-
Name
2,3-Dimethylbromobenzene
- EINECS 209-398-2
- CAS No. 576-23-8
- Article Data29
- CAS DataBase
- Density 1.339 g/cm3
- Solubility Not miscible or difficult to mix in water.
- Melting Point -20.49°C (estimate)
- Formula C8H9Br
- Boiling Point 211 °C at 760 mmHg
- Molecular Weight 185.063
- Flash Point 84 °C
- Transport Information
- Appearance Clear colourless to light yellow liquid
- Safety 26-36/37/39-24/25
- Risk Codes 36/37/38
-
Molecular Structure
-
Hazard Symbols
 Xi
Xi
- Synonyms o-Xylene,3-bromo- (6CI,7CI,8CI);1-Bromo-2,3-dimethylbenzene;
- PSA 0.00000
- LogP 3.06590
Synthetic route

| Conditions | Yield |
|---|---|
| With bromine; acetic acid | A 82% B 18% |
| With bromine; iodine In dichloromethane at -10 - 20℃; for 7.58333h; | A 75% B 15% |
| With hydrogenchloride; sodium hypochlorite; sodium bromide In water at 29.85℃; for 8h; Product distribution; Further Variations:; Temperatures; | A 24.6% B 68.5% |

| Conditions | Yield |
|---|---|
| With nitrogen(II) oxide; copper(ll) bromide In acetonitrile at -78 - 25℃; | 73% |
| With hydrogen bromide diazotization; | 40% |
| With hydrogen bromide Diazotization.Erwaermen der Diazoniumsalz-Loesung mit Kupfer-Pulver; |

| Conditions | Yield |
|---|---|
| With bromine; triphenylphosphine In acetonitrile | 60% |
| With bromine; triphenylphosphine In acetonitrile at 25℃; for 48h; | 41.1% |
-
-
593-53-3
Methyl fluoride

-
-
95-46-5
2-methylphenyl bromide

-
A
-
576-22-7
2-Bromo-m-xylene

-
B
-
553-94-6
4-bromo-m-xylene

-
C
-
583-70-0
1-bromo-2,4-dimethylbenzene

-
D
-
576-23-8
2,3-dimethylbromobenzene

| Conditions | Yield |
|---|---|
| With oxygen at 40℃; under 720 Torr; Mechanism; Irradiation; | A n/a B n/a C 13.7% D 32.8% |

-
-
22364-25-6
4-bromo-2,3-dimethylaniline

-
-
576-23-8
2,3-dimethylbromobenzene

| Conditions | Yield |
|---|---|
| With hydrogen bromide; sodium nitrite anschliessend Behandeln mit Natriumhypophosphit und wss. Bromwasserstoffsaeure; |
-
-
75366-04-0
2-methyl-1H-cyclopropabenzene

-
A
-
576-22-7
2-Bromo-m-xylene

-
B
-
576-23-8
2,3-dimethylbromobenzene

| Conditions | Yield |
|---|---|
| With bromine; tri-n-butyl-tin hydride Product distribution; Mechanism; multistep reaction; reaction regioselectivity; a series of reagents; |

| Conditions | Yield |
|---|---|
| With aluminium trichloride In tetrachloromethane; nitromethane |
-
-
95-47-6
o-xylene

-
A
-
583-71-1
4-bromo-o-xylene

-
B
-
576-23-8
2,3-dimethylbromobenzene

-
C
-
89-92-9
2-methylbenzyl bromide

-
D
-
91-13-4
α,α'-dibromo-o-xylene

| Conditions | Yield |
|---|---|
| With bromine In water for 0.333333h; Ambient temperature; Irradiation; | A 3 % Chromat. B 2 % Chromat. C 16 % Chromat. D 79 % Chromat. |


-
-
576-23-8
2,3-dimethylbromobenzene

| Conditions | Yield |
|---|---|
| With steam; sulfuric acid |

-
-
576-23-8
2,3-dimethylbromobenzene

| Conditions | Yield |
|---|---|
| With steam; sulfuric acid |

-
-
576-23-8
2,3-dimethylbromobenzene

| Conditions | Yield |
|---|---|
| With sulfuric acid Destillieren mit ueberhitztem Wasserdampf; |

-
-
576-23-8
2,3-dimethylbromobenzene

| Conditions | Yield |
|---|---|
| With sulfuric acid Destillieren mit ueberhitztem Wasserdampf; |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: Fe, Br2 / CCl4 2: AlCl3 / CCl4; nitromethane View Scheme |
-
-
576-23-8
2,3-dimethylbromobenzene

-
-
127168-82-5
1-bromo-2,3-bis(bromomethyl)benzene

| Conditions | Yield |
|---|---|
| With N-Bromosuccinimide In tetrachloromethane Irradiation; | 100% |
| With N-Bromosuccinimide; Perbenzoic acid In tetrachloromethane for 0.5h; Heating; Irradiation; | 94.1% |
| With N-Bromosuccinimide; 2,2'-azobis(isobutyronitrile) In tetrachloromethane for 0.5h; Heating; | 88% |
-
-
576-23-8
2,3-dimethylbromobenzene

-
-
78-95-5
chloroacetone

-
-
42432-45-1
2-(2,3-dimethylphenyl)-2-methyloxirane

| Conditions | Yield |
|---|---|
| Stage #1: 2,3-dimethylbromobenzene With n-butyllithium In tetrahydrofuran; hexane at -78℃; for 0.5h; Stage #2: chloroacetone In tetrahydrofuran; hexane; toluene at -78 - 20℃; for 1.33333h; Product distribution / selectivity; | 100% |
| Stage #1: 2,3-dimethylbromobenzene With n-butyllithium In tetrahydrofuran; hexane at -78℃; for 0.5h; Stage #2: chloroacetone In tetrahydrofuran; hexane; toluene at -78 - 20℃; Product distribution / selectivity; | 100% |
-
-
576-23-8
2,3-dimethylbromobenzene

-
-
176721-02-1
4-<(2,3-dimethylphenyl)carbonyl>-1-(triphenylmethyl)imidazole

| Conditions | Yield |
|---|---|
| Stage #1: 2,3-dimethylbromobenzene With iodine; magnesium In tetrahydrofuran at 65 - 70℃; for 1h; Stage #2: morpholine[1-(triphenylmethyl)-1H-imidazol-4-yl]methanone In tetrahydrofuran at 0 - 40℃; for 1h; | 100% |

| Conditions | Yield |
|---|---|
| With tetrakis(triphenylphosphine) palladium(0); potassium carbonate In N,N-dimethyl-formamide at 105℃; for 24h; | 96% |

| Conditions | Yield |
|---|---|
| With potassium carbonate; PdCl2[diethyl(o-ethylphenyl)phosphane]2 In N,N-dimethyl-formamide at 150℃; for 0.5h; Suzuki-Miyaura coupling reaction; microwave irradiation; | 96% |
| With C21H25ClN2Pd; potassium carbonate In toluene at 80℃; for 2h; Suzuki-Miyaura Coupling; | 82% |
| With caesium carbonate; cis-dibromobis[3-(2-propenyl)benzothiazolin-2-ylidene]Pd(II) In N,N-dimethyl-formamide at 100℃; for 19h; Suzuki-Miyaura coupling; | 80 % Chromat. |
| With potassium phosphate; C17H14O*C108H156N3O6P9*3Pd In tetrahydrofuran at 79.84℃; for 24h; Suzuki coupling; Inert atmosphere; | 99 %Spectr. |
-
-
576-23-8
2,3-dimethylbromobenzene

-
-
68-12-2, 33513-42-7
N,N-dimethyl-formamide

-
-
5779-93-1
2,3-dimethylbenzaldehyde

| Conditions | Yield |
|---|---|
| Stage #1: 2,3-dimethylbromobenzene With magnesium In tetrahydrofuran for 1h; Inert atmosphere; Reflux; Stage #2: N,N-dimethyl-formamide In tetrahydrofuran at 20℃; for 2h; Concentration; Temperature; | 96% |
-
-
576-23-8
2,3-dimethylbromobenzene

-
-
116795-55-2
1-(1-triphenylmethyl-1H-imidazol-4-yl)-1-ethanone

-
-
176721-03-2
4-<1-(2,3-dimethylphenyl)-1-hydroxyethyl>-1-(triphenylmethyl)imidazole

| Conditions | Yield |
|---|---|
| Stage #1: 2,3-dimethylbromobenzene With magnesium In tetrahydrofuran for 1h; Reflux; Stage #2: 1-(1-triphenylmethyl-1H-imidazol-4-yl)-1-ethanone In tetrahydrofuran at 20℃; for 2h; Time; | 89.7% |
-
-
576-23-8
2,3-dimethylbromobenzene


| Conditions | Yield |
|---|---|
| With tris-(dibenzylideneacetone)dipalladium(0); sodium t-butanolate; ruphos In water; tert-butyl alcohol at 100℃; for 18h; Suzuki Coupling; Schlenk technique; Inert atmosphere; | 89% |
-
-
1068471-18-0
methyl 3-(4-ethynylphenyl)propanoate

-
-
576-23-8
2,3-dimethylbromobenzene

-
-
1082059-34-4
methyl 3-(4-((2,3-dimethylphenyl)ethynyl)phenyl)propanoate

| Conditions | Yield |
|---|---|
| With copper(l) iodide; sodium tetrachloropalladate(II); 2-[bis(1,1-dimethylethyl)phosphino]-1-phenyl-1H-indole In 2,3-dimethyl-2,3-diaminobutane at 80℃; Sonogashira coupling; Inert atmosphere; | 86% |
| With bis-triphenylphosphine-palladium(II) chloride; copper(l) iodide; triethylamine In N,N-dimethyl-formamide at 50℃; Sonogashira coupling; Inert atmosphere; | 14% |

| Conditions | Yield |
|---|---|
| Stage #1: 2,3-dimethylbromobenzene With iodine; magnesium In tetrahydrofuran for 1h; Reflux; Stage #2: 4(5)formylimidazole at 50℃; for 4h; | 86% |

| Conditions | Yield |
|---|---|
| Stage #1: 2,3-dimethylbromobenzene With tert.-butyl lithium In diethyl ether Stage #2: carbon dioxide | 84% |
| (i) Mg, (ii) /BRN= 1900390/; Multistep reaction; |

| Conditions | Yield |
|---|---|
| With tris-(dibenzylideneacetone)dipalladium(0); tri-tert-butyl phosphine In tetrahydrofuran; toluene at 20℃; for 1h; Schlenk technique; Inert atmosphere; | 84% |
-
-
576-23-8
2,3-dimethylbromobenzene

-
-
134640-85-0
2,3-dimethylphenylmagnesium bromide

-
-
7495-46-7
2,2′,3,3′-tetramethylbiphenyl

| Conditions | Yield |
|---|---|
| triphenylphosphine; nickel dibromide In tetrahydrofuran Heating; | 83% |

| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine; tris-(dibenzylideneacetone)dipalladium(0); tri-tert-butyl phosphine In N,N-dimethyl-formamide; toluene at 100℃; for 18h; Product distribution / selectivity; | 83% |

| Conditions | Yield |
|---|---|
| With bis(tri-t-butylphosphine)palladium(0) In toluene at 20℃; for 1h; | 83% |
-
-
576-23-8
2,3-dimethylbromobenzene

-
-
666829-97-6
2-(N,N-diethylcarboxamido)-4-methoxy-3-trimethylsilylphenylboronic acid

| Conditions | Yield |
|---|---|
| With tetrakis(triphenylphosphine) palladium(0); sodium carbonate In 1,2-dimethoxyethane; ethanol; water at 100℃; for 16h; Suzuki-Miyaura Coupling; | 83% |
-
-
576-23-8
2,3-dimethylbromobenzene

-
-
106877-28-5
2-(Nonafluorobutyl)aniline

| Conditions | Yield |
|---|---|
| Stage #1: 2,3-dimethylbromobenzene With magnesium In tetrahydrofuran at 70℃; Stage #2: 2-(Nonafluorobutyl)aniline In tetrahydrofuran at -70 - 23℃; for 13h; Inert atmosphere; Stage #3: With water; oxygen In tetrahydrofuran | 81% |

| Conditions | Yield |
|---|---|
| Stage #1: 2,3-dimethylbromobenzene With iodine; magnesium In tetrahydrofuran at 22℃; for 0.5h; Inert atmosphere; Stage #2: bromobenzene With nickel dichloride In tetrahydrofuran at 66℃; for 3h; Inert atmosphere; | 80% |
-
-
576-23-8
2,3-dimethylbromobenzene

-
-
57090-88-7
1H-imidazole-4-carbonitrile

| Conditions | Yield |
|---|---|
| Stage #1: 2,3-dimethylbromobenzene With magnesium; ethylene dibromide In tetrahydrofuran at 40 - 65℃; for 1h; Stage #2: 1H-imidazole-4-carbonitrile With isopropylmagnesium chloride In tetrahydrofuran at -5 - 0℃; for 2h; Stage #3: With sulfuric acid In tetrahydrofuran; water at 30℃; for 3h; | 80% |
-
-
3612-20-2
1-phenylmethyl-4-piperidone

-
-
576-23-8
2,3-dimethylbromobenzene

-
-
1370256-31-7
1-benzyl-4-(2,3-dimethylphenyl)piperidin-4-ol

| Conditions | Yield |
|---|---|
| Stage #1: 2,3-dimethylbromobenzene With iodine; magnesium In tetrahydrofuran Grignard reaction; Inert atmosphere; Reflux; Stage #2: 1-phenylmethyl-4-piperidone In tetrahydrofuran at 20℃; for 3h; Grignard reaction; Inert atmosphere; Cooling with ice; | 79% |
| With iodine; magnesium In tetrahydrofuran at 0 - 70℃; for 1.5h; Inert atmosphere; |
2,3-Dimethylbromobenzene Chemical Properties
IUPAC Name: 1-Bromo-2,3-dimethylbenzene
Synonyms of 2,3-Dimethylbromobenzene (CAS NO.576-23-8): 3-Bromo-1,2-dimethylbenzene ; 3-Bromo-o-xylene
CAS NO: 576-23-8
The Molecular Formula: C8H9Br
The Molecular Weight: 185.06
Molecular Structure : 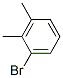
EINECS: 209-398-2
H bond acceptors: 0
H bond donors: 0
Freely Rotating Bonds: 0
Polar Surface Area: Å2
Index of Refraction: 1.543
Molar Refractivity: 43.59 cm3
Molar Volume: 138.1 cm3
Surface Tension: 33.7 dyne/cm
Density: 1.339 g/cm3
Flash Point: 84 °C
Enthalpy of Vaporization: 42.91 kJ/mol
Boiling Point: 211 °C at 760 mmHg
Vapour Pressure: 0.27 mmHg at 25°C
Appearance: Clear colourless to light yellow liquid
Product Categories of 2,3-Dimethylbromobenzene (CAS NO.576-23-8): Aromatic Hydrocarbons (substituted) & Derivatives;Halogen toluene;Benzene derivates;Bromine Compounds
SMILES: Cc1cccc(Br)c1C
InChI: InChI=1/C8H9Br/c1-6-4-3-5-8(9)7(6)2/h3-5H,1-2H3
InChIKey: WLPXNBYWDDYJTN-UHFFFAOYAR
Std. InChI: InChI=1S/C8H9Br/c1-6-4-3-5-8(9)7(6)2/h3-5H,1-2H3
Std. InChIKey: WLPXNBYWDDYJTN-UHFFFAOYSA-N
2,3-Dimethylbromobenzene Safety Profile
Safety Information about 2,3-Dimethylbromobenzene (CAS NO.576-23-8):
Hazard Codes:  Xi
Xi
Risk Statements: 36/37/38
R36/37/38:Irritating to eyes, respiratory system and skin.
Safety Statements: 26-36/37/39-24/25
S26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice.
S24/25: Avoid contact with skin and eyes.
S36/37/39: Wear suitable protective clothing, gloves and eye/face protection.
RIDADR: UN 2810 6.1/PG 3
WGK Germany: 3
Hazard Note: Irritant
2,3-Dimethylbromobenzene Specification
General Information: As in any fire, wear a self-contained breathing apparatus in pressure-demand, MSHA/NIOSH (approved or equivalent), and full protective gear. Will burn if involved in a fire. Combustible liquid.
Extinguishing Media: Use water spray, dry chemical, carbon dioxide, or chemical foam.
Handling: Avoid breathing dust, vapor, mist, or gas. Avoid contact with skin and eyes. Avoid ingestion and inhalation.
Storage: Keep 2,3-Dimethylbromobenzene (CAS NO.576-23-8) away from sources of ignition. Store in a cool, dry place. Store in a tightly closed container.
Related Products
- 20,22-DIHYDRODIGITOXIN
- 20,29,30-Trinorlupane,(17alpha)-
- 20-ETHYL-6-β,8-DIHYDROXY-1-α-METHOXY-4-METHYLHETERATISAN-14-ONE
- 20-Ethylprostaglandin F2-alpha
- 20-Isopropylcholanthrene
- 20-METHYLCHOLANTHREN-15-ONE
- 20-METHYLCHOLANTHRENE PICRATE
- 20-METHYLCHOLANTHRENE-TRINITRO-BENZENE
- 20(S)-Ginsenoside C-K
- 2,10-DIFLUOROBENZO(rst)PENTAPHENE
- 576-24-9
- 57-62-5
- 5762-56-1
- 57625-74-8
- 57625-97-5
- 576-26-1
- 5762-64-1
- 57627-75-5
- 57632-67-4
- 5763-44-0
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View