|
| In water | 86% |
-
-
5329-14-6
aminosulfonic acid

-
-
13823-50-2
potassium sulfamate

| Conditions | Yield |
|---|---|
| With potassium hydroxide In water at 20℃; for 0.416667h; | 86% |
-
-
1310-73-2
sodium hydroxide

-
-
5329-14-6
aminosulfonic acid

-
-
7647-15-6
sodium bromide

-
-
134509-56-1
N-bromosulfamic acid sodium salt

| Conditions | Yield |
|---|---|
| Stage #1: sodium hydroxide; aminosulfonic acid; sodium bromide In water at 26.6567 - 32.2122℃; pH=2; Stage #2: With trichloroisocyanuric acid In water for 0.25 - 0.5h; Product distribution / selectivity; | 85% |
| Stage #1: aminosulfonic acid; sodium bromide In water at 26.6567 - 32.2122℃; pH=2; Stage #2: With calcium hypochlorite In water for 0.25 - 0.5h; Stage #3: sodium hydroxide In water at 26.6567 - 32.2122℃; Product distribution / selectivity; |

-
-
178050-26-5
1-(2,6-dinitro-4-trifluoromethylphenyl)-2-chloro-3-cyano-4-dichlorofluoro-methylsulfenyl-5-methylpyrrole

-
-
5329-14-6
aminosulfonic acid

-
-
183288-91-7
2-chloro-3-cyano-1-(2,6-dichloro-4-trifluoromethylphenyl)-4-(dichlorofluoromethylsulfenyl)-5-methylpyrrole

| Conditions | Yield |
|---|---|
| With lithium chloride In 1-methyl-pyrrolidin-2-one | 85% |

-
-
374562-42-2
2-amino-6-(1-hydroxyethyl)-benzoic acid (sodium salt)

-
-
5329-14-6
aminosulfonic acid

-
-
7783-06-4
hydrogen sulfide

-
-
135217-37-7
7-mercapto-3-methyl-3H-isobenzofuran-1-one

| Conditions | Yield |
|---|---|
| With hydrogenchloride; sodium hydroxide; sodium hydrogen sulphite; acetic acid; triphenylphosphine In water; toluene | 85% |


| Conditions | Yield |
|---|---|
| In water | 85% |

| Conditions | Yield |
|---|---|
| With sulfuric acid; sodium nitrite; palladium diacetate In water | 84% |

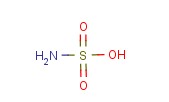
Sulfamic acid Chemical Properties
Molecular structure of Sulfamic acid (CAS NO.5329-14-6) is:
Product Name: Sulfamic acid
CAS Registry Number: 5329-14-6
IUPAC Name: sulfamic acid
Molecular Weight: 97.09372 [g/mol]
Molecular Formula: H3NO3S
XLogP3-AA: -1.6
H-Bond Donor: 2
H-Bond Acceptor: 4
EINECS: 226-218-8
Melting Point: 215-225 °C (dec.)(lit.)
Surface Tension: 101.5 dyne/cm
Density: 1.913 g/cm3
Water Solubility: 146.8 g/L (20 °C)
Stability: Stable.
Product Categories: Intermediates of Dyes and Pigments;INORGANIC & ORGANIC CHEMICALS;Intermediates;Inorganics
Sulfamic acid Uses
Sulfamic acid (CAS NO.5329-14-6) is uaually used as herbicides, fire agents, paper and textile softeners,and metal cleaning agent. It can be used as standard in alkalimetry, in acid cleaning, in nitrite removal, in chlorine stabilization for use in swimming pools, cooling towers, paper mills.
Sulfamic acid Toxicity Data With Reference
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| guinea pig | LD50 | oral | 1050mg/kg (1050mg/kg) | BEHAVIORAL: ALTERED SLEEP TIME (INCLUDING CHANGE IN RIGHTING REFLEX) BEHAVIORAL: EXCITEMENT BEHAVIORAL: REGIDITY | Gigiena i Sanitariya. For English translation, see HYSAAV. Vol. 52(10), Pg. 88, 1987. |
| mouse | LD50 | oral | 1312mg/kg (1312mg/kg) | BEHAVIORAL: ALTERED SLEEP TIME (INCLUDING CHANGE IN RIGHTING REFLEX) BEHAVIORAL: EXCITEMENT BEHAVIORAL: REGIDITY | Gigiena i Sanitariya. For English translation, see HYSAAV. Vol. 52(10), Pg. 88, 1987. |
| rat | LD50 | oral | 3160mg/kg (3160mg/kg) | "Sbornik Vysledku Toxixologickeho Vysetreni Latek A Pripravku," Marhold, J.V., Institut Pro Vychovu Vedoucicn Pracovniku Chemickeho Prumyclu Praha, Czechoslovakia, 1972Vol. -, Pg. 18, 1972. | |
| rat | LDLo | intraperitoneal | 100mg/kg (100mg/kg) | Journal of Industrial Hygiene and Toxicology. Vol. 25, Pg. 26, 1943. |
Sulfamic acid Safety Profile
Hazard Codes:  Xi
Xi
Risk Statements: 36/38-52/53
R36/38:Irritating to eyes and skin.
R52/53:Harmful to aquatic organisms, may cause long-term adverse effects in the aquatic environment.
Safety Statements: 26-28-61-28A
S26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice.
S28:After contact with skin, wash immediately with plenty of soap-suds.
S61:Avoid release to the environment. Refer to special instructions / safety data sheets.
S28:After contact with skin, wash immediately with plenty of soap-suds.
RIDADR: UN 2967 8/PG 3
WGK Germany: 1
RTECS: WO5950000
HazardClass: 8
PackingGroup: III
HS Code: 28111980
Sulfamic acid Specification
Sulfamic acid , its cas register number is 5329-14-6. It also can be called Amidosulfonic acid ; Amidosulfuric acid ; Aminosulfonic acid .It is a white crystalline solid.It reacts exothermically with bases. Aqueous solutions are acidic and corrosive.
Related Products
- Sulfamic acid
- Sulfamic acid,2-aminoethyl ester
- Sulfamic acid,N-(4-chlorophenyl)-
- Sulfamic acid,N-cyclohexyl-, compd. with N-(3-phenylpropoxy)guanidine (1:1)
- Sulfamic acid,N-hydroxy-, sodium salt (1:1)
- Sulfamic acid,N-methyl-
- 5329-22-6
- 53292-89-0
- 5329-29-3
- 532932-99-3
- 5329-33-9
- 532-93-4
- 53293-43-9
- 532-94-5
- 53295-44-6
- 53296-64-3
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View