Simagchem Corporation
Welcome to Simagchem, your partner in China as a premier supply of bulk specialty chemicals for industry and life science. We introduce experienced quality product and exceptional JIT service with instant market intelligence in China to benefit our
Cas:2052-07-5
Min.Order:1 Metric Ton
Negotiable
Type:Manufacturers
inquiryHangzhou Dingyan Chem Co., Ltd
Items Standard Result Assay (Ursolic acid) 98%min 98.22% ----------------------------------------------------------------
Cas:2052-07-5
Min.Order:1 Gram
FOB Price: $100.0 / 500.0
Type:Trading Company
inquiryHenan Allgreen Chemical Co.,Ltd
The company has a professional R & D team, mature technology, very competitive prices and stable high quality products for customers to order high quality and low price product efforts! Appearance:White or off-white Solid Storage:Sealed, dry, m
Ality Chemical Corporation
The above product is Ality Chemical's strong item with best price, good quality and fast supply. Ality Chemical has been focusing on the research and production of this field for over 14 years. At the same time, we are always committed to providi
Xi'an Xszo Chem Co., Ltd.
1. Factory price and high quality must be guaranteed, base on 8 years of production and R&D experience2. Free samples will be provided,ensure specifications and quality are right for customer3. Customers will receive the most professional technical s
Cas:2052-07-5
Min.Order:1 Gram
FOB Price: $0.1
Type:Manufacturers
inquiryDayang Chem (Hangzhou) Co.,Ltd.
As a leading manufacturer and supplier of chemicals in China, DayangChem not only supply popular chemicals, but also DayangChem’s R&D center offer custom synthesis according to the contract research and development services for the fine c
Cas:2052-07-5
Min.Order:1 Kilogram
FOB Price: $15.0 / 16.0
Type:Lab/Research institutions
inquiryChemwill Asia Co., Ltd.
Our main production base is located in Xuzhou industry park. We are certified both to the ISO 9001 and ISO 14001 Standards, have a safety management system in place.Our R&D team masters core technology for process-design of target building block
Cas:2052-07-5
Min.Order:5 Kiloliter
FOB Price: $1.2 / 5.0
Type:Manufacturers
inquiryHenan Tianfu Chemical Co., Ltd.
TIANFUCHEM--2052-07-5--2-Bromobiphenyl factory price Our company was built in 2009 with an ISO certificate.In the past 10 years, we have grown up as a famous fine chemicals supplier in China And we had established stable business relati
Cas:2052-07-5
Min.Order:1 Metric Ton
FOB Price: $2000.0
Type:Lab/Research institutions
inquiryJinan Finer Chemical Co., Ltd
Product Description Product name 2-Bromobiphenyl CAS 2052-07-5 Assay 99% Appearance Colorless liquid
Cas:2052-07-5
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryHebei Nengqian Chemical Import and Export Co., LTD
With our good experience, we offer detailed technical support and advice to assist customers. We communicate closely with customers to establish their quality requirements. Consistent Quality Our plant has strict quality control in each manufacturin
Cas:2052-07-5
Min.Order:1 Kilogram
FOB Price: $5.0 / 10.0
Type:Trading Company
inquiryZhuozhou Wenxi import and Export Co., Ltd
WITH US,YOUR MONEY IN SAFE,YOUR BUSINESS IN SAFE 1)Quick Response Within 12 hours; 2)Quality Guarantee: All products are strictly tested by our QC, confirmed by QA and approved by third party lab in China, USA, Canada, Germany, UK, Italy, France et
Cas:2052-07-5
Min.Order:1 Kilogram
FOB Price: $139.0 / 210.0
Type:Trading Company
inquiryHenan Sinotech Import&Export Corporation
Product name:2-Bromobiphenyl CAS No:2052-07-5 Appearance:Pale yellow liquid Content:98% Appearance:Pale yellow liquid Storage:Preserve in well-closed, light-resistant and airtight containers.
Hubei Langyou International Trading Co., Ltd
1. Guaranteed purity; 2. Large quantity in stock; 3. Largest manufacturer; 4. Best service after shipment with email; 5. High quality & competitive price; Appearance:White crystalline granules or powder. Storage:Store in seale
Cas:2052-07-5
Min.Order:100 Gram
Negotiable
Type:Other
inquiryShanghai Upbio Tech Co.,Ltd
1.No Less 8 years exporting experience. Clients can 100% received goods 2.Lower Price with higher quality 3,Free sample 4,We are sincerely responsible for the "product quality" and "After Service" Upbio is Specialized
Cas:2052-07-5
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryLeader Biochemical Group
About Product Details Items Specifications Test Results Appearance White to white crystalline powde
Cas:2052-07-5
Min.Order:100 Kilogram
FOB Price: $1.0 / 2.0
Type:Lab/Research institutions
inquiryBaoji Guokang Healthchem co.,ltd
Our company has been in existence for 10 years since its establishment. We have our own unique team. The company integrates independent research and development, production and sales. We have established famous brands at home and abroad. At prese
Cas:2052-07-5
Min.Order:1 Kilogram
FOB Price: $86.4 / 92.1
Type:Trading Company
inquiryQingdao Beluga Import and Export Co., LTD
2-Bromobiphenyl CAS: 2052-07-5 Qingdao Belugas Import and Export Co., Ltd. is a scientific and technological company integrating research and development, production and trade of chemical intermediates, specializing in high quality organic intermedi
Cas:2052-07-5
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryShandong Hanjiang Chemical Co., Ltd.
Hello, dear friend! I'm Hansen and Allen from China. Welcome to my lookchem mall! The following is a brief introduction of our company's products and services. If you are interested in our products, please contact us by emai
Cas:2052-07-5
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryHubei Jiutian Bio-medical Technology Co., Ltd
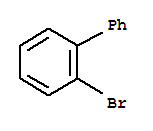
Name: 1,1'-Biphenyl, 2-bromo- Synonyms: PBB 1; NSC 67353; o-Bromobiphenyl; 2-Biphenyl bromide; m-Biphenyl bromide; 2-Biphenylyl bromide; 2-Phenylbromobenzene; 2-Bromo-1,1'-biphenyl; 1-Bromo-2-phenylbenzene; Biphenyl,2-bromo- (6CI,7CI,8CI) CAS
Cas:2052-07-5
Min.Order:1 Metric Ton
Negotiable
Type:Lab/Research institutions
inquiryHenan Wentao Chemical Product Co., Ltd.
Henan Wentao Chemical Product Co.,Ltd is Located in Zhengzhou High-tech Development Zone with import and export license, We passed ISO 9001:2008 as well, Henan Wentao has developed more than 1000 compounds, which are widely used in the fields of prod
Triumph International Development Limilted
Triumph has the complete production of G- KG - MT service chain,we can make the new technology into productivity quickly in the research and development of new products. Main Service 1.Own made fine chemical products 2.Out sourcing and qua
Hangzhou Keyingchem Co.,Ltd
Hangzhou KeyingChem Co., Ltd. exported this product to many countries and regions at best price. If you are looking for the material’s manufacturer or supplier in China, KeyingChem is your best choice. Pls contact with us freely for getting det
Cas:2052-07-5
Min.Order:0 Metric Ton
Negotiable
Type:Lab/Research institutions
inquiryHangzhou J&H Chemical Co., Ltd.
J&H CHEM R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. J&H CHEM has some Manufacturing base in Jia
Shanghai Massive Chemical Technology Co., Ltd.
Massive Chemical is certified with ISO9001 and ISO14001 manufacturer for this product. We will offer all documents as requirement for the materials which includes, Certificate of Analysis, Material Safety Data Sheet, and Method of Analysis and
Cas:2052-07-5
Min.Order:1 Gram
FOB Price: $1.0
Type:Lab/Research institutions
inquiryZibo Hangyu Biotechnology Development Co., Ltd
Zibo Hangyu Biotechnology Development Co., Ltd is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemi
Cas:2052-07-5
Min.Order:10 Gram
FOB Price: $100.0
Type:Lab/Research institutions
inquiryHangzhou Lingrui Chemical Co.,Ltd.
2-BromobiphenylAppearance:white to off-white powder Storage:cool temperature Package:according to customers' requirements Application:intermediate Transportation:By air(EMS or EUB or FedEx or TNT ect...) or by sea(FOB or CIF or CNF ect...)or accordin
Zibo Dorne chemical technology co. LTD
Product Details Grade: pharmaceutical grade Purity:99%+ ProductionCapacity: 1000 Kilogram/Month Scope of use: For scientific research only(The product must be used legally) Our Advantage 1. Best quality with competitive price. 2. Quick shipping,
Cas:2052-07-5
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryXiamen Jenny Chemical Technology Co., Ltd.
GMP standard, high purity, competitive price, in stock 1. Quick Response: within 6 hours after receiving your email. 2. Quality Guarantee: All products are strictly tested by our QC, confirmed by QA, and approved by a third-party lab in China, USA,
Shanghai Minstar Chemical Co., Ltd
2-Bromobiphenyl Basic information Product Name: 2-Bromobiphenyl Synonyms: 2-Biphenylyl bromide;2-Phenylbromobenzene;Biphenyl-2-yl bromide;2-Bromobiphenyl,98%;2-BROMOBIPHENYL FOR SYNTHESIS;1-broMo-2-phenylbenzene;2-BroMobiphenyl, 98% 5GR;2-Brom
Hangzhou Huarong Pharm Co., Ltd.
Hangzhou Huarong Pharm Co., Ltd.established since 2006 , has been actively developing specialty products for Finished Dosages, APIs, Intermediates, and Fine chemicals markets in North America, Europe, Korea, Japan, Mid-East and all over the World. Hu
Synthetic route

| Conditions | Yield |
|---|---|
| With C84H60Cl6Fe3N6Pd3; triethylamine at 20℃; for 0.416667h; | 96% |
| With tetra-butylammonium acetate; Pd EnCat-30TM In ethanol at 120℃; for 0.166667h; Suzuki cross-coupling; microwave irradiation; | 86% |
-
-
1538-62-1, 34716-94-4
triphenylantimony(V) diacetate

-
-
244205-40-1
(2-bromophenyl)boronic acid

-
-
2052-07-5
2-Bromobiphenyl

| Conditions | Yield |
|---|---|
| tetrakis(triphenylphosphine) palladium(0) In 1,4-dioxane at 60℃; for 18h; Suzuki-type cross-coupling reaction; | 95% |

| Conditions | Yield |
|---|---|
| With bis-triphenylphosphine-palladium(II) chloride; potassium carbonate In methanol; toluene Suzuki-Miyaura Coupling; Inert atmosphere; Reflux; | 93% |
| With bis-triphenylphosphine-palladium(II) chloride; potassium carbonate In 1,2-dimethoxyethane; water at 80℃; for 5h; Inert atmosphere; | 90% |
| With bis-triphenylphosphine-palladium(II) chloride; potassium carbonate In 1,2-dimethoxyethane; water at 80℃; for 5h; Inert atmosphere; | 90% |

| Conditions | Yield |
|---|---|
| With manganese triacetate for 0.5h; Heating; | 90% |
| With potassium permanganate; acetic acid Heating; | 90% |
| With manganese triacetate; acetic acid for 0.5h; Microwave irradiation; | 81% |
| With di-tert-butyl peroxide at 80℃; for 24h; Inert atmosphere; | 50% |
| With potassium permanganate; acetic acid at 100℃; for 24h; |
-
-
583-53-9
1,2-dibromobenzene

-
-
98-80-6
phenylboronic acid

-
A
-
2052-07-5
2-Bromobiphenyl

-
B
-
84-15-1
o-terphenyl

| Conditions | Yield |
|---|---|
| With bis(1,5-cyclooctadiene)nickel (0); potassium phosphate; tricyclohexylphosphine In tetrahydrofuran for 20h; Suzuki Coupling; Inert atmosphere; | A n/a B 90% |
| With 1-((dimethylamino)(phenyl)methyl)naphthalen-2-ol; tetrabutylammomium bromide; palladium diacetate; potassium carbonate In 1,4-dioxane; water at 40℃; for 5h; Suzuki coupling; Sonication; | A 10% B 87% |
| With palladium diacetate In ethanol at 20℃; for 0.25h; Suzuki-Miyaura Coupling; chemoselective reaction; | A 74% B 13% |
| With sodium carbonate In N,N-dimethyl-formamide at 110℃; for 6h; Suzuki-Miyaura coupling reaction; Inert atmosphere; | A 30% B 55% |
| With disodium[(N,N’-bis(2-hydroxy-5-sulfonatobenzyl)-1,2-diphenyl-1,2-diaminoethano)palladate(II)]; caesium carbonate In water at 80℃; for 1h; Suzuki-Miyaura Coupling; | A 17% B 34% |


| Conditions | Yield |
|---|---|
| With dipotassium peroxodisulfate; silver nitrate In acetonitrile at 120℃; for 10h; Inert atmosphere; Sealed tube; Green chemistry; | 84% |

| Conditions | Yield |
|---|---|
| Stage #1: 2-bromoaniline With hydrogenchloride; sodium nitrite In water; acetone at 0 - 5℃; for 0.116667h; Stage #2: benzene In water; acetone at 25℃; for 5h; Sonication; | 84% |

| Conditions | Yield |
|---|---|
| With potassium acetate; 18-crown-6 ether for 1.5h; Ambient temperature; | 81% |
| With potassium acetate; tetra(n-butyl)ammonium hydrogensulfate for 24h; Ambient temperature; | 51 % Chromat. |

| Conditions | Yield |
|---|---|
| With diethyl bromomethylmalonate; [4,4′-di-tert-butyl-2,2′-bipyridine-N1,N1′]bis{3,5-difluoro-2-[5-(trifluoromethyl)-2-pyridinyl-N]phenyl-C}iridium(III) hexafluorophosphate; caesium carbonate In acetonitrile at 55℃; for 22h; Reagent/catalyst; Temperature; Irradiation; | 81% |

| Conditions | Yield |
|---|---|
| With hydrogen bromide; copper(I) bromide; sodium nitrite In water for 2h; Ambient temperature; | 70% |
| With hydrogen bromide; copper(I) bromide Diazotization; | |
| With hydrogenchloride Diazotization.Behandlung der Diazoniumsalz-Loesung mit CuBr; |

| Conditions | Yield |
|---|---|
| With manganese triacetate Heating; | 70% |

| Conditions | Yield |
|---|---|
| With tetrabutyl ammonium fluoride; palladium diacetate In water at 95℃; for 3h; Suzuki coupling; | 70% |

| Conditions | Yield |
|---|---|
| With palladium diacetate In water at 20℃; for 12h; Suzuki-Miyaura Coupling; | 69% |
-
-
583-55-1
1-Bromo-2-iodobenzene

-
-
98-80-6
phenylboronic acid

-
A
-
2052-07-5
2-Bromobiphenyl

-
B
-
84-15-1
o-terphenyl

| Conditions | Yield |
|---|---|
| With palladium diacetate In ethanol at 20℃; for 0.333333h; Suzuki-Miyaura Coupling; | A 24% B 68% |
| With dichloro bis(acetonitrile) palladium(II); C10H11N3S; potassium carbonate In water; N,N-dimethyl-formamide at 20℃; for 1h; Suzuki Coupling; Schlenk technique; Inert atmosphere; | A 36% B 62% |
| With sodium carbonate; palladium diacetate In ethanol; water at 35℃; for 3h; Suzuki; Title compound not separated from byproducts; |


| Conditions | Yield |
|---|---|
| With palladium diacetate In water at 20℃; for 24h; Suzuki-Miyaura Coupling; Inert atmosphere; | 65% |

| Conditions | Yield |
|---|---|
| With sodium carbonate In tetrahydrofuran; water at 80℃; Inert atmosphere; | 62% |
| Stage #1: 2,3-dibromobenzene; phenylboronic acid With potassium carbonate In tetrahydrofuran; toluene for 0.25h; Suzuki coupling; Inert atmosphere; Stage #2: With tetrakis(triphenylphosphine) palladium(0) In tetrahydrofuran; toluene at 110℃; for 24h; Suzuki coupling; Inert atmosphere; | 41.8% |
| With sodium carbonate at 30℃; for 16h; Suzuki Coupling; Schlenk technique; Inert atmosphere; | |
| With C36H36Cl2Fe2NPPd; sodium carbonate In N,N-dimethyl-formamide at 100℃; for 18h; Suzuki-Miyaura Coupling; Inert atmosphere; Schlenk technique; | 91 %Chromat. |

| Conditions | Yield |
|---|---|
| With potassium tert-butylate; vasicine at 20 - 110℃; for 72h; Schlenk technique; Inert atmosphere; | A 60% B 6% |

-
-
583-55-1
1-Bromo-2-iodobenzene

-
-
71-43-2
benzene

-
A
-
92-52-4
biphenyl

-
B
-
2052-07-5
2-Bromobiphenyl

-
C
-
84-15-1
o-terphenyl

-
D
-
2113-51-1
2-iodobiphenyl

| Conditions | Yield |
|---|---|
| With 6,13,14,21-tetraphenyl-22H-tribenzo[b,g,m]-[14]triphyrin(2.1.1); potassium hydroxide; tert-butyl alcohol at 180℃; for 24h; Darkness; | A 19 %Chromat. B 8% C 60% D 11% |

-
-
108-86-1
bromobenzene

-
-
108-95-2
phenol

-
A
-
2052-07-5
2-Bromobiphenyl

-
B
-
92-66-0
4-bromo-1,1'-biphenyl

-
C
-
2113-57-7
3-bromobiphenyl

| Conditions | Yield |
|---|---|
| With tert.-butylhydroperoxide; iodine at 289.9℃; for 0.0272222h; Product distribution; | A 22.7% B 18.4% C 58.9% |


| Conditions | Yield |
|---|---|
| With tert.-butylhydroperoxide; 1,2-Dinitrobenzene In decane at 150℃; for 12h; Inert atmosphere; Sealed tube; | 58% |
-
-
129112-25-0
2-(trifluoromethanesulfonyl)oxy-bromobenzene

-
-
98-80-6
phenylboronic acid

-
A
-
2052-07-5
2-Bromobiphenyl

-
B
-
84-15-1
o-terphenyl

-
C
-
17763-65-4
biphenyl-2-yl trifluoromethanesulfonate

| Conditions | Yield |
|---|---|
| With tris-(dibenzylideneacetone)dipalladium(0); sodium carbonate; 1,4-di(diphenylphosphino)-butane In water; toluene for 16h; Suzuki-Miyaura Coupling; Inert atmosphere; Heating; | A 57.1% B 8.8% C 30.9% |


| Conditions | Yield |
|---|---|
| With tert.-butylhydroperoxide; 1,2-Dinitrobenzene In decane at 150℃; for 12h; Inert atmosphere; Sealed tube; | 54% |

| Conditions | Yield |
|---|---|
| Stage #1: bromobenzene With tert.-butyl lithium In tetrahydrofuran; hexane; pentane for 1h; Inert atmosphere; Cooling; Stage #2: 2,3-dibromobenzene In tetrahydrofuran; hexane; pentane at -50℃; for 2h; Reagent/catalyst; Solvent; Inert atmosphere; | 44% |
| Stage #1: bromobenzene With tert.-butyl lithium In toluene; pentane at -80℃; for 1h; Inert atmosphere; Stage #2: With lithium bromide In 1,2-dimethoxyethane; toluene; pentane at -80℃; for 0.0833333h; Inert atmosphere; Stage #3: 2,3-dibromobenzene In 1,2-dimethoxyethane; toluene; pentane at -50℃; for 0.75h; Solvent; Inert atmosphere; |
-
-
108-86-1
bromobenzene

-
-
583-53-9
1,2-dibromobenzene

-
A
-
2052-07-5
2-Bromobiphenyl

-
B
-
13029-09-9
2,2'-dibromobiphenyl

| Conditions | Yield |
|---|---|
| Stage #1: bromobenzene With tert.-butyl lithium In tetrahydrofuran at -80℃; for 1h; Inert atmosphere; Stage #2: 2,3-dibromobenzene In tetrahydrofuran at -50℃; for 2h; Inert atmosphere; | A 44% B 9% |

-
-
583-55-1
1-Bromo-2-iodobenzene

-
-
143-66-8
sodium tetraphenyl borate

-
A
-
2052-07-5
2-Bromobiphenyl

-
B
-
84-15-1
o-terphenyl

| Conditions | Yield |
|---|---|
| With palladium diacetate; sodium carbonate In water; acetone at 25℃; for 50h; | A 43% B 17% |
| With palladium diacetate; sodium carbonate In water; acetone at 25℃; for 0.666667h; Product distribution; other solvent composition, reaction time; | A 77 % Chromat. B 14 % Chromat. |


| Conditions | Yield |
|---|---|
| Stage #1: phenyllithium In tetrahydrofuran; dibutyl ether; cyclohexane at -78℃; for 1h; Inert atmosphere; Stage #2: In tetrahydrofuran; dibutyl ether; cyclohexane at -50℃; for 2h; Inert atmosphere; Stage #3: 2,3-dibromobenzene In tetrahydrofuran; dibutyl ether; cyclohexane at -50℃; for 2h; Solvent; Inert atmosphere; | 42% |

| Conditions | Yield |
|---|---|
| With 1-(2-hydroxyethyl)piperazine; potassium tert-butylate at 100℃; for 24h; Sealed tube; Inert atmosphere; | A 34% B 30% |

-
-
108-86-1
bromobenzene

-
-
17420-03-0
N-sulphinylphenylhydrazine

-
A
-
2052-07-5
2-Bromobiphenyl

-
B
-
92-66-0
4-bromo-1,1'-biphenyl

-
C
-
2113-57-7
3-bromobiphenyl

-
D
-
882-33-7
diphenyldisulfane

| Conditions | Yield |
|---|---|
| at 135℃; for 72h; Further byproducts given; | A 20.9% B 6.9% C 12.7% D 1.1% E n/a |
| at 135℃; for 72h; Further byproducts given; | A 19.5% B 8.1% C 12.9% D 1.1% E n/a |

-
-
108-86-1
bromobenzene

-
-
17420-03-0
N-sulphinylphenylhydrazine

-
A
-
1212-08-4
S-Phenyl benzenethiosulfonate

-
B
-
2052-07-5
2-Bromobiphenyl

-
C
-
92-66-0
4-bromo-1,1'-biphenyl

-
D
-
2113-57-7
3-bromobiphenyl

| Conditions | Yield |
|---|---|
| at 135℃; for 72h; Further byproducts given; | A 0.2% B 20.9% C 6.9% D 12.7% E n/a |


| Conditions | Yield |
|---|---|
| Stage #1: 2-Bromobiphenyl With n-butyllithium; butyl magnesium bromide In tetrahydrofuran at 0℃; for 0.5h; Stage #2: allyl bromide With copper(I) cyanide lithium chloride In tetrahydrofuran at -78℃; for 0.5h; | 100% |
| Stage #1: 2-Bromobiphenyl With N,N,N,N,-tetramethylethylenediamine; C4H9Mg(1+)*2C8H17O(1-)*Li(1+) In toluene at 25℃; for 0.25h; Schlenk technique; Inert atmosphere; Stage #2: With copper(l) iodide In toluene at 0℃; for 1h; Schlenk technique; Inert atmosphere; Stage #3: allyl bromide In toluene at 0℃; for 1h; Inert atmosphere; Schlenk technique; | 92% |
| (i) Mg, (ii) /BRN= 605308/; Multistep reaction; |

| Conditions | Yield |
|---|---|
| With LiCrH4*2LiCl*2THF In tetrahydrofuran at 25℃; | 100% |
| With vanadium monochloride In tetrahydrofuran at 25℃; for 12h; Inert atmosphere; | 100% |
| With [ruthenium(II)(η6-1-methyl-4-isopropyl-benzene)(chloride)(μ-chloride)]2; potassium tert-butylate; isopropyl alcohol at 100℃; Schlenk technique; Inert atmosphere; | 96% |

| Conditions | Yield |
|---|---|
| With cesium pivalate; palladium(0)bis(tricyclohexylphosphine) In N,N-dimethyl-formamide at 110℃; under 760.051 Torr; for 7h; Carbonylation; cyclization; | 100% |
| With tristricyclohexylphosphine palladium(0); cesium pivalate In N,N-dimethyl-formamide at 110℃; under 760.051 Torr; for 7h; | 100% |
| With palladium diacetate; sodium carbonate; magnesium sulfate; tricyclohexylphosphine tetrafluoroborate; Trimethylacetic acid In toluene at 110℃; under 760.051 Torr; for 25h; Sealed tube; Inert atmosphere; | 86% |

| Conditions | Yield |
|---|---|
| Stage #1: 2-Bromobiphenyl; tri-n-butyllithium magnesate complex In tetrahydrofuran; hexane at 0℃; for 0.5h; Stage #2: allyl bromide With CuCN*2LiCl In tetrahydrofuran; hexane at -78℃; for 0.5h; Further stages.; | 100% |


| Conditions | Yield |
|---|---|
| Stage #1: 2-Bromobiphenyl With tert.-butyl lithium In tetrahydrofuran at -78℃; for 0.5h; Stage #2: N,N-dimethyl-formamide In tetrahydrofuran at -78 - 20℃; | 100% |
| Stage #1: 2-Bromobiphenyl With n-butyllithium In tetrahydrofuran; hexane at -80℃; for 0.5h; Stage #2: N,N-dimethyl-formamide In tetrahydrofuran; hexane at -80 - 20℃; | 96% |
-
-
2052-07-5
2-Bromobiphenyl

-
-
107035-97-2
2,5-dimethyl-3-(1-methylethyl)-5-hexen-3-ol

| Conditions | Yield |
|---|---|
| With caesium carbonate; Tri(p-tolyl)phosphine; palladium diacetate In N,N-dimethyl-formamide; toluene at 200℃; for 0.25h; microwave irradiation; | 100% |
-
-
2052-07-5
2-Bromobiphenyl

-
-
593-51-1
methylamine hydrochloride

-
-
14925-09-8
2-(N-methylamino)-1,1'-biphenyl

| Conditions | Yield |
|---|---|
| With (R)-(-)-1-[(SP)-2-(dicyclohexylphosphino)ferrocenyl]ethyldi-tert-butylphosphine; bis(tri-ortho-tolylphosphine)palladium(0); sodium t-butanolate In 1,4-dioxane at 100℃; for 12h; Inert atmosphere; Glovebox; | 99% |

| Conditions | Yield |
|---|---|
| With C30H43O2P*C13H12N(1-)*CH3O3S(1-)*Pd(2+); lithium hexamethyldisilazane In 1,4-dioxane at 100℃; for 16h; Inert atmosphere; | 99% |

| Conditions | Yield |
|---|---|
| With copper(l) iodide; potassium iodide; N,N`-dimethylethylenediamine In toluene at 130℃; for 24h; Rosenmund-von Braun reaction; | 98% |
| Stage #1: 2-Bromobiphenyl; sodium cyanide With potassium iodide; N,N`-dimethylethylenediamine; copper(l) iodide In toluene at 110 - 130℃; for 24h; Stage #2: With ammonia In water; ethyl acetate; toluene at 25℃; for 0.166667h; | 68% |

| Conditions | Yield |
|---|---|
| With tetrabutyl ammonium fluoride; palladium dichloride; tricyclohexylphosphine In water; dimethyl sulfoxide at 80℃; for 3h; | 98% |
| Stage #1: triallyl(phenyl)silane With tetrabutyl ammonium fluoride In water; dimethyl sulfoxide at 20℃; for 1h; Stage #2: 2-Bromobiphenyl With tricyclohexylphosphine; palladium dichloride In water; dimethyl sulfoxide at 80℃; for 3h; | 98% |
-
-
2052-07-5
2-Bromobiphenyl

-
-
40244-90-4
Chlorodiisopropylphosphane

| Conditions | Yield |
|---|---|
| Stage #1: 2-Bromobiphenyl With n-butyllithium at -78℃; Stage #2: Chlorodiisopropylphosphane at -78 - 20℃; Further stages.; | 98% |
| Stage #1: 2-Bromobiphenyl With n-butyllithium In diethyl ether; hexane at -10℃; for 3h; Schlenk technique; Inert atmosphere; Stage #2: Chlorodiisopropylphosphane In diethyl ether; hexane at -10 - 20℃; Schlenk technique; Inert atmosphere; | 68.1% |
| Stage #1: 2-Bromobiphenyl With n-butyllithium In diethyl ether; hexane at 0℃; for 2h; Stage #2: Chlorodiisopropylphosphane In diethyl ether; hexane at 20℃; for 1h; | |
| Stage #1: 2-Bromobiphenyl With n-butyllithium In diethyl ether; hexane at 0℃; for 2h; Stage #2: Chlorodiisopropylphosphane In diethyl ether; hexane at 20℃; for 1h; |

| Conditions | Yield |
|---|---|
| With tri-tert-butyl phosphine; bis(dibenzylideneacetone)-palladium(0); sodium t-butanolate In toluene at 20℃; for 7h; Inert atmosphere; | 98% |
| With tris-(dibenzylideneacetone)dipalladium(0); potassium tert-butylate In toluene at 0 - 20℃; for 7h; Inert atmosphere; | 98% |
| With tri-tert-butyl phosphine; palladium diacetate; sodium t-butanolate In toluene for 2h; Buchwald-Hartwig Coupling; Inert atmosphere; Reflux; | 92% |
-
-
109-12-6
2-aminopyrimidine

-
-
2052-07-5
2-Bromobiphenyl

-
-
1372776-06-1
N-([1,1'-biphenyl]-2-yl)pyrimidin-2-amine

| Conditions | Yield |
|---|---|
| With tris-(dibenzylideneacetone)dipalladium(0); 4,5-bis(diphenylphos4,5-bis(diphenylphosphino)-9,9-dimethylxanthenephino)-9,9-dimethylxanthene; sodium t-butanolate In toluene at 95℃; for 18h; Buchwald-Hartwig Coupling; Schlenk technique; Inert atmosphere; | 98% |
-
-
2052-07-5
2-Bromobiphenyl

-
-
84127-04-8
bis(4-methoxyphenyl)phosphine

| Conditions | Yield |
|---|---|
| Stage #1: bis(4-methoxyphenyl)phosphine With N,N’-dimethyl-2,6-bis(aminomethyl)pyridine; copper(l) iodide In toluene Glovebox; Schlenk technique; Inert atmosphere; Stage #2: 2-Bromobiphenyl With potassium tert-butylate In tetrahydrofuran; toluene at 100℃; for 24h; Schlenk technique; Sealed tube; | 98% |
-
-
2052-07-5
2-Bromobiphenyl

-
-
71075-24-6
(1R,2R,3R,4S)-bicyclo[2.2.1]hept-5-ene-2,3-diylbis(diphenylphosphane)dioxide

| Conditions | Yield |
|---|---|
| With tetrakis(triphenylphosphine) palladium(0); caesium carbonate In N,N-dimethyl-formamide at 105℃; for 48h; Schlenk technique; Inert atmosphere; regioselective reaction; | 98% |
-
-
2052-07-5
2-Bromobiphenyl

| Conditions | Yield |
|---|---|
| With bis-triphenylphosphine-palladium(II) chloride; potassium carbonate In ethanol; toluene for 12h; Schlenk technique; Inert atmosphere; Heating; | 98% |

| Conditions | Yield |
|---|---|
| Stage #1: 2-Bromobiphenyl; dibenzosuberenon With n-butyllithium In tetrahydrofuran at -78 - 20℃; Stage #2: With hydrogenchloride; acetic acid In water for 0.333333h; Heating; | 97% |
| Stage #1: 2-Bromobiphenyl With n-butyllithium In tetrahydrofuran; hexane at -78℃; for 0.5h; Stage #2: dibenzosuberenon In tetrahydrofuran; hexane for 6h; Stage #3: With hydrogenchloride; acetic acid In water for 12h; Reflux; | 94% |
| Stage #1: 2-Bromobiphenyl; dibenzosuberenon With tert.-butyl lithium In tetrahydrofuran; hexane at -78℃; for 6.5h; Stage #2: With hydrogenchloride; acetic acid In water for 12h; Reflux; | 94% |
| Stage #1: 2-Bromobiphenyl In tetrahydrofuran; hexane at -78℃; for 0.5h; Stage #2: dibenzosuberenon In tetrahydrofuran; hexane for 6h; Stage #3: With hydrogenchloride; acetic acid In water for 12h; Reflux; | 94% |
-
-
944129-32-2
3,7-dibromo-5H-dibenzo[a,d][7]annulen-5-one

-
-
2052-07-5
2-Bromobiphenyl

-
-
944129-33-3
3,7-dibromo-5,5-spirofluorenyl-5H-dibenzo[a,d]cycloheptene

| Conditions | Yield |
|---|---|
| Stage #1: 2-Bromobiphenyl With n-butyllithium In tetrahydrofuran; hexane at -78℃; for 0.5h; Stage #2: 3,7-dibromo-5H-dibenzo[a,d][7]annulen-5-one In tetrahydrofuran; hexane at -78 - 20℃; Stage #3: With acetic acid; hydrogenchloride for 0.5h; Heating; Further stages.; | 97% |
| Stage #1: 2-Bromobiphenyl With n-butyllithium In tetrahydrofuran; hexane at -78℃; for 0.5h; Inert atmosphere; Stage #2: 3,7-dibromo-5H-dibenzo[a,d][7]annulen-5-one In tetrahydrofuran; hexane at -78 - 20℃; Inert atmosphere; | 97% |
| Stage #1: 3,7-dibromo-5H-dibenzo[a,d][7]annulen-5-one; 2-Bromobiphenyl With n-butyllithium at -78 - 20℃; Stage #2: With hydrogenchloride; acetic acid for 8h; Reflux; | 95% |
| Stage #1: 2-Bromobiphenyl With n-butyllithium In tetrahydrofuran; hexane at -78℃; for 0.5h; Inert atmosphere; Stage #2: 3,7-dibromo-5H-dibenzo[a,d][7]annulen-5-one In tetrahydrofuran; hexane at -78 - 20℃; Inert atmosphere; Stage #3: With hydrogenchloride; acetic acid In water for 0.5h; Reflux; | 90% |
| Stage #1: 2-Bromobiphenyl With n-butyllithium In tetrahydrofuran at -78℃; for 1h; Inert atmosphere; Stage #2: 3,7-dibromo-5H-dibenzo[a,d][7]annulen-5-one In tetrahydrofuran at -78 - 20℃; Inert atmosphere; Stage #3: With hydrogenchloride; acetic acid In water for 1h; Reflux; | 90% |
-
-
686-69-1
diethylchlorophosphine

-
-
2052-07-5
2-Bromobiphenyl

-
-
1085850-83-4
[1,1'-biphenyl-2-yl]diethylphosphine

| Conditions | Yield |
|---|---|
| Stage #1: 2-Bromobiphenyl With n-butyllithium In diethyl ether at -10 - 0℃; for 3h; Stage #2: diethylchlorophosphine In diethyl ether at 20℃; Further stages.; | 97% |
| Stage #1: 2-Bromobiphenyl With n-butyllithium In diethyl ether; hexane at -10℃; for 3h; Schlenk technique; Inert atmosphere; Stage #2: diethylchlorophosphine In diethyl ether; hexane at -10 - 20℃; Schlenk technique; Inert atmosphere; | 70% |
| Stage #1: 2-Bromobiphenyl With n-butyllithium In diethyl ether; hexane at 0℃; for 2h; Stage #2: diethylchlorophosphine In diethyl ether; hexane at 20℃; for 1h; | |
| Stage #1: 2-Bromobiphenyl With n-butyllithium In diethyl ether; hexane at 0℃; for 2h; Stage #2: diethylchlorophosphine In diethyl ether at 20℃; for 1h; |

| Conditions | Yield |
|---|---|
| With methanol; Methyl formate; copper(l) chloride at 115℃; for 7h; Autoclave; Green chemistry; | 97% |

| Conditions | Yield |
|---|---|
| With 1,1'-bis-(diphenylphosphino)ferrocene; dichloro(1,1'-bis(diphenylphosphanyl)ferrocene)palladium(II)*CH2Cl2; sodium t-butanolate In toluene at 90℃; for 6h; Inert atmosphere; | 97% |
Related products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View
 Xi,
Xi,  Xn
Xn