-
Name
4-Bromobiphenyl
- EINECS 202-176-6
- CAS No. 92-66-0
- Article Data321
- CAS DataBase
- Density 1.363 g/cm3
- Solubility insoluble in water
- Melting Point 82-86 °C(lit.)
- Formula C12H9Br
- Boiling Point 309.8 °C at 760 mmHg
- Molecular Weight 233.107
- Flash Point 141.8 °C
- Transport Information UN 3152 9/PG 2
- Appearance white powder
- Safety 22-24/25-37/39-26
- Risk Codes 36/37/38
-
Molecular Structure
-
Hazard Symbols
 Xi
Xi
- Synonyms Biphenyl,4-bromo- (6CI,8CI);(4-Bromophenyl)benzene;1-Bromo-4-phenylbenzene;4-Biphenylbromide;4-Biphenylyl bromide;4-Bromo-1,1'-biphenyl;4-Bromobiphenyl;4-Bromodiphenyl;4'-Bromobiphenyl;NSC 406933;PBB 3;p-Biphenylyl bromide;p-Bromobiphenyl;p-Bromodiphenyl;p-Phenylbromobenzene;p-Phenylphenyl bromide;
- PSA 0.00000
- LogP 4.11610
Synthetic route

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 20℃; for 21h; | A 100% B n/a C n/a |


| Conditions | Yield |
|---|---|
| With sodium carbonate In 1-methyl-pyrrolidin-2-one at 100℃; for 6h; Suzuki-Miyaura Coupling; Inert atmosphere; | 99% |
| With potassium carbonate In water at 90℃; for 1h; Suzuki-Miyaura coupling; chemoselective reaction; | 98% |
| With potassium carbonate In ethanol; water at 80℃; for 0.583333h; Catalytic behavior; Suzuki-Miyaura Coupling; | 96% |

| Conditions | Yield |
|---|---|
| With sodium nitrite In acetonitrile at 80℃; for 1h; Sealed tube; | 99% |
| With N-Bromosuccinimide In acetonitrile at 80℃; for 12h; | 98% |
| With tetrabutylammomium bromide; copper(ll) bromide In water at 100℃; Sealed tube; | 88% |
| With N-Bromosuccinimide; potassium acetate In acetonitrile at 50℃; for 4h; | 85% |
-
-
143-66-8
sodium tetraphenyl borate

-
-
65826-67-7
bis(4-bromophenyl)iodonium bromide

-
-
92-66-0
4-bromo-1,1'-biphenyl

| Conditions | Yield |
|---|---|
| palladium dichloride In water for 0.00555556h; Suzuki coupling; microwave irradiation; | 97% |
| for 0.0833333h; Suzuki coupling; microwave irradiation; | 94% |
| In water at 100℃; for 0.05h; Suzuki coupling reaction; microwave irradiation; | 91% |
| In water at 100℃; for 0.05h; Suzuki coupling; microwave irradiation; | 91% |

| Conditions | Yield |
|---|---|
| With potassium carbonate In ethanol; water at 50℃; for 2.5h; Suzuki-Miyaura Coupling; Inert atmosphere; Schlenk technique; | 97% |
| With potassium carbonate In ethanol; water at 100℃; for 1.5h; Suzuki-Miyaura Coupling; Green chemistry; | 94% |
| With potassium carbonate In ethanol at 80℃; for 3h; Suzuki-Miyaura Coupling; Schlenk technique; | 92% |

| Conditions | Yield |
|---|---|
| With C84H60Cl6Fe3N6Pd3; triethylamine at 20℃; for 0.383333h; | 97% |
| With sodium carbonate In water at 80℃; for 4.5h; Catalytic behavior; Suzuki Coupling; Green chemistry; | 90% |
| With tetrabutylammomium bromide In ethanol; water at 90℃; for 0.666667h; Catalytic behavior; Suzuki Coupling; Inert atmosphere; | 84% |
| With potassium carbonate In ethanol; water at 50℃; for 12h; Catalytic behavior; Suzuki-Miyaura Coupling; Green chemistry; | 65% |

| Conditions | Yield |
|---|---|
| With sodium carbonate In water at 80℃; for 4.16667h; Catalytic behavior; Suzuki Coupling; Green chemistry; | 93% |

| Conditions | Yield |
|---|---|
| With copper diacetate; palladium dichloride In dimethyl sulfoxide; acetonitrile at 90℃; for 2h; | 92% |

| Conditions | Yield |
|---|---|
| With tetrabutylammomium bromide; nickel; sodium carbonate In ethanol at 80℃; for 6h; Suzuki-Miyaura reaction; | 91% |
| With potassium carbonate In N,N-dimethyl-formamide at 120℃; for 1h; Suzuki Coupling; | 85% |
| With aluminum oxide; potassium fluoride; palladium for 0.0333333h; Suzuki reaction; Heating; microwave irradiation; | 80% |
-
-
589-87-7
1,4-bromoiodobenzene

-
-
24341-81-9
2-phenyl-2,3-dihydro-1H-naphtho[1,8-de][1,3,2]diazaborinine

-
-
92-66-0
4-bromo-1,1'-biphenyl

| Conditions | Yield |
|---|---|
| With tris(dibenzylideneacetone)dipalladium(0) chloroform complex; potassium tert-butylate; triphenylphosphine In tetrahydrofuran; toluene at 70℃; for 24h; Suzuki-Miyaura Coupling; | 91% |

| Conditions | Yield |
|---|---|
| With copper(l) iodide; triphenyl-arsane; 1-butyl-3-methylimidazolium Tetrafluoroborate; bis(benzonitrile)palladium(II) dichloride at 80℃; for 18h; Stille coupling; | 90% |

| Conditions | Yield |
|---|---|
| With bis-triphenylphosphine-palladium(II) chloride In tetrahydrofuran for 2h; Heating; | 90% |
| With 3-aminopropyl-functionalized silica supported MCM-41-immobilized palladium(0)-[(pyridin-2-yl)methylidene]amine complex In tetrahydrofuran at 68℃; for 2h; Inert atmosphere; | 87% |
| tetrakis(triphenylphosphine) palladium(0) In tetrahydrofuran Heating; |

| Conditions | Yield |
|---|---|
| palladium diacetate In tetrahydrofuran at 20℃; for 1h; Stille reaction; | 90% |

| Conditions | Yield |
|---|---|
| With palladium diacetate; potassium carbonate; 4,5-bis(diphenylphos4,5-bis(diphenylphosphino)-9,9-dimethylxanthenephino)-9,9-dimethylxanthene In tert-butyl methyl ether at 60℃; for 6h; Suzuki Coupling; | 90% |

| Conditions | Yield |
|---|---|
| With copper diacetate; palladium dichloride In dimethyl sulfoxide; acetonitrile at 90℃; for 2h; | 90% |

| Conditions | Yield |
|---|---|
| at 100℃; for 12h; Sealed tube; | 90% |
-
-
768-32-1
trimethylphenylsilane

-
-
673-40-5
4-bromobenzenediazonium tetrafluoroborate

-
-
92-66-0
4-bromo-1,1'-biphenyl

| Conditions | Yield |
|---|---|
| With [bis(trifluoromethanesulfonyl)imidate](triphenylphosphine)gold(I) In acetonitrile at -78 - 20℃; Inert atmosphere; Irradiation; | 90% |
-
-
4482-01-3
o-benzenedisulfonimide

-
-
106-40-1
4-bromo-aniline

-
-
98-80-6
phenylboronic acid

-
-
92-66-0
4-bromo-1,1'-biphenyl

| Conditions | Yield |
|---|---|
| Stage #1: o-benzenedisulfonimide; 4-bromo-aniline With acetic acid at 0 - 5℃; for 0.166667h; Inert atmosphere; Stage #2: With isopentyl nitrite for 0.166667h; Inert atmosphere; Stage #3: phenylboronic acid With bis[(trifluoromethanesulfonyl)imidate]-2-(dicyclohexyl(2’,6’-dimethoxybiphenyl))phosphine gold(I); caesium carbonate In tetrahydrofuran at 20℃; for 2h; Suzuki-Miyaura Coupling; Inert atmosphere; chemoselective reaction; | 90% |


| Conditions | Yield |
|---|---|
| With hydrogen In methanol at 20℃; for 24h; | 100% |
| Stage #1: 4-bromo-1,1'-biphenyl With n-butyllithium In tetrahydrofuran at -50℃; for 0.5h; Stage #2: With phenylacetonitrile In tetrahydrofuran at -50 - 20℃; for 0.5h; Further stages; | 99% |
| With [RhCl2(p-cymene)]2; potassium tert-butylate In isopropyl alcohol at 20 - 100℃; for 24h; Inert atmosphere; | 97% |

| Conditions | Yield |
|---|---|
| With potassium phosphate; naphthidine di(radical cation)s-stabilized Pd nanoparticles In 1,4-dioxane at 80℃; for 5h; Suzuki-Miyaura cross-coupling reaction; | 100% |
| With potassium carbonate; [1,1'-bis{(S)-4-isopropyloxazolin-2-yl}ferrocene]Pd(II)Cl2 In toluene at 60℃; for 2h; Suzuki cross-coupling reaction; | 99% |
| With potassium phosphate; Pd (sulfur-containing palladacycle); tetra(n-tert-butyl)ammonium bromide In N,N-dimethyl-formamide at 130℃; for 2h; Suzuki cross-coupling; | 99% |
-
-
92-66-0
4-bromo-1,1'-biphenyl

-
-
150-76-5
4-methoxy-phenol

-
-
40843-54-7
4-(4-methoxyphenoxy)-1,1'-biphenyl

| Conditions | Yield |
|---|---|
| With copper(l) iodide; N,N-dimethylglycine hydrochoride; caesium carbonate In 1,4-dioxane at 90℃; for 18h; Product distribution / selectivity; | 100% |
| With copper(l) iodide; N,N-dimethylglycine hydrochoride; caesium carbonate In 1,4-dioxane at 90℃; | 91% |
| With silver(I) hexafluorophosphate; rhodium(II) acetate dimer; N,N-dicyclohexylbenzimidazolium hexafluorophosphate; sodium t-butanolate In toluene at 100℃; for 24h; | 60% |
| With potassium phosphate; (4s,6s)-2,4,5,6-tetra(9H-carbazol-9-yl)isophthalonitrile; [nickel(II) (4,4'-di-tert-butyl-2,2'-bipyridine)(bromide)2] at 25℃; for 36h; Schlenk technique; Inert atmosphere; Sealed tube; Irradiation; | 50% |
-
-
92-66-0
4-bromo-1,1'-biphenyl

-
-
5471-63-6
1,3-diphenylisobenzofuran

-
-
1006715-11-2
2-phenyl-9,10-diphenyl-9,10-epoxyanthracene

| Conditions | Yield |
|---|---|
| With Me2Zn(TMP)Li In tetrahydrofuran at 20℃; for 6h; | 100% |
-
-
45347-82-8
azetidine-3-ol

-
-
92-66-0
4-bromo-1,1'-biphenyl

-
-
1380499-22-8
1-biphenyl-4-yl-3-hydroxylcyclobutylamine

| Conditions | Yield |
|---|---|
| With potassium phosphate; copper(l) iodide; CVT-2537 In dimethyl sulfoxide at 90℃; for 24h; Inert atmosphere; | 100% |
-
-
92-66-0
4-bromo-1,1'-biphenyl

-
-
16419-60-6
2-Methylphenylboronic acid

-
-
31158-32-4
2-methyl-1,1’:4’,1’’-terphenyl

| Conditions | Yield |
|---|---|
| Stage #1: 2-Methylphenylboronic acid With potassium phosphate; 2C2F6NO4S2(1-)*C44H66Cl2N2P2Pd(2+); 1-butyl-methylpyrrolidinium bis(trifluoromethylsulfonyl)amide In water at 65℃; Suzuki-Miyaura reaction; Inert atmosphere; Stage #2: 4-bromo-1,1'-biphenyl In water at 65℃; for 2h; Suzuki-Miyaura reaction; Inert atmosphere; | 99% |
| With potassium phosphate; water In toluene at 100℃; for 1h; Suzuki coupling reaction; | 85% |
| With potassium carbonate In ethanol; water at 70℃; for 14h; Suzuki coupling; | 70% |
-
-
92-66-0
4-bromo-1,1'-biphenyl

-
-
1461-22-9
tributyltin chloride

-
-
51533-89-2
[1,1′-biphenyl]-4-yltributylstannane

| Conditions | Yield |
|---|---|
| Stage #1: 4-bromo-1,1'-biphenyl With n-butyllithium In tetrahydrofuran; hexane at -78℃; for 0.5h; Stage #2: tributyltin chloride In tetrahydrofuran; hexane at -78℃; for 1h; | 99% |
| With n-butyllithium In water | 78% |
-
-
1073-13-8
4,4-dimethylcyclohexenone

-
-
92-66-0
4-bromo-1,1'-biphenyl

-
-
1263002-06-7
4-(4,4-dimethylcyclohexa-1,5-dienyl)biphenyl

| Conditions | Yield |
|---|---|
| Stage #1: 4,4-dimethylcyclohexenone With tris-(dibenzylideneacetone)dipalladium(0); toluene-4-sulfonic acid hydrazide; lithium tert-butoxide; XPhos In 1,4-dioxane for 0.0166667h; Inert atmosphere; Stage #2: 4-bromo-1,1'-biphenyl In 1,4-dioxane at 110℃; Inert atmosphere; | 99% |
-
-
1295-35-8
bis(1,5-cyclooctadiene)nickel (0)

-
-
110-18-9
N,N,N,N,-tetramethylethylenediamine

-
-
92-66-0
4-bromo-1,1'-biphenyl

| Conditions | Yield |
|---|---|
| In toluene at 23℃; for 2h; | 99% |

-
-
590-28-3
potassium cyanate

-
-
201230-82-2
carbon monoxide

-
-
92-66-0
4-bromo-1,1'-biphenyl

-
-
67-63-0
isopropyl alcohol

| Conditions | Yield |
|---|---|
| With dichloro(1,5-cyclooctadiene)palladium(II); 4,5-bis(diphenylphos4,5-bis(diphenylphosphino)-9,9-dimethylxanthenephino)-9,9-dimethylxanthene at 70℃; for 8h; | 99% |

-
-
594-20-7
2,2-dichloropropane

-
-
92-66-0
4-bromo-1,1'-biphenyl

-
-
41081-31-6
dimethyl(4-biphenyl)silyl chloride

| Conditions | Yield |
|---|---|
| Stage #1: 4-bromo-1,1'-biphenyl With n-butyllithium In diethyl ether; hexane at -78 - 20℃; for 3h; Schlenk technique; Glovebox; Stage #2: 2,2-dichloropropane In diethyl ether; hexane at -78 - 20℃; for 15h; Schlenk technique; Glovebox; | 99% |
4-Bromobiphenyl Chemical Properties
Empirical Formula: C12H9Br
Molecular Weight: 233.1039 g/mol
EINECS: 202-176-6
Index of Refraction: 1.6
Density: 1.363 g/cm3
Flash Point: 141.8 °C
Melting point: 82-86 °C(lit.)
Water solubility: Insoluble
Appearance: White powder
Enthalpy of Vaporization: 52.86 kJ/mol
Boiling Point: 309.8 °C at 760 mmHg
Vapour Pressure: 0.00114 mmHg at 25 °C
Structure of 4-Bromobiphenyl (CAS NO.92-66-0):
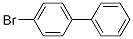
IUPAC Name: 1-Bromo-4-phenylbenzene
Product Category of 4-Bromobiphenyl (CAS NO.92-66-0): Aromatic Compounds;Bromides;Liquid Crystal intermediates;Biphenyl & Diphenyl ether;Alpha Sort;Halogenated;Volatiles/ Semivolatiles
4-Bromobiphenyl Uses
4-Bromobiphenyl (CAS NO.92-66-0) can be used for materials of liquid crystal and intermediates.
4-Bromobiphenyl Toxicity Data With Reference
| 1. | mma-sat 50 µg/plate | BCPCA6 Biochemical Pharmacology. 27 (1978),1245. |
4-Bromobiphenyl (CAS NO.92-66-0) hasn't been listed as a carcinogen by NTP, IARC, ACGIH, or CA Prop 65. And the toxicological properties have not been fully investigated. You can see actual entry in RTECS for complete information.
4-Bromobiphenyl Consensus Reports
Reported in EPA TSCA Inventory.
4-Bromobiphenyl Safety Profile
Mutation data reported. When heated to decomposition it emits toxic vapors of Br−.
Hazard Codes:  Xi
Xi
Risk Statements: 36/37/38
R36/37/38:Irritating to eyes, respiratory system and skin.
Safety Statements: 22-24/25-37/39-26
S22:Do not breathe dust.
S24/25:Avoid contact with skin and eyes.
S26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice.
S37/39:Wear suitable gloves and eye/face protection.
4-Bromobiphenyl Specification
4-Bromobiphenyl , its cas register number is 92-66-0. It also can be called 1,1'-biphenyl, 4-bromo- . It is hazardous, so the first aid measures and others should be known. Such as: When on the skin: first, should flush skin with plenty of water immediately for at least 15 minutes while removing contaminated clothing. Secondly, get medical aid. Or in the eyes: Flush eyes with plenty of water for at least 15 minutes, occasionally lifting the upper and lower eyelids. Then get medical aid soon. While, it's inhaled: Remove from exposure and move to fresh air immediately. Give artificial respiration while not breathing. When breathing is difficult, give oxygen. And as soon as to get medical aid. Then you have the ingesting of the product : If victim is conscious and alert, give 2-4 cupfuls of milk or water. Get medical aid immediately. Notes to physician: Treat supportively and symptomatically.
In addition, 4-Bromobiphenyl (CAS NO.92-66-0) is not compatible with strong oxidizing agents, and you must not take it with incompatible materials. And also prevent it to broken down into hazardous decomposition products: hydrogen chloride, carbon monoxide, carbon dioxide.
Related Products
- 4-Bromobiphenyl
- 4-Bromobiphenyl-4'-boronic acid
- 92660-81-6
- 926-61-4
- 926622-52-8
- 926622-53-9
- 926-62-5
- 926-63-6
- 926642-55-9
- 926-64-7
- 92665-29-7
- 926657-23-0
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View