-
Name
CHLORINE TRIFLUORIDE
- EINECS 232-230-4
- CAS No. 7790-91-2
- Density 3.18 (Relative vapour density (air = 1))
- Solubility reaction in water
- Melting Point -76ºC
- Formula ClF3
- Boiling Point 12ºC
- Molecular Weight 92.4482
- Flash Point
- Transport Information
- Appearance NEARLY COLOURLESS COMPRESSED LIQUEFIED GAS , WITH CHARACTERISTIC ODOUR.
-
Safety
Human poison by inhalation. An eye irritant. See also FLUORIDES, CHLORINE, and FLUORINE. Spontaneously flammable. A powerful oxidant which may react violently with oxidizable materials. A rocket propellant.
Explosive reaction with water, bis(trifluoro methyl)sulfide or -disulfide, polychlorotrifluoroethylene, trifluoromethanesulfenyl chloride, and other hydrogen- containing materials (e.g., ammonia, coal gas, hydrogen, hydrogen sulfide, methane, acetic acid, benzene, ether, cotton, paper, wood). Forms shock-sensitive explosive mixtures with highly chlorinated compounds (e.g., carbon tetrachloride), nitroaryl compounds (e.g., trinitrotoluene, hexanitrobiphenyl, hexanitrodiphenyl amine, hexanitro diphenyl sulfide, hexanitrodiphenyl ether). Reaction with ammonium fluoride or ammonium hydrogen fluoride forms explosive gaseous products.
Ignition on contact with boron-containing materials, iodine, finely divided refractory materials (e.g., asbestos, glass wool, sand, tungsten carbide), fluorinated polymers (with flowing trifluoride).
Violent reaction with acids (e.g., nitric or sulfuric), chromium trioxide, ruthenium, selenium tetrafluoride (above 106°C), metals, metal oxides, metal salts, nonmetals, nonmetal salts, organic matter, glass wool, acetic acid, Al, Sb, As, Cu, Ir, Fe, Pb, Mg, Mo, Os, P, K, Rh, Se, Si, Ag, Na, S, Te, Sn, W, Zn, oxides, CO, graphite, HgI2, HNO3, K2CO3, KI, rubber, AgN3, AgNO3, NaOH, V2P5, WO3. Incompatible with fuels, nitro compounds. When heated to decomposition or in reaction with water or steam it emits toxic fumes of F− and Cl−.
- Risk Codes 8-35
-
Molecular Structure
- Hazard Symbols Explodes in contact with organic materials or with water. Dangerous fire risk. A poison, very toxic, corrosive to skin. TLV: ceiling 0.1 ppm.
- Synonyms Chlorinefluoride (Cl2F6); Chlorine trifluoride; Chlorine trifluoride (ClF3);Chlorotrifluoride; Trifluorochlorine
- PSA 0.00000
- LogP 1.95010
Synthetic route

| Conditions | Yield |
|---|---|
| In gas at 119 - 232°C in Al-tubes about 30 - 45 min;; | A 95% B n/a |
| In gas at 170°C in Ni-tubes about 40 min;; | A 12% B n/a |
| nickel(II) fluoride In gas Kinetics; at 467, 497 and 530°K in Ni-tubes;; |


| Conditions | Yield |
|---|---|
| In neat (no solvent) equilibrium react.;; | |
| In neat (no solvent) Kinetics; equilibrium at 500 - 800°K;; | |
| In neat (no solvent) Kinetics; equilibrium at 100 - 380°C;; |

| Conditions | Yield |
|---|---|
| In neat (no solvent) heated (nickel labyrinth crucible, dry N2 atm., heating rate 4-6 K/min.); |
-
-
19195-69-8
potassium tetrafluoridochlorate(III)

-
B
-
7790-91-2
chlorine trifluoride

| Conditions | Yield |
|---|---|
| In neat (no solvent) heated (nickel labyrinth crucible, dry N2 atm., heating rate 4-6 K/min.); |

| Conditions | Yield |
|---|---|
| In neat (no solvent) heating S2Cl2 and NaF under refluxat 136°C; hydrolysis at -120°C;; | 20-45 |

-
-
184851-42-1, 478167-78-1, 13637-63-3, 676353-68-7, 478167-76-9
chlorine pentafluoride

-
A
-
7790-91-2
chlorine trifluoride

-
B
-
7782-41-4
fluorine

| Conditions | Yield |
|---|---|
| In neat (no solvent) equilibrium;; | |
| In neat (no solvent) equilibrium between 211 - 271°K;; | |
| In neat (no solvent) equilibrium between 211 - 271°K;; | |
| In neat (no solvent) equilibrium;; |
-
-
38785-48-7
ClF2 bismuth hexafluoride

-
A
-
7787-62-4
bismuth pentafluoride

-
B
-
7790-91-2
chlorine trifluoride

| Conditions | Yield |
|---|---|
| In neat (no solvent) dry air atmosphere; heating (90-150°C, 4-6 K/min); TGA; |

| Conditions | Yield |
|---|---|
| With sodium fluoride In melt placing the fluorides in a Monel cylinder with a Ni-dip tube and outlet for gaseous phase (3 cooper traps, temp. from dry ice to room temp.); protecting from atmosphere; heating with electric furnace to 725°C; streaming Cl2 through melt for 5h; detecting ClF along with ClF3 by IR spectroscopy; |

-
-
13773-81-4
krypton difluoride

-
A
-
7790-91-2
chlorine trifluoride

-
-
184851-42-1, 478167-78-1, 13637-63-3, 676353-68-7, 478167-76-9
chlorine pentafluoride

| Conditions | Yield |
|---|---|
| With polyethylene observed by IR and raman spectroscopy of KrF2;; |


| Conditions | Yield |
|---|---|
| other Radiation; α-radiation; | |
| other Radiation; α-radiation; |

| Conditions | Yield |
|---|---|
| With chlorine In neat (no solvent) Kinetics; addn. of Cl2 to - 130°C cooled O2F2;; |

| Conditions | Yield |
|---|---|
| In neat (no solvent) above 140°C;; | |
| In neat (no solvent) above 140°C;; |
-
-
7783-44-0
dioxygen difluoride

-
-
7790-89-8
chlorine monofluoride

-
A
-
7790-91-2
chlorine trifluoride

-
B
-
80937-33-3
oxygen

| Conditions | Yield |
|---|---|
| In neat (no solvent) at 140°K;; | |
| In neat (no solvent) at 140°K;; |

-
-
7783-44-0
dioxygen difluoride

-
-
7790-89-8
chlorine monofluoride

-
A
-
7790-91-2
chlorine trifluoride

-
B
-
80937-33-3
oxygen

-
C
-
7782-41-4
fluorine

| Conditions | Yield |
|---|---|
| In neat (no solvent) above 140°K;; |

-
-
14884-42-5
chromium pentafluoride

-
-
7782-50-5
chlorine

-
A
-
7788-97-8
chromium(III) fluoride

-
B
-
7790-91-2
chlorine trifluoride

-
C
-
10049-11-3
chromium tetrafluoride

-
D
-
7790-89-8
chlorine monofluoride

| Conditions | Yield |
|---|---|
| heating at 185°C for 20 h; |

-
-
75-69-4
trichlorofluoromethane

-
-
7782-41-4
fluorine

-
A
-
7790-91-2
chlorine trifluoride

-
B
-
75-72-9
chlorotrifluoromethane

-
C
-
75-71-8
Dichlorodifluoromethane

| Conditions | Yield |
|---|---|
| In neat (no solvent) Kinetics; reaction in a pyrex vessel and He; mechanism discussed;; |


| Conditions | Yield |
|---|---|
| In neat (no solvent) at 270 - 280°C;; | |
| In gas in nickel autoclave for 3 h at 280°C; strictly stoich. quantities; low-temp. distillation in vac.; DTA; chromy.; | |
| In neat (no solvent) apparatus described;; |
-
-
7782-50-5
chlorine

-
-
7782-41-4
fluorine

-
A
-
7790-91-2
chlorine trifluoride

-
B
-
7790-89-8
chlorine monofluoride

| Conditions | Yield |
|---|---|
| In neat (no solvent) reaction of Cl2 and F2 (1:3) at 300°C; apparatus described;; distillation;; | |
| In neat (no solvent) reaction of Cl2 and F2 (1:3) at 300°C; apparatus described;; distillation;; | |
| In neat (no solvent) reaction of Cl2 and F2 in Cu pig at 200°C;; fractionated distillation: at -70°C ClF3, at -150°C ClF; purification of ClF3 in vacuum at 20 Torr;; |


| Conditions | Yield |
|---|---|
| Kinetics; Irradiation (UV/VIS); cylindrical silica cell; λ = 260-370 nm, 293 K; spectrophotometry; |
-
-
14104-20-2
silver tetrafluoroborate

-
A
-
7790-91-2
chlorine trifluoride

-
B
-
7790-89-8
chlorine monofluoride

| Conditions | Yield |
|---|---|
| With chlorine In neat (no solvent) byproducts: AgF, AgCl, BF3; packing AgBF4 in a copper tube connected to traps and on-line IR cell; evacuating; passing Cl2 through AgBF4; heating to 150°C (or 200 or 250°C) for 3h; detecting ClF and ClF3 by IR spectroscopy; |
-
-
7783-44-0
dioxygen difluoride

-
-
7790-89-8
chlorine monofluoride

-
A
-
7790-91-2
chlorine trifluoride

-
-
12133-60-7, 38680-84-1
chlorine dioxide trifluoride

-
C
-
80937-33-3
oxygen

-
D
-
7782-41-4
fluorine

| Conditions | Yield |
|---|---|
| In neat (no solvent) distillation of O2F2 into boron silicate glass in vacuum; cooling at 90°K; addn. of equimolar amount ClF at 119 - 140°K; cooling at 90°K; evacuation of F2 and Cl2;; |


| Conditions | Yield |
|---|---|
| In gas | |
| In neat (no solvent, gas phase) |


| Conditions | Yield |
|---|---|
| In gas | |
| In neat (no solvent, gas phase) |


| Conditions | Yield |
|---|---|
| In neat (no solvent) | |
| In neat (no solvent) |


| Conditions | Yield |
|---|---|
| In neat (no solvent) in 10 min;; | A n/a B >90 |
| In neat (no solvent) in 10 min;; | A n/a B >90 |


| Conditions | Yield |
|---|---|
| In neat (no solvent) | |
| In neat (no solvent) |

-
-
12133-60-7, 38680-84-1
chlorine dioxide trifluoride

-
A
-
7790-91-2
chlorine trifluoride

-
B
-
80937-33-3
oxygen

| Conditions | Yield |
|---|---|
| In neat (no solvent) dissociation above 140°K;; |
-
-
421-17-0
Trifluoromethylsulfenyl chloride

-
-
7790-91-2
chlorine trifluoride

-
-
374-10-7
trifluoro(trifluoromethyl)sulfur(IV)

| Conditions | Yield |
|---|---|
| In dichloromethane -78°C; | 95% |
| In dichloromethane -78°C; | 95% |
| In further solvent(s) in CF2Cl2 at -196 to -78°C; | |
| In further solvent(s) in CF2Cl2 at -196 to -78°C; |
-
-
7790-91-2
chlorine trifluoride

-
-
372-64-5
Bis(trifluoromethyl)disulfid

-
-
374-10-7
trifluoro(trifluoromethyl)sulfur(IV)

| Conditions | Yield |
|---|---|
| In dichloromethane -78°C; | 95% |
| In dichloromethane -78°C; | 95% |
| In Dichlorodifluoromethane excess ClF3, -196 to -78°C; | |
| In Dichlorodifluoromethane excess ClF3, -196 to -78°C; |
-
-
7790-91-2
chlorine trifluoride

-
-
7789-20-0
water-d2

-
A
-
80937-33-3
oxygen

-
B
-
7782-50-5
chlorine

-
C
-
14333-26-7
hydrogen fluoride

| Conditions | Yield |
|---|---|
| In neat (no solvent) ClF3 was delivered to D2O through a nickel capillary (reactor shouldl becooled), DF was condensed to container at 77 K; | A n/a B n/a C 90% |


| Conditions | Yield |
|---|---|
| In neat (no solvent) byproducts: ClF; condensation of ClF3 at -196°C to U in a reactor; warming to room temp. (cooling reactor with compressed air); pumping off volatile material after 1 h at -78°C; warming the bomb to -50°C; pumping off traces of ClF3;; sublimation; | 85% |
| In hydrogen fluoride Kinetics; at 30°C; influence of HF, formed UF6, ClF3 and HF concn. and rise in temp. at 60°C in presence of BrF3;; | |
| In neat (no solvent) Kinetics; above 60 - 80°C;; |
-
-
20816-12-0
osmium(VIII) oxide

-
-
7790-91-2
chlorine trifluoride

-
A
-
19184-41-9
osmium pentafluoride oxide

-
B
-
24762-17-2, 58800-18-3
osmium trioxide difluoride

| Conditions | Yield |
|---|---|
| In neat (no solvent) OsO4 was condensed into a reaction tube at -196°C; ClF3 was condensed at the same temp. onto the osmium oxide; the mixt. was then allowed to warm up to ambient temp. and stored for 1 d;; OsOF5 was vaporized at -10°C and condensed into a trap at -196°C; OsO3F2 was sublimed in a static vac. in passivated sapphire tube at 130°C;; | A 15.6% B 82.7% |


| Conditions | Yield |
|---|---|
| In neat (no solvent) | 80% |
| In neat (no solvent) at room temp. in presence of moisture;; | 80% |

| Conditions | Yield |
|---|---|
| With chlorine at 300℃; for 48h; Autoclave; Inert atmosphere; | 72% |
| With chlorinated hydrocarbons In neat (no solvent) | |
| With chlorine In neat (no solvent) heating 1:1 mixture at 180 - 350°C;; |

| Conditions | Yield |
|---|---|
| In water-d2 byproducts: O2, HF, Cl2; ClF3 addn. to D2O (77 K), heating to room temp., pumping off of O2 and HF (77 K); fractional condensation (170 K); 70-95% (18)O; | 50% |

| Conditions | Yield |
|---|---|
| In neat (no solvent) at room temp. in presence of moisture;; | 45% |

| Conditions | Yield |
|---|---|
| In neat (no solvent) at room temp. in presence of moisture;; | 12% |

| Conditions | Yield |
|---|---|
| With catalyst: charcoal CO dild. with a double amt. of N2, -18°C; | 10% |
-
-
7790-91-2
chlorine trifluoride

-
-
10049-17-9
rhenium hexafluoride

-
-
4648-54-8
trimethylsilylazide

-
A
-
84159-30-8
ReF5(NCl)

-
B
-
84159-29-5
rhenium tetrafluoride nitride

| Conditions | Yield |
|---|---|
| In 1,1,2-Trichloro-1,2,2-trifluoroethane ReF6 was reacted with ligand at -50°C in CF3ClCFCl2, allowed to warm to 0°C, solvent was removed, residue was warmed to 70°C in vac., sublimed, involatile residue was reacted with ClF3 at 0°C; sublimed at 70°C in vac.; | A n/a B 10% |


| Conditions | Yield |
|---|---|
| In neat (no solvent) at ambient temp.;; | |
| room temp.; | |
| In neat (no solvent) at room temp. in presence of moisture, (in absence of moisture not quantitative react.);; | >99 |

| Conditions | Yield |
|---|---|
| In neat (no solvent) at room temp. in presence of moisture, (in absence of moisture not quantitative react.);; | >99 |
-
-
7790-91-2
chlorine trifluoride

-
-
7553-56-2
iodine

-
-
676353-70-1, 51096-45-8, 7783-66-6
iodine pentafluoride

-
B
-
13873-84-2
iodine monofluoride

| Conditions | Yield |
|---|---|
| In neat (no solvent) |


| Conditions | Yield |
|---|---|
| In neat (no solvent) passing ClF3 into Br2 at 20 - 30°C;; | 60-80 |
| In neat (no solvent) bubbling ClF3 through liq. Br2; aerated with N2, distd.; | >99 |
| In neat (no solvent) byproducts: Cl2; distd. (segregating 125-127°C fraction; cooled cyclically three times to 0°C in nickel container; chromy.; DTA; |
-
-
7790-91-2
chlorine trifluoride

-
-
7726-95-6
bromine

-
-
177570-08-0, 7789-30-2, 676353-69-8
bromine pentafluoride

| Conditions | Yield |
|---|---|
| In neat (no solvent) passing excess of ClF3 into Br2 at 20 - 30°C;; |

-
-
7790-91-2
chlorine trifluoride

-
A
-
13637-87-1
chlorodifluoroamine

-
B
-
7727-37-9
nitrogen

-
C
-
7782-50-5
chlorine

| Conditions | Yield |
|---|---|
| In neat (no solvent) at -50 - -5°C;; | |
| In neat (no solvent) at -50 - -5°C;; |


| Conditions | Yield |
|---|---|
| In neat (no solvent) solvolysis at 100°C;; | |
| In neat (no solvent) solvolysis at 100°C;; |

| Conditions | Yield |
|---|---|
| In neat (no solvent) solvolysis at 100°C;; | |
| In neat (no solvent) solvolysis at 100°C;; |

| Conditions | Yield |
|---|---|
| In hydrogen fluoride byproducts: NaHF2; anhydrous HF; | |
| In hydrogen fluoride byproducts: NaHF2; HF (liquid); anhydrous HF; |


| Conditions | Yield |
|---|---|
| In neat (no solvent) heating at 250°C;; | |
| In neat (no solvent) at 250°C;; | |
| In neat (no solvent) heating at 250°C;; | |
| In neat (no solvent) at 250°C;; |

| Conditions | Yield |
|---|---|
| byproducts: Cl2; |

| Conditions | Yield |
|---|---|
| In neat (no solvent) solvolysis at 100°C;; | |
| In neat (no solvent) solvolysis at 100°C;; |

| Conditions | Yield |
|---|---|
| In neat (no solvent) at 25 - 180°C and 0.3 - 1 atm;; | |
| In neat (no solvent) at 25 - 180°C and 0.3 - 1 atm;; |

| Conditions | Yield |
|---|---|
| In neat (no solvent) decompn. of ClF3 on heating (Ni wire);; |

| Conditions | Yield |
|---|---|
| With metal In neat (no solvent) | |
| In neat (no solvent) byproducts: Cl2; other Radiation; α-particles; at ambient temp.;; | |
| In neat (no solvent) other Radiation; α-radiation;; detection by mass spectroscopy;; |

| Conditions | Yield |
|---|---|
| In neat (no solvent) at 12°C;; | |
| In neat (no solvent) at 120°C;; |
Chlorine trifluoride Chemical Properties
Molecular Structure of Chlorine trifluoride (CAS NO. 7790-91-2):
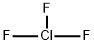
EINECS: 232-230-4
Chemical Name: Chlorine trifluoride
Molecular Formula: ClF3
XLogP3-AA: 2.5
H-Bond Donor: 0
H-Bond Acceptor: 3
InChI: InChI=1S/ClF3/c2-1(3)4
InChIKey: JOHWNGGYGAVMGU-UHFFFAOYSA-N
Density: 1.8 g/cm3
Molecular Weight: 92.448210 g/mol
Enthalpy of Vaporization: 27.53 kJ/mol
Boiling Point: 11.3 °C at 760 mmHg
Vapour Pressure: 1310 mmHg at 25 °C
Melting Point: -83 °C
Water Solubility: 7840 mg/L at 25 °C
Chlorine trifluoride Uses
Chlorine trifluoride is mainly used to produce uranium hexafluoride :
U + 3ClF3 → UF6 + 3ClF
Before the start of World War II, Chlorine trifluoride under the code name of N-stoff was investigated for military applications by the Kaiser Wilhelm Institute in Nazi German. It was found to be an effective combined incendiary weapon and poison gas. Now, it is never used in war. Chlorine trifluoride has also been investigated as a high-performance storable oxidizer in rocket propellant systems due to its hypergolic properties with every known fuel even things like cloth, wood, and test engineers. Chlorine trifluoride is used in the semiconductor industry to clean chemical vapour deposition chambers without having to dismantle the chamber. It does not need to be activated by plasma as the heat of the chamber is enough to make it decompose and react with the semiconductor material.
Chlorine trifluoride Production
Chlorine trifluoride was prepared by fluorination of chlorine first reported by Ruff and Krug. The reaction also produces ClF, then the mixture was separated by distillation.
3F2 + Cl2 → 2ClF3
Chlorine trifluoride Toxicity Data With Reference
| 1. | eye-rat 21 ppm/12H-I | AMIHAB AMA Archives of Industrial Health. 12 (1955),515. | ||
| 2. | eye-dog 21 ppm/12H-I | AMIHAB AMA Archives of Industrial Health. 12 (1955),515. | ||
| 3. | ihl-hmn LCLo:50 ppm | 34ZIAG Toxicology of Drugs and Chemicals, Deichmann, W.B.,New York, NY.: Academic Press, Inc.,1966,651. | ||
| 4. | ihl-rat LCLo:400 ppm/30M | TXAPA9 Toxicology and Applied Pharmacology. 27 (1974),527. | ||
| 5. | ihl-mus LC50:178 ppm/1H | AMRL** Aerospace Medical Research Laboratory Report. (Aerospace Technical Div., Air Force Systems Command, Wright-Patterson Air Force Base, OH 45433) TR-70-55 ,1970. | ||
| 6. | ibl-mky LC50:230 ppm/1H | AMRL** Aerospace Medical Research Laboratory Report. (Aerospace Technical Div., Air Force Systems Command, Wright-Patterson Air Force Base, OH 45433) TR-70-77 ,1970. |
Chlorine trifluoride Consensus Reports
Reported in EPA TSCA Inventory.
Chlorine trifluoride Safety Profile
Safety Information of Chlorine trifluoride (CAS NO. 7790-91-2):
Hazard Codes: O 
Hazard Note: Oxidising agent
Risk Statements: 8-35
R8 :Contact with combustible material may cause fire
R35:Causes severe burns
Safety Statements: 17-38
S17:Keep away from combustible material
S38:In case of insufficient ventilation, wear suitable respiratory equipment
RIDADR: 1749
HazardClass: 2.3
Human poison by inhalation. An eye irritant. See also FLUORIDES, CHLORINE, and FLUORINE. Spontaneously flammable. A powerful oxidant which may react violently with oxidizable materials. A rocket propellant.
Explosive reaction with water, bis(trifluoro methyl)sulfide or -disulfide, polychlorotrifluoroethylene, trifluoromethanesulfenyl chloride, and other hydrogen- containing materials (e.g., ammonia, coal gas, hydrogen, hydrogen sulfide, methane, acetic acid, benzene, ether, cotton, paper, wood). Forms shock-sensitive explosive mixtures with highly chlorinated compounds (e.g., carbon tetrachloride), nitroaryl compounds (e.g., trinitrotoluene, hexanitrobiphenyl, hexanitrodiphenyl amine, hexanitro diphenyl sulfide, hexanitrodiphenyl ether). Reaction with ammonium fluoride or ammonium hydrogen fluoride forms explosive gaseous products.
Ignition on contact with boron-containing materials, iodine, finely divided refractory materials (e.g., asbestos, glass wool, sand, tungsten carbide), fluorinated polymers (with flowing trifluoride).
Violent reaction with acids (e.g., nitric or sulfuric), chromium trioxide, ruthenium, selenium tetrafluoride (above 106°C), metals, metal oxides, metal salts, nonmetals, nonmetal salts, organic matter, glass wool, acetic acid, Al, Sb, As, Cu, Ir, Fe, Pb, Mg, Mo, Os, P, K, Rh, Se, Si, Ag, Na, S, Te, Sn, W, Zn, oxides, CO, graphite, HgI2, HNO3, K2CO3, KI, rubber, AgN3, AgNO3, NaOH, V2P5, WO3. Incompatible with fuels, nitro compounds. When heated to decomposition or in reaction with water or steam it emits toxic fumes of F− and Cl−.
Chlorine trifluoride Standards and Recommendations
OSHA PEL: CL 0.1 ppm
ACGIH TLV: CL 0.1 ppm
DFG MAK: 0.1 ppm (0.38 mg/m3)
DOT Classification: 2.3; Label: Poison Gas, Oxidizer, Corrosive
Chlorine trifluoride Specification
Chlorine trifluoride with cas registry number of 7790-91-2 is also called for Chlorine fluoride ; Chlorine fluoride (ClF3) ; Chlorine trifluoride (ClF3) ; Chlorotrifluoride ; HSDB 970 ; Trifluorure de chlore ; Trifluorure de chlore [French] ; Chlorine trifluoride [UN1749] [Poison gas] ; UN1749 . It is A colorless gas or green liquid with a pungent odor. Pure Chlorine trifluoride is stable to 180° in glass vessels; above this temperature it decomposes by a free radical mechanism to the elements.
Related Products
- Chlorine
- Chlorine azide
- Chlorine dioxide
- CHLORINE DIOXYGEN TRIFLUORIDE
- Chlorine nitrate
- Chlorine pentafluoride
- Chlorine perchlorate
- Chlorine trifluoride
- CHLORINE(1)TRIFLUOROMETHANE-SULFONATE
- Chlorine, atomic
- 7790-93-4
- 7790-94-5
- 7790-98-9
- 7790-99-0
- 7791-03-9
- 7791-07-3
- 7791-08-4
- 7791-11-9
- 7791-12-0
- 7791-13-1
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View