-
Name
4-Dimethylaminobenzaldehyde
- EINECS 202-819-0
- CAS No. 100-10-7
- Article Data274
- CAS DataBase
- Density 1.072 g/cm3
- Solubility 0.3 g/L (20 °C) in water
- Melting Point 72-75 °C(lit.)
- Formula C9H11NO
- Boiling Point 266.5 °C at 760 mmHg
- Molecular Weight 149.192
- Flash Point 103.3 °C
- Transport Information UN 2920 8/PG 2
- Appearance White to off white crystalline powder
- Safety 7-16-24/25-26-61-45-39-36/37/39-36
- Risk Codes 36/37/38-52/53-22-67-41-37/38-10-66-37-34-20-20/21/22
-
Molecular Structure
-
Hazard Symbols
 C,
C, Xn,
Xn, Xi
Xi
- Synonyms p-Dimethylaminobenzaldehyde;Benzaldehyde,p-(dimethylamino)- (8CI);4-(Dimethylamino)benzaldehyde;4-(Dimethylamino)benzenecarbonal;4-(N,N-Dimethylamino)benzaldehyde;4-Formyl-N,N-dimethylaniline;Erlich reagent;N,N-Dimethyl-4-aminobenzaldehyde;N,N-Dimethyl-4-formylaniline;N,N-Dimethyl-p-aminobenzaldehyde;NSC 5517;p-(Dimethylamino)benzaldehyde;p-(N,N-Dimethylamino)benzaldehyde;p-DAB;p-Formyl-N,N-dimethylaniline;p-Formyldimethylaniline;Benzaldehyde,4-(dimethylamino)-;
- PSA 20.31000
- LogP 1.56510
Synthetic route

-
-
100-10-7
4-dimethylamino-benzaldehyde

| Conditions | Yield |
|---|---|
| With hydrogenchloride for 0.0416667h; Product distribution; Ambient temperature; pH = 4-6, regeneration of aldehyde; | 100% |
-
-
459-57-4
4-fluorobenzaldehyde

-
-
68-12-2, 33513-42-7
N,N-dimethyl-formamide

-
-
100-10-7
4-dimethylamino-benzaldehyde

| Conditions | Yield |
|---|---|
| With PdCl(2-HO-C6H4-CH(Ph)-NH-(CH2)3-SeC6H5); potassium carbonate; phenylboronic acid In water Heating; | 100% |

| Conditions | Yield |
|---|---|
| With silica-supported Jones reagent In dichloromethane for 0.00269444h; | 99.3% |
| With potassium carbonate In water at 90℃; for 3h; | 99% |
| With potassium hydroxide In toluene at 110℃; for 4h; Catalytic behavior; | 99% |
-
-
24588-75-8
4-(N,N'-dimethylamino)phenyl-1,3-dithiane

-
-
100-10-7
4-dimethylamino-benzaldehyde

| Conditions | Yield |
|---|---|
| With bis-[(trifluoroacetoxy)iodo]benzene In methanol; water for 0.166667h; Ambient temperature; | 99% |
| With silica gel In neat (no solvent) at 20℃; for 0.0583333h; | 90% |

| Conditions | Yield |
|---|---|
| Stage #1: 4-(1,3-dioxolan-2-yl)phenyl trifluoromethanesulfonate With potassium phosphate; tris-(dibenzylideneacetone)dipalladium(0); XPhos In tetrahydrofuran at 80℃; for 0.0833333h; Buchwald-Hartwig Coupling; Inert atmosphere; Sealed tube; Stage #2: dimethyl amine In tetrahydrofuran at 80℃; for 16h; Buchwald-Hartwig Coupling; Inert atmosphere; Sealed tube; | 99% |
-
-
2929-84-2, 37961-71-0, 77145-76-7
4-dimethylaminobenzaldehyde oxime

-
-
100-10-7
4-dimethylamino-benzaldehyde

| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid at 20℃; for 0.0166667h; neat (no solvent); | 97% |
| With water; Dess-Martin periodane In dichloromethane at 5℃; for 0.333333h; | 92% |
| With dihydrogen peroxide; tripropylammonium fluorochromate (VI) In acetone at -10℃; for 0.166667h; | 90% |
-
-
201230-82-2
carbon monoxide

-
-
586-77-6
4-bromo-N,N-dimethylaniline

-
-
100-10-7
4-dimethylamino-benzaldehyde

| Conditions | Yield |
|---|---|
| With N,N,N,N,-tetramethylethylenediamine; hydrogen; palladium diacetate; propyl di-tert-butylphosphinite In toluene at 100℃; under 3750.38 Torr; for 20h; Inert atmosphere; | 95% |
| With Cr(CO)3; hydrogen; triethylamine; triphenylphosphine; bis-triphenylphosphine-palladium(II) chloride In toluene at 130℃; | 35% |
| With 4-methoxy-N'-tetramethylethylenediamine; hydrogen; catacxium A; palladium diacetate In toluene at 100℃; under 3750.3 Torr; for 16h; | 98 % Chromat. |
| With N,N,N,N,-tetramethylethylenediamine; hydrogen; catacxium A; palladium diacetate In toluene at 100℃; under 3750.38 Torr; for 16h; | 98 % Chromat. |
| With N,N,N,N,-tetramethylethylenediamine; hydrogen; palladium diacetate; catacxium A In toluene at 20 - 100℃; under 3750.38 Torr; Autoclave; | 99 %Chromat. |
-
-
86459-85-0
4-(N,N-dimethylamino)benzaldehyde dimethyl acetal

-
-
100-10-7
4-dimethylamino-benzaldehyde

| Conditions | Yield |
|---|---|
| With iron(III) p-toluenesulfonate hexahydrate In water at 20℃; for 0.75h; | 94% |
| bismuth(III) iodide In water at 20℃; for 2h; | 85% |
| With copper(II) sulfate; sodium iodide In acetone at 20℃; for 3h; | 82% |
-
-
92310-70-8
4-formylphenyl N,N-dimethylcarbamate

-
-
100-10-7
4-dimethylamino-benzaldehyde

| Conditions | Yield |
|---|---|
| With bis(1,5-cyclooctadiene)nickel (0) In toluene at 160℃; for 14h; Inert atmosphere; Sealed tube; Glovebox; | 93% |
-
-
31362-12-6
p-(1,3-dithiolan-2-yl)-N,N-dimethylaniline

-
-
100-10-7
4-dimethylamino-benzaldehyde

| Conditions | Yield |
|---|---|
| With silica gel In neat (no solvent) at 20℃; for 0.0666667h; | 92% |
| With boron trifluoride diethyl etherate; water; mercury(II) oxide In tetrahydrofuran for 1h; Ambient temperature; | 90% |
| With Oxone; potassium bromide In water; acetonitrile at 20℃; for 0.333333h; | 82% |

| Conditions | Yield |
|---|---|
| With hexaaquairon(III) perchlorate for 2h; | 91% |

| Conditions | Yield |
|---|---|
| With pyrrolidine; water In acetonitrile at 20℃; for 12h; Inert atmosphere; | 91% |
| With morpholine; palladium 10% on activated carbon; oxygen; copper(l) chloride In isopropyl alcohol at 100℃; for 24h; regioselective reaction; | 87% |
-
-
121-69-7
N,N-dimethyl-aniline

-
-
68-12-2, 33513-42-7
N,N-dimethyl-formamide

-
-
100-10-7
4-dimethylamino-benzaldehyde

| Conditions | Yield |
|---|---|
| With bis(trichloromethyl) carbonate In tetrachloromethane at 40 - 50℃; for 2h; Vilsmeier-Haack formylation; | 90% |
| With trichlorophosphate for 0.0833333h; Vilsmeier-Haack reaction; Microwave irradiation; | 80% |
| With silica gel; trichlorophosphate for 0.025h; Formylation; Microwave irradiation (300 W); | 78% |

| Conditions | Yield |
|---|---|
| 90% | |
| formylation; | 90% |
-
-
1369488-99-2
4-(azidomethyl)-N,N-dimethylaniline

-
-
100-10-7
4-dimethylamino-benzaldehyde

| Conditions | Yield |
|---|---|
| With iron(III) chloride; dihydrogen peroxide In dichloromethane; water for 14h; Reflux; Air; | 90% |
| Stage #1: 4-(azidomethyl)-N,N-dimethylaniline With sodium hydride In dimethyl sulfoxide; mineral oil at 20℃; for 4h; Inert atmosphere; Stage #2: With water In dimethyl sulfoxide; mineral oil for 0.25h; Inert atmosphere; | 80% |
-
-
17087-90-0, 149742-33-6
N-(4-dimethylaminobenzylidene)-p-toluidine

-
-
100-10-7
4-dimethylamino-benzaldehyde

| Conditions | Yield |
|---|---|
| With tribromo-isocyanuric acid In acetonitrile for 1h; Reflux; | 90% |
-
-
15795-57-0
5-(4-N,N-dimethylaminobenzylidene)-2,2-dimethyl-1,3-dioxane-4,6-dione

-
-
100-10-7
4-dimethylamino-benzaldehyde

| Conditions | Yield |
|---|---|
| With oxone In water; acetonitrile at 45℃; for 1h; | 89% |
-
-
889-37-2, 1613-99-6
N,N-dimethyl-4-((phenylimino)methyl)aniline

-
-
100-10-7
4-dimethylamino-benzaldehyde

| Conditions | Yield |
|---|---|
| With hexaaquairon(III) perchlorate for 2h; | 88% |
-
-
140111-19-9
Ru3(CO)10H(OCC6H4N(CH3)2)

-
A
-
15243-33-1
dodecacarbonyl-triangulo-triruthenium

-
B
-
100-10-7
4-dimethylamino-benzaldehyde

| Conditions | Yield |
|---|---|
| With CO In benzene High Pressure; heating (3 bar CO, 90 min, 80°C); chromy. (hexane); | A 88% B 81% |

-
-
100-10-7
4-dimethylamino-benzaldehyde

| Conditions | Yield |
|---|---|
| With sodium tetrahydroborate; nickel dichloride In methanol at -10 - 10℃; | 87% |

| Conditions | Yield |
|---|---|
| With iodine; triethylamine; triphenylphosphine In toluene at 80℃; Inert atmosphere; Sealed tube; | 87% |

| Conditions | Yield |
|---|---|
| With hexaaquairon(III) perchlorate for 2h; | 86% |

-
-
100-10-7
4-dimethylamino-benzaldehyde

| Conditions | Yield |
|---|---|
| With potassium hydrogencarbonate; dimethyl sulfoxide for 0.0583333h; Microwave irradiation; | 86% |
-
-
3724-43-4, 149409-22-3
Vilsmeier reagent

-
-
121-69-7
N,N-dimethyl-aniline

-
-
100-10-7
4-dimethylamino-benzaldehyde

| Conditions | Yield |
|---|---|
| Stage #1: Vilsmeier reagent; N,N-dimethyl-aniline In N,N-dimethyl-formamide at 70℃; for 1h; Stage #2: With water; sodium carbonate In N,N-dimethyl-formamide | 84% |
| und anschliessend mit Wasser; |
-
-
117227-36-8
4-{[1-(4-Dimethylamino-phenyl)-meth-(E)-ylidene]-amino}-3,6,6-trimethyl-4,6-dihydro-1H-cyclopenta[1,2,4]triazine-5,7-dicarboxylic acid dimethyl ester

-
A
-
100-10-7
4-dimethylamino-benzaldehyde

-
B
-
1197-19-9
4-cyano-N,N-dimethylaniline

| Conditions | Yield |
|---|---|
| With sodium methylate In ethanol for 3h; Product distribution; Heating; | A 12% B 84% |

| Conditions | Yield |
|---|---|
| With hexaaquairon(III) perchlorate for 2h; | 84% |
| With sodium perborate In acetic acid at 40℃; for 1.5h; Oxidation; |
-
-
17078-28-3
2-[4-(dimethylamino)phenyl]acetic acid

-
-
100-10-7
4-dimethylamino-benzaldehyde

| Conditions | Yield |
|---|---|
| With mercury(II) fluoride; oxygen In acetonitrile at 25℃; for 24h; Irradiation; | 84% |
-
-
6425-24-7
1-(dibromomethyl)-4-fluorobenzene

-
-
124-40-3
dimethyl amine

-
-
100-10-7
4-dimethylamino-benzaldehyde

| Conditions | Yield |
|---|---|
| With water at 60℃; for 2h; | 82% |
-
-
42060-28-6
N-(4-methylbenzyl)methanesulfonamide

-
-
100-10-7
4-dimethylamino-benzaldehyde

| Conditions | Yield |
|---|---|
| With MS3 Angstroem; oxygen; sodium acetate; triphenylphosphine; palladium dichloride In N,N-dimethyl-formamide at 80℃; under 760 Torr; for 14h; | 82% |
-
-
67-52-7
BARBITURIC ACID

-
-
100-10-7
4-dimethylamino-benzaldehyde

-
-
1753-47-5
5-(4-dimethylaminobenzylidene)barbituric acid

| Conditions | Yield |
|---|---|
| In methanol Knoevenagel Condensation; Reflux; | 100% |
| In methanol at 20℃; Knoevenagel condensation; | 98% |
| With 1-butyl-3-methylimidazolium Tetrafluoroborate at 20℃; for 0.166667h; Knoevenagel condensation; | 98% |
-
-
5217-47-0
N,N'-diethyl-2-thiobarbituric acid

-
-
100-10-7
4-dimethylamino-benzaldehyde

-
-
86872-78-8
5-(4-(dimethylamino)benzylidene)-1,3-diethyl-2-thioxodihydropyrimidine-4,6(1H,5H)-dione

| Conditions | Yield |
|---|---|
| at 20℃; for 1h; Knoevenagel condensation; | 100% |
| In water for 0.5h; Reflux; | 93% |
| In ethanol at 20℃; Condensation; Knoevenagel condensation; | 87% |

-
-
65786-13-2, 6342-97-8
2'-hydroxy-4-dimethylaminochalcone

| Conditions | Yield |
|---|---|
| Stage #1: o-hydroxyacetophenone With sodium hydroxide In ethanol; water at 0 - 5℃; Stage #2: 4-dimethylamino-benzaldehyde In ethanol; water | 100% |
| With potassium hydroxide In ethanol at 25℃; | 85% |
| With potassium hydroxide In ethanol at 25℃; | 85% |
-
-
105-56-6
ethyl 2-cyanoacetate

-
-
100-10-7
4-dimethylamino-benzaldehyde

-
-
1886-52-8, 74897-86-2, 14394-77-5
ethyl (E)-2-cyano-3-[4-(dimethylamino)phenyl]prop-2-enoate

| Conditions | Yield |
|---|---|
| With third generation polystyrene supported poly(amidoamine) dendrimer In ethanol at 50℃; for 0.25h; Knoevenagel condensation; | 100% |
| With diazabicyclo[5.4.0]undec-7-ene-water complex at 20℃; for 1h; Knoevenagel condensation; | 100% |
| With polyacrylonitrile fiber functionalized with N,N-dimethyl-1,3-propanediamine In ethanol for 1.5h; Knoevenagel condensation; Reflux; | 99% |
-
-
139-65-1
4,4'-thiobisaniline

-
-
100-10-7
4-dimethylamino-benzaldehyde

-
-
3430-66-8
4,4'-(((thiobis(4,1-phenylene))bis(azanylylidene))bis(methanylylidene))bis(N,N-dimethylaniline)

| Conditions | Yield |
|---|---|
| With piperidine In ethanol | 100% |
| With acetic acid In ethanol for 4h; Reflux; | 85% |
| With ethanol; zinc(II) chloride |

| Conditions | Yield |
|---|---|
| In diethyl ether for 1h; Ambient temperature; | 100% |
-
-
100-10-7
4-dimethylamino-benzaldehyde

-
-
67-64-1
acetone

-
-
5432-53-1
4-p-dimethylaminophenyl-3-buten-2-one

| Conditions | Yield |
|---|---|
| With sodium hydroxide for 72h; Ambient temperature; | 100% |
| With water; potassium hydroxide at 20℃; for 0.2h; | 82% |
| With sodium hydroxide In water at 0 - 20℃; Aldol condensation; | 67.73% |
-
-
100-10-7
4-dimethylamino-benzaldehyde

-
-
74-89-5
methylamine

-
-
877-79-2
4-(dimethylamino)-N-methylbenzylimine

| Conditions | Yield |
|---|---|
| at 20℃; under 750.06 Torr; Solid phase reaction; gas-solid reaction; | 100% |
| at 20℃; for 12h; | 100% |
| With benzene |
-
-
100-10-7
4-dimethylamino-benzaldehyde

-
-
109-77-3
malononitrile

-
-
2826-28-0
p-(N-dimethylamino benzylidene) malononitrile

| Conditions | Yield |
|---|---|
| With third generation polystyrene supported poly(amidoamine) dendrimer In ethanol at 30℃; for 0.333333h; Knoevenagel condensation; | 100% |
| With polymer supported poly(propylene imine)dendrimer In ethanol at 20℃; for 0.0833333h; Knoevenagel Condensation; Green chemistry; | 100% |
| With potassium hydrogen phthalate In water at 20℃; for 0.116667h; Knoevenagel Condensation; Green chemistry; | 99% |
-
-
100-10-7
4-dimethylamino-benzaldehyde

-
-
105-34-0
methyl 2-cyanoacetate

-
-
3785-86-2, 13432-69-4
methyl (E)-2-cyano-3-(4-(N,N-dimethylamino)phenyl)-2-propenoate

| Conditions | Yield |
|---|---|
| at 170℃; for 1h; Knoevenagel condensation; | 100% |
| With silica sodium carbonate nanoparticles In acetonitrile at 70℃; Knoevenagel Condensation; Sonication; Green chemistry; | 95% |
| With tetra(n-butyl)ammonium hydroxide In ethanol; water at 20℃; for 0.166667h; Knoevenagel condensation; | 94% |
-
-
100-10-7
4-dimethylamino-benzaldehyde

-
-
2929-84-2, 37961-71-0, 77145-76-7
4-dimethylaminobenzaldehyde oxime

| Conditions | Yield |
|---|---|
| With hydroxylamine hydrochloride; sodium acetate In acetonitrile for 4h; Reflux; | 100% |
| With sodium hydroxide; hydroxylamine hydrochloride at 20℃; for 0.5h; grinding; | 98% |
| With 3 A molecular sieve; hydroxylamine hydrochloride; sodium acetate In ethanol for 0.25h; | 94% |

| Conditions | Yield |
|---|---|
| With 1-methyl-pyrrolidin-2-one; hydroxylamine hydrochloride at 100℃; for 0.25h; Condensation; microwave irradiation; | 100% |
| With ammonia; iodine In water; N,N-dimethyl-formamide at 20℃; for 0.666667h; | 99% |
| With sodium azide; trifluorormethanesulfonic acid In acetonitrile at 20℃; for 0.0333333h; Schmidt reaction; | 98% |
-
-
26104-68-7
(p-cyanobenzyl)triphenylphosphonium bromide

-
-
100-10-7
4-dimethylamino-benzaldehyde

-
-
2844-17-9
4-(dimethylamino)-4'-cyanostilbene

| Conditions | Yield |
|---|---|
| With potassium tert-butylate In methanol for 24h; Heating; | 100% |
| (i) LiOMe, MeOH, (ii) I2, xylene; Multistep reaction; |
-
-
100-10-7
4-dimethylamino-benzaldehyde

-
-
107-91-5
cyanoacetic acid amide

-
-
97006-42-3
(E)-2-cyano-3-[4-(dimethylamino)phenyl]-2-propenamide

| Conditions | Yield |
|---|---|
| With triethylamine at 20℃; for 24h; Knoevenagel condensation; | 100% |
| With tetra(n-butyl)ammonium hydroxide In ethanol; water at 20℃; for 0.166667h; Knoevenagel condensation; | 94% |
| With Tonsil Actisil FF at 80℃; for 0.333333h; Irradiation; | 55% |
| With potassium-exchanged zirconium hydrogen phosphate at 100℃; for 4h; Knoevenagel condensation; | 49% |
| With diethylamine |
-
-
110-91-8
morpholine

-
-
100-10-7
4-dimethylamino-benzaldehyde

-
-
87294-98-2
(4-(dimethylamino)phenyl)(morpholino)methanone

| Conditions | Yield |
|---|---|
| With bromobenzene; potassium carbonate; triphenylphosphine; palladium diacetate In 1,2-dimethoxyethane for 24h; Heating; | 100% |
-
-
110-91-8
morpholine

-
-
100-10-7
4-dimethylamino-benzaldehyde

-
-
5925-53-1
4-<4-(Dimethylamino)thiobenzoyl>morpholine

| Conditions | Yield |
|---|---|
| With sulfur for 0.0666667h; Willgerodt-Kindler reaction; microwave irradiation; | 100% |
| With 4-methyl-morpholine; sulfur In N,N-dimethyl-formamide at 135℃; for 6h; Willgerodt-Kindler reaction; | 92% |
| With sulfur In dimethyl sulfoxide at 20℃; for 16h; | 89% |
-
-
3949-36-8
3-acetylcoumarin

-
-
100-10-7
4-dimethylamino-benzaldehyde

-
-
91527-77-4
1-(3'-coumarinyl)-3-(4''-dimethylaminophenyl)-2-propen-1-one

| Conditions | Yield |
|---|---|
| With potassium hydroxide In ethanol Reflux; | 100% |
| With piperidine In chloroform for 7h; Heating; | 97% |
| With piperidine at 45 - 50℃; for 0.5h; | 91% |
-
-
108-91-8
cyclohexylamine

-
-
100-10-7
4-dimethylamino-benzaldehyde

-
-
31235-64-0
N-<<4-(dimethylamino)phenyl>methylene>cyclohexylamine

| Conditions | Yield |
|---|---|
| With sodium sulfate In methanol for 4h; Ambient temperature; | 100% |
-
-
107-10-8
propylamine

-
-
100-10-7
4-dimethylamino-benzaldehyde

-
-
59488-01-6
p-Dimethylamino-N-n-propyl-iminomethylbenzol

| Conditions | Yield |
|---|---|
| at 20℃; for 12h; | 100% |
| In methanol at 25℃; Mechanism; Rate constant; Thermodynamic data; ΔH(excit.), ΔS(excit.); kinetic solvent isotope effect; |
-
-
21209-71-2
2-(hydroxyimino)propionic acid

-
-
100-10-7
4-dimethylamino-benzaldehyde

-
-
93563-01-0
1-Carboxy-N-(4-dimethylaminophenylmethylen)ethylamin-N-oxid

| Conditions | Yield |
|---|---|
| In ethanol Heating; | 100% |
-
-
52-90-4
L-Cysteine

-
-
100-10-7
4-dimethylamino-benzaldehyde

-
-
222404-26-4
(2RS,4R)-2-(4-dimethyloamino-phenyl)-thiazolidine-4-carboxylic acid

| Conditions | Yield |
|---|---|
| In ethanol at 20℃; for 5h; | 100% |
| In methanol; water | 84% |
| In ethanol; water at 20℃; |

| Conditions | Yield |
|---|---|
| With 4 A molecular sieve In dichloromethane at 20℃; for 70h; | 100% |

-
-
100-10-7
4-dimethylamino-benzaldehyde

| Conditions | Yield |
|---|---|
| In acetic anhydride at 120℃; for 0.5h; | 100% |
-
-
100-10-7
4-dimethylamino-benzaldehyde

-
-
67-64-1
acetone

-
-
30625-58-2
4-(4-N,N-dimethylaminophenyl)-3-buten-2-one

| Conditions | Yield |
|---|---|
| With sodium hydroxide In water at 0 - 25℃; for 3h; | 100% |
| With sodium hydroxide In water at 5 - 10℃; for 0.583333h; | 98% |
| With sodium hydroxide for 2.5h; Ambient temperature; | 96% |
-
-
75-31-0
isopropylamine

-
-
100-10-7
4-dimethylamino-benzaldehyde

-
-
27976-83-6
<4-Dimethylamino-benzyliden>-isopropylamin

| Conditions | Yield |
|---|---|
| at 20℃; for 12h; | 100% |
| at 20℃; for 48h; | 79% |
-
-
26138-64-7
3-acetyl-2,4-dihydroxyquinoline

-
-
100-10-7
4-dimethylamino-benzaldehyde

| Conditions | Yield |
|---|---|
| 100% |

| Conditions | Yield |
|---|---|
| With phosphoric acid In water; isopropyl alcohol for 6h; Heating; | 100% |
-
-
100-10-7
4-dimethylamino-benzaldehyde

-
-
146476-37-1
(2S)-1-(diphenylphosphino)-3-methylbutan-2-amine

| Conditions | Yield |
|---|---|
| In toluene Ambient temperature; | 100% |
| In toluene at 20℃; Substitution; |
-
-
6332-77-0
N-(4-nitrophenyl)ethylenediamine

-
-
100-10-7
4-dimethylamino-benzaldehyde

-
-
250386-77-7
N-[1-(4-Dimethylamino-phenyl)-meth-(E)-ylidene]-N'-(4-nitro-phenyl)-ethane-1,2-diamine

| Conditions | Yield |
|---|---|
| In methanol at 25℃; for 1h; Condensation; | 100% |
4-Dimethylaminobenzaldehyde Consensus Reports
4-Dimethylaminobenzaldehyde Specification
The 4-Dimethylaminobenzaldehyde is also known as Benzaldehyde,4-(dimethylamino)-. The IUPAC name is 4-(Dimethylamino)benzaldehyde. It belongs to product categories of Intermediates of Dyes and Pigments; Aromatic Aldehydes & Derivatives (substituted); Benzaldehyde; Exciton Chirality CD Method (for Primary Amino Groups); Absolute Configuration Determination (Exciton Chirality CD Method); Analytical Chemistry; Enantiomer Excess & Absolute Configuration Determination. This chemical is a white to off white crystalline powder and soluble in alcohol, ether, acetone, chloroform and acetic acid, slightly soluble in water. What's more, it should be sealed in ventilated and cool place away from fire, heat, oxidants, alkali without air.
Physical properties about 4-Dimethylaminobenzaldehyde are: (1)ACD/LogP: 1.81; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): 1.81; (4)ACD/LogD (pH 7.4): 1.81; (5)ACD/BCF (pH 5.5): 13.98; (6)ACD/BCF (pH 7.4): 13.98; (7)ACD/KOC (pH 5.5): 229.91; (8)ACD/KOC (pH 7.4): 229.95; (9)#H bond acceptors: 2; (10)#Freely Rotating Bonds: 2; (11)Index of Refraction: 1.595; (12)Molar Refractivity: 47.31 cm3; (13)Molar Volume: 139 cm3; (14)Surface Tension: 42.1 dyne/cm; (15)Density: 1.072 g/cm3; (16)Flash Point: 103.3 °C; (17)Enthalpy of Vaporization: 50.45 kJ/mol; (18)Boiling Point: 266.5 °C at 760 mmHg; (19)Vapour Pressure: 0.00861 mmHg at 25 °C.
Preparation of 4-Dimethylaminobenzaldehyde: 4-Dimethylaminobenzaldehyde is prepared by reaction of 4-bromo-N,N-dimethyl-aniline with carbon monoxide. This reaction needs reagents Cr(CO)3, PPh3, Et3N, H2, catalyst Pd(PPh3)2Cl2 and solvent toluene at the temperature of 130 °C. The yield is about 35%.

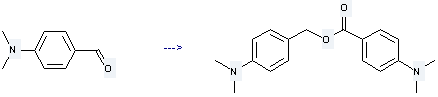
Uses of 4-Dimethylaminobenzaldehyde: 4-Dimethylaminobenzaldehyde is used as a dye intermediate. Besides, it can be used as a reagent for determining urinary choline, indole, alkaloids and also can be used as chromatography reagent. Furthermore, it is used to produce 4-dimethylamino-benzoic acid 4-dimethylamino-benzyl ester. The reaction occurs with reagents Na, biphenyl and solvents benzene, hexane with other condition of heating for 12 hours. The yield is about 86%.
When you are using 4-Dimethylaminobenzaldehyde, please be cautious about it. As a chemical, it is harmful by inhalation, in contact with skin and if swallowed. There is risk of serious damage to eyes. 4-Dimethylaminobenzaldehyde is also harmful to aquatic organisms that may cause long-term adverse effects in the aquatic environment and repeated exposure may cause skin dryness or cracking. As it is flammable, it causes burns and its vapours may cause drowsiness and dizziness. During using it, wear suitable protective clothing, gloves and eye/face protection. Avoid contact with skin and eyes and release to the environment. If contact with eyes accidently, rinse immediately with plenty of water and seek medical advice. In case of accident or if you feel unwell seek medical advice immediately. After using it, keep container tightly closed away from sources of ignition.
You can still convert the following datas into molecular structure:
1) Canonical SMILES: CN(C)C1=CC=C(C=C1)C=O
2) InChI: InChI=1S/C9H11NO/c1-10(2)9-5-3-8(7-11)4-6-9/h3-7H,1-2H3
3) InChIKey: BGNGWHSBYQYVRX-UHFFFAOYSA-N
The toxicity data is as follows:
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| mouse | LD50 | intraperitoneal | 200mg/kg (200mg/kg) | BEHAVIORAL: GENERAL ANESTHETIC BEHAVIORAL: ATAXIA BEHAVIORAL: SOMNOLENCE (GENERAL DEPRESSED ACTIVITY) | National Technical Information Service. Vol. OTS0533441, |
| mouse | LD50 | oral | 800mg/kg (800mg/kg) | BEHAVIORAL: ATAXIA BEHAVIORAL: GENERAL ANESTHETIC BEHAVIORAL: SOMNOLENCE (GENERAL DEPRESSED ACTIVITY) | National Technical Information Service. Vol. OTS0533441, |
| rat | LD50 | intraperitoneal | 620mg/kg (620mg/kg) | Hine Laboratories Reports. Vol. AF33(657)-11756, Pg. 1964, | |
| rat | LDLo | oral | 500mg/kg (500mg/kg) | Journal of Pharmacology and Experimental Therapeutics. Vol. 90, Pg. 260, 1947. |
Related Products
- 4-Dimethylaminobenzaldehyde
- 10010-93-2
- 1001095-14-2
- 100111-07-7
- 100114-24-7
- 100114-41-8
- 100114-57-6
- 100114-58-7
- 100-11-8
- 1001183-84-1
- 1001200-61-8
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View