-
Name
o-Xylene
- EINECS 202-422-2
- CAS No. 95-47-6
- Article Data469
- CAS DataBase
- Density 0.87 g/cm3
- Solubility miscible with ethanol, ether, acetone and benzene, insoluble in water
- Melting Point -26--23 °C
- Formula C8H10
- Boiling Point 145.949 °C at 760 mmHg
- Molecular Weight 106.167
- Flash Point 28.882 °C
- Transport Information UN 1307 3/PG 3
- Appearance colourless liquid
- Safety 25-45-36/37-16-7
- Risk Codes 10-20/21-38-39/23/24/25-23/24/25-11
-
Molecular Structure
-
Hazard Symbols
 Xn,
Xn, T,
T, F
F
- Synonyms o-Xylene;o-Xylene(8CI);1,2-Xylene;2-Methyltoluene;3,4-Dimethylbenzene;NSC 60920;Orthoxylene;o-Dimethylbenzene;o-Methyltoluene;o-Xylol;ortho-Xylene;
- PSA 0.00000
- LogP 2.30340
Synthetic route

-
-
13991-08-7
1,2-Bis(diphenylphosphino)benzene

-
A
-
85318-49-6
bis(1,2-bis(diphenylphosphino)benzene)palladium

-
B
-
95-47-6
o-xylene

| Conditions | Yield |
|---|---|
| In benzene-d6 under N2; in NMR tube; react. of Pd complex and ligand (excess) in benzene-d6 at 40 for 2 h or at 90°C for < 10 min; monitored by (1)H NMR spectra; | A n/a B 99% |


| Conditions | Yield |
|---|---|
| With palladium dichloride In methanol at 40℃; for 18h; Inert atmosphere; Green chemistry; chemoselective reaction; | 99% |
| With 2,4,6-trimethyl-pyridine; 4,4'-dimethoxyphenyl disulfide; iridium(lll) bis[2-(2,4-difluorophenyl)-5-methylpyridine-N,C20]-4,40-di-tert-butyl-2,20-bipyridine hexafluorophosphate; triphenylphosphine In toluene for 24h; Irradiation; | 66 %Chromat. |

| Conditions | Yield |
|---|---|
| With palladium dichloride In methanol at 40℃; for 1h; Green chemistry; chemoselective reaction; | 99% |
-
-
2808-75-5, 58396-30-8
2-methyl-1-methylenecyclohexane

-
-
95-47-6
o-xylene

| Conditions | Yield |
|---|---|
| With n-butyllithium; potassium 2-methylbutan-2-olate Mechanism; 1) r.t., 16 h, 2) reflux, 6 h; further reagent: D2O; | 97% |

| Conditions | Yield |
|---|---|
| With palladium/alumina; hydrogen In hexane at 325℃; Flow reactor; Green chemistry; | A 96.2% B 3.8% |
| With hydrogen In hexane at 325℃; Flow reactor; Green chemistry; | A 15.9% B 84.2% |
-
-
55262-10-7
1,2-dimethyl-2,5-cyclohexadiene-1-carboxylic acid

-
-
95-47-6
o-xylene

| Conditions | Yield |
|---|---|
| With chlorosulfonic acid In dichloromethane at 0℃; for 0.166667h; | 95% |

| Conditions | Yield |
|---|---|
| With hydrogen; aluminum oxide; nickel at 150℃; under 760 Torr; various Nitriles; | 92% |
| With nickel-copper at 240℃; Hydrogenation; |

| Conditions | Yield |
|---|---|
| With hydrogen; 5%-palladium/activated carbon at 290 - 320℃; for 1h; | 90% |
-
-
201230-82-2
carbon monoxide

-
-
612-14-6
phthalyl alcohol

-
A
-
4385-35-7
isochroman-3-one

-
B
-
95-47-6
o-xylene

-
C
-
644-36-0
o-methylphenylacetic acid

| Conditions | Yield |
|---|---|
| With hydrogen iodide; tetrakis(triphenylphosphine) palladium(0) In acetone at 90℃; under 68400 Torr; for 42h; Carbonylation; reduction; | A 88% B n/a C 9% |
| With hydrogen iodide; tetrakis(triphenylphosphine) palladium(0) In 1,4-dioxane at 120℃; under 68400 Torr; for 42h; Carbonylation; reduction; | A 13% B n/a C 52% |

-
-
7397-06-0
4-t-butyl-o-xylene

-
A
-
766-39-2
2,3-Dimethylmaleic anhydride

-
B
-
32703-79-0
4-tert-butylphthalic anhydride

-
C
-
74186-27-9
6-(tert-butyl)isobenzofuran-1(3H)-one

-
D
-
95-47-6
o-xylene

| Conditions | Yield |
|---|---|
| With oxygen; phosphorus; titanium; oxides of V; aluminium at 345℃; for 0.000119444h; Product distribution; oth. temperature, oth. time, gas phase; | A 3% B 83% C 1.8% D n/a E n/a |

-
-
18022-66-7
3,4-dimethylcyclohex-3-ene-1-carboxaldehyde

-
A
-
95-47-6
o-xylene

-
B
-
95-63-6
1,2,4-Trimethylbenzene

| Conditions | Yield |
|---|---|
| With hydrogen In hexane at 325℃; Flow reactor; Green chemistry; | A 17.5% B 80.9% |
| With palladium/alumina; hydrogen In hexane at 325℃; Flow reactor; Green chemistry; | A 73% B 27% |

| Conditions | Yield |
|---|---|
| With tetrakis(triphenylphosphine)palladium dichloride In benzene at 80℃; | A 80% B 13% |

-
-
104-87-0
4-methyl-benzaldehyde

-
-
552-45-4
1-chloromethyl-2-methylbenzene

-
A
-
95-47-6
o-xylene

-
B
-
589-18-4
4-Methylbenzyl alcohol

-
C
-
952-80-7
1,2-bis(2-methylphenyl)ethane

-
D
-
113312-69-9
1-(4-methylphenyl)-2-(2-methylphenyl)ethanol

| Conditions | Yield |
|---|---|
| With lithium In diethyl ether at 0℃; for 0.75h; Further byproducts given. Yields of byproduct given; | A n/a B n/a C n/a D 75% |


| Conditions | Yield |
|---|---|
| With magnesium at 600℃; | A 15% B 73% |

| Conditions | Yield |
|---|---|
| With polymethylhydrosiloxane; iron(III) chloride hexahydrate In 1,2-dichloro-ethane at 120℃; for 1h; Microwave irradiation; | 73% |
-
-
110-54-3
hexane

-
A
-
95-47-6
o-xylene

-
B
-
106-42-3
para-xylene

-
C
-
100-41-4
ethylbenzene

-
D
-
108-38-3
m-xylene

-
E
-
108-88-3
toluene

-
F
-
71-43-2
benzene

| Conditions | Yield |
|---|---|
| 540 degC; treated 16 h; U type zeolite at 450℃; Product distribution; | A n/a B n/a C n/a D n/a E 10.8% F 72.2% |
| 540 degC; treated 16 h; UHS type zeolite at 450℃; Product distribution; | A n/a B n/a C n/a D n/a E 33.3% F 50% |


| Conditions | Yield |
|---|---|
| With magnesium at 600℃; | A 20% B 70% |
-
-
89-92-9
2-methylbenzyl bromide

-
A
-
95-47-6
o-xylene

-
B
-
694-87-1
benzocyclobutene

-
C
-
952-80-7
1,2-bis(2-methylphenyl)ethane

-
D
-
36888-18-3
(E)-2,2'-dimethylstilbene

| Conditions | Yield |
|---|---|
| With magnesium at 600℃; under 0.15 Torr; | A n/a B n/a C 70% D n/a |

-
-
201230-82-2
carbon monoxide

-
-
89-92-9
2-methylbenzyl bromide

-
A
-
95-47-6
o-xylene

-
B
-
644-36-0
o-methylphenylacetic acid

-
C
-
952-80-7
1,2-bis(2-methylphenyl)ethane

| Conditions | Yield |
|---|---|
| With sodium hydroxide; cobalt tricarbonyl nitrosyl; trimethyldodecylammonium chloride In benzene under 760 Torr; Ambient temperature; | A 4% B 68% C 12% D 10% |

-
-
89-92-9
2-methylbenzyl bromide

-
A
-
95-47-6
o-xylene

-
B
-
644-36-0
o-methylphenylacetic acid

-
C
-
952-80-7
1,2-bis(2-methylphenyl)ethane

| Conditions | Yield |
|---|---|
| With sodium hydroxide; cobalt tricarbonyl nitrosyl; carbon monoxide; trimethyldodecylammonium chloride In benzene under 760 Torr; Ambient temperature; | A 4% B 68% C 12% D 10% |

-
-
55262-10-7
1,2-dimethyl-2,5-cyclohexadiene-1-carboxylic acid

-
A
-
95-47-6
o-xylene

| Conditions | Yield |
|---|---|
| With oxygen; 5,15,10,20-tetraphenylporphyrin In chloroform at -30℃; for 24h; Irradiation; | A 19% B 10% C 68% |

-
-
108-93-0
cyclohexanol

-
A
-
95-47-6
o-xylene

-
B
-
96-37-7
methyl-cyclopentane

-
C
-
110-82-7
cyclohexane

-
D
-
71-43-2
benzene

| Conditions | Yield |
|---|---|
| With hydrogen; nickel(II) oxide; aluminum oxide; molybdenum(VI) oxide at 450℃; under 21001.7 Torr; for 0.75h; Further byproducts given; | A n/a B 20% C 65% D n/a |

-
-
137092-35-4
Bis(dithiopivalinsaeure)-1,2-xylylenester

-
A
-
95-47-6
o-xylene

-
B
-
40920-16-9
methyl dithiopivalate

| Conditions | Yield |
|---|---|
| In methanol cathodic reduction, Pt cathode; | A 61% B 50% |
-
-
91-13-4
α,α'-dibromo-o-xylene

-
A
-
95-47-6
o-xylene

-
B
-
952-80-7
1,2-bis(2-methylphenyl)ethane

-
C
-
113245-36-6
α,α'-diethoxy-o-xylene

| Conditions | Yield |
|---|---|
| With sodium In toluene for 24h; Heating; | A 60% B n/a C n/a D n/a |


| Conditions | Yield |
|---|---|
| With tert-butylethylene; (iPr4PCOP)IrHCl; sodium t-butanolate at 200℃; for 120h; Concentration; Reagent/catalyst; Temperature; | A 11% B 60% C 7% |

-
-
142-82-5
n-heptane

-
A
-
95-47-6
o-xylene

-
B
-
106-42-3
para-xylene

-
C
-
100-41-4
ethylbenzene

-
D
-
108-38-3
m-xylene

-
E
-
108-88-3
toluene

-
F
-
71-43-2
benzene

| Conditions | Yield |
|---|---|
| 540 degC; treated 16 h; U type zeolite at 450℃; Product distribution; | A n/a B n/a C n/a D n/a E 58.5% F 6% |
| 540 degC; treated 16 h; UHS type zeolite at 450℃; Product distribution; | A n/a B n/a C n/a D n/a E 42.8% F 8.8% |


| Conditions | Yield |
|---|---|
| Stage #1: 3,4-Dimethylphenol With fluorosulfonyl fluoride; triethylamine In dimethyl sulfoxide at 20℃; for 3h; Stage #2: With formic acid; 1,3-bis-(diphenylphosphino)propane; palladium diacetate In dimethyl sulfoxide at 20℃; for 2h; | 58% |

| Conditions | Yield |
|---|---|
| With lithium; nickel dichloride In tetrahydrofuran at 20℃; Reduction; | 57% |
-
-
3227-91-6
<4>Radialene

-
A
-
292638-84-7
styrene

-
B
-
95-47-6
o-xylene

-
C
-
536-74-3
phenylacetylene

-
D
-
108-88-3
toluene

-
E
-
71-43-2
benzene

| Conditions | Yield |
|---|---|
| at 880℃; under 0.001 Torr; | A 55% B 6% C n/a D n/a E n/a |


| Conditions | Yield |
|---|---|
| With oxygen at 340 - 360℃; | 100% |
| With oxygen | 100% |
| With oxygen | 100% |

| Conditions | Yield |
|---|---|
| With hydrogen; [(norbornadiene)rhodium(I)chloride]2; phosphinated polydiacetylene In n-heptane at 30℃; under 60800 Torr; for 1.8h; | 100% |
| With Ti8O8(14+)*6C8H4O4(2-)*4O(2-)*3.3Li(1+)*0.7Co(2+)*0.7C4H8O*0.7H(1-); hydrogen In neat (no solvent) at 120℃; under 37503.8 Torr; for 18h; | 100% |
| With hydrogen In water at 100℃; under 15001.5 Torr; for 5h; | 99% |

| Conditions | Yield |
|---|---|
| With oxygen at 340 - 360℃; Product distribution / selectivity; | A 0.01% B 100% |
| With oxygen at 340 - 360℃; | A 0.1% B 100% |
| With oxygen at 340 - 360℃; | A 0.03% B 100% |
-
-
95-47-6
o-xylene

| Conditions | Yield |
|---|---|
| With chlorine; manganese trisacetylacetonate; 10H-phenothiazine-10-carbonyl chloride at 10℃; under 760.051 Torr; for 0.833333h; Product distribution / selectivity; | 100% |
-
-
95-47-6
o-xylene

| Conditions | Yield |
|---|---|
| With chlorine; manganese trisacetylacetonate; 10H-phenothiazine-10-carbonyl chloride at 10℃; under 760.051 Torr; for 0.666667h; Product distribution / selectivity; | 100% |

| Conditions | Yield |
|---|---|
| With iron(III) chloride; chloro-trimethyl-silane In 1,2-dichloro-ethane at 20℃; for 0.166667h; Friedel-Crafts Alkylation; Inert atmosphere; | 100% |

| Conditions | Yield |
|---|---|
| With phosphotungstic acid at 120℃; for 3h; Reagent/catalyst; Friedel-Crafts Alkylation; | 100% |
-
-
95-47-6
o-xylene

| Conditions | Yield |
|---|---|
| With trifluorormethanesulfonic acid In dichloromethane at -40℃; for 6h; | 100% |

| Conditions | Yield |
|---|---|
| With cross-linked poly acidic ionic liquids In N,N-dimethyl-formamide at 120℃; for 3h; Solvent; Temperature; High pressure; | 99.67% |
| With phosphotungstic acid at 120℃; for 3h; Reagent/catalyst; | 98.3% |
| With 12-tungstophosphoric acid supported 30percent triethylammonium acetate, P123 in micro/mesoporous silica at 119.84℃; for 3h; Catalytic behavior; Reagent/catalyst; Temperature; Friedel-Crafts Alkylation; | 93.8% |

| Conditions | Yield |
|---|---|
| With iodine at 50℃; Temperature; | 99.3% |
| With iron(III) chloride at 0 - 20℃; | 85% |
| With aluminium trichloride | 69% |

| Conditions | Yield |
|---|---|
| With aluminum (III) chloride In neat (no solvent) for 1h; Friedel-Crafts Acylation; Milling; | 99% |
| With [bmim]Cl*AlCl3 at 30℃; for 3h; Friedel-Crafts reaction; | 95% |
| With aluminium trichloride for 0.166667h; Heating; | 37% |
-
-
95-47-6
o-xylene

-
-
625-36-5
2-chloropropionyl chloride

-
-
22422-23-7
3-chloro-1-(3,4-dimethyl-phenyl)-propan-1-one

| Conditions | Yield |
|---|---|
| With aluminum (III) chloride; nitromethane at 19 - 22℃; under 760.051 Torr; Friedel Crafts acylation; Inert atmosphere; | 99% |
| With aluminum (III) chloride In dichloromethane at 0 - 20℃; Friedel-Crafts acylation; Inert atmosphere; | 98% |
| With aluminum (III) chloride In dichloromethane at 25℃; for 3h; | 96% |
-
-
95-47-6
o-xylene

-
-
25145-43-1, 36240-11-6, 59981-93-0, 22414-26-2
2-phenylpropionyl chloride

-
-
135584-30-4
1-(3,4-dimethylphenyl)-2-phenyl-1-propanone

| Conditions | Yield |
|---|---|
| With aluminium trichloride In carbon disulfide for 14h; Ambient temperature; | 99% |
-
-
95-47-6
o-xylene

| Conditions | Yield |
|---|---|
| With chlorine; manganese trisacetylacetonate; 10H-phenothiazine-10-carbonyl chloride at 10℃; under 760.051 Torr; for 0.583333h; Product distribution / selectivity; | 99% |
-
-
95-47-6
o-xylene

-
-
199620-14-9, 13007-92-6
chromium(0) hexacarbonyl

-
-
12129-29-2
(η6-o-xylene)tricarbonylchromium(0)

| Conditions | Yield |
|---|---|
| In tetrahydrofuran; dibutyl ether refluxing for 1 wk; evapn. of solvents under vac., removing unreacted (Cr(CO)6) by sublimation, recrystn. from CH2Cl2 under N2; | 99% |
| With catalyst: CH3COOC4H9 In decalin byproducts: CO; (Ar); refluxed for 3 h; cooled to 20°C; filtered through Kieselguhr; catalyst evapd.; chromy.; evapd.; crystd. from benzene/pertoleum ether at -18°C; | 98% |
| With catalyst: butyl acetate In decalin byproducts: CO; refluxing for 2.5 h (catalyst: butyl acetate); freezing out at -18°C or quick chromy. of the decaline soln. on a SiO2 column; | 98% |

| Conditions | Yield |
|---|---|
| In o-xylene Schlenkware or glovebox; o-xylene was added to solid carborane acid; mixt. was stirred for few min; solvent removed (vac.); elem. anal.; | 99% |
-
-
95-47-6
o-xylene

-
-
18734-63-9
perfluoro-o-phenylenemercury trimer

-
-
649559-06-8
perfluoro-ortho-phenylene mercury trimer - ortho-xylene (1:1)

| Conditions | Yield |
|---|---|
| In further solvent(s) Hg-compound was dissolved by boiling in o-xylene, cooled; slow evapd. in a well-aerated fume hood; elem. anal.; | 99% |

| Conditions | Yield |
|---|---|
| With tetra-(n-butyl)ammonium iodide In water at 80℃; for 3h; | 99% |
| With di-tert-butyl peroxide; C11H23N2(1+)*Br4Fe(1-) at 110℃; for 24h; | 99% |
| With tert.-butylhydroperoxide; choline chloride; tetra-(n-butyl)ammonium iodide; urea In water at 80℃; for 12h; Green chemistry; | 90% |
-
-
95-47-6
o-xylene

-
-
3972-65-4
1-bromo-4-tert-butylbenzene

-
-
70663-11-5
1-(4-(tert-butyl) benzyl)-2-methylbenzene

| Conditions | Yield |
|---|---|
| With Xantphos-Pd-G3; potassium hexamethylsilazane at 110℃; for 12h; Inert atmosphere; Sealed tube; | 99% |
-
-
677-71-4
hexafluoroacetone hydrate

-
-
95-47-6
o-xylene

-
-
65294-20-4
4,4'-(hexafluoroisopropylidene)-bis-(o-xylene)

| Conditions | Yield |
|---|---|
| With hydrogen fluoride at 100℃; under 7980.54 Torr; for 13h; Concentration; Temperature; Autoclave; | 98.7% |

| Conditions | Yield |
|---|---|
| With aluminum (III) chloride In dichloromethane at 20℃; for 2h; Friedel-Crafts Acylation; | 98% |
| With [bmim]Cl*AlCl3 at 30℃; for 3h; Friedel-Crafts reaction; | 95% |
| With aluminium trichloride In dichloromethane | 86% |

| Conditions | Yield |
|---|---|
| With H5CoW12O40 supported on rice husk ash extracted nano silica at 60℃; for 0.5h; Reagent/catalyst; Green chemistry; | 98% |
| With boron trifluoride diethyl etherate; water at 80℃; for 2h; regioselective reaction; | 93% |
| With (diethylamino)difluorosulfonium tetrafluoroborate In dichloromethane at 20℃; for 48h; Inert atmosphere; | 89% |
| With phosphorus pentoxide | |
| With 1-butyl-3-methylimidazolium tetrachloridoferrate(III) at 80℃; for 12h; Friedel-Crafts type benzylation; regioselective reaction; | 56 %Chromat. |

| Conditions | Yield |
|---|---|
| With bromine In tetrachloromethane Solvent; | 98% |
| With bromine In tetrachloromethane for 1.5h; Ambient temperature; | 97% |
| With 2,4,4,6-Tetrabromo-2,5-cyclohexadien-1-one; silica gel for 4.5h; UV-irradiation; | 82% |

| Conditions | Yield |
|---|---|
| With potassium carbonate; palladium on activated charcoal at 120℃; for 12h; | 98% |
| With zinc(II) chloride Substitution; Heating; | 92% |
| With zinc(II) chloride for 10h; Heating; | 90% |
| With molybdenum hexacarbonyl at 120℃; for 2h; Sealed tube; regioselective reaction; | 72% |

| Conditions | Yield |
|---|---|
| With pyrographite for 24h; Heating; | 98% |

| Conditions | Yield |
|---|---|
| pyrographite for 24h; Heating; | 98% |
-
-
95-47-6
o-xylene

| Conditions | Yield |
|---|---|
| With C42H63O3P*Au(1+)*C7H5N*F6Sb(1-) at 20℃; for 0.05h; Schlenk technique; | 98% |
1,2-Dimethylbenzene Consensus Reports
1,2-Dimethylbenzene Standards and Recommendations
ACGIH TLV: TWA 100 ppm; STEL 150 ppm; BEI: methyl hippuric acids in urine at end of shift 1.5 g/g creatinine; Not Classifiable as a Human Carcinogen
NIOSH REL: (Xylene) TWA 100 ppm; CL 200 ppm/10M
DOT Classification: 3; Label: Flammable Liquid
1,2-Dimethylbenzene Specification
1,2-Dimethylbenzene, also known o-Xylene, is colourless, flammable liquid with aromatic smell. It is an aromatic hydrocarbon, based on benzene with two methyl substituents bonded to adjacent carbon atoms in the aromatic ring (the ortho configuration). What's more, 1,2-Dimethylbenzene is miscible with ethanol, ether, acetone and benzene, insoluble in water.
Preparation: 1,2-Dimethylbenzene was mainly obtained from coal tar originally, but now the majority of domestic and overseas extract this product from oil catalytic reforming and aromatics thermal cracking. As the structure of 1,2-Dimethylbenzene, p-xylene, m-xylene is very similar, their physical parameters vary little. In industry, the separation of 1,2-Dimethylbenzene uses superfractionation. The rectification requires about 150 tower plates and reflux ratio 5-8 and consumes more energy.
Use industrial 1,2-dimethylbenzene as raw material. First, use industrial concentrated sulfuric acid to wash to acid layer colorless. Then wash the mixture with 10% sodium hydroxide solution and water to be qualified. After the separation of water layer, dry with anhydrous calcium chloride. And then rectify until distillate clear. Finally, collecting the midbarrel, the product is pure.
Uses: 1,2-Dimethylbenzene is mainly used as chemical raw material and solvent. It can be used in the production of phthalic anhydride, phthalic nitrile, dimethyl phenol, dimethylaniline, dyes, pesticide, drug (such as vitamin) and so on. It is also used as aviation gasoline additive. In addition, 1,2-Dimethylbenzene is used in organic synthesis. For example: it can react with benzoyl chloride to get 3,4-dimethyl-benzophenone. This reaction which is a kind of Friedel-Crafts acylation needs reagent BiCl3 at temperature of 120 °C. The reaction time is 12 hours. The yield is 93%.
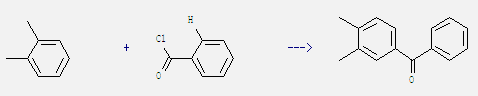
Safty: 1,2-Dimethylbenzene is harmful by inhalation and in contact with skin. It is specially irritating to skin. And people also should avoid contact with eyes. If you want to contact this product, you must wear suitable protective clothing and gloves. In case of accident or if you feel unwell, seek medical advice immediately (show the label whenever possible.)
Structure Descriptors:
1. Smiles:c1(c(cccc1)C)C
2. InChI:InChI=1/C8H10/c1-7-5-3-4-6-8(7)2/h3-6H,1-2H3
Toxicity:
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| human | LCLo | inhalation | 6125ppm/12H (6125ppm) | Gekkan Yakuji. Pharmaceuticals Monthly. Vol. 22, Pg. 883, 1980. | |
| mammal (species unspecified) | LDLo | intraperitoneal | 1500mg/kg (1500mg/kg) | Journal of Pathology and Bacteriology. Vol. 46, Pg. 95, 1938. | |
| mammal (species unspecified) | LDLo | subcutaneous | 2500mg/kg (2500mg/kg) | Journal of Pathology and Bacteriology. Vol. 46, Pg. 95, 1938. | |
| mouse | LCLo | inhalation | 30gm/m3 (30000mg/m3) | BEHAVIORAL: GENERAL ANESTHETIC LUNGS, THORAX, OR RESPIRATION: RESPIRATORY DEPRESSION CARDIAC: OTHER CHANGES | Naunyn-Schmiedeberg's Archiv fuer Experimentelle Pathologie und Pharmakologie. Vol. 143, Pg. 223, 1929. |
| mouse | LD50 | intraperitoneal | 1550uL/kg (1.55mL/kg) | Archives of Toxicology. Vol. 58, Pg. 106, 1985. | |
| rat | LCLo | inhalation | 6125ppm/12H (6125ppm) | Journal of Pathology and Bacteriology. Vol. 46, Pg. 95, 1938. | |
| rat | LDLo | oral | 5gm/kg (5000mg/kg) | Gekkan Yakuji. Pharmaceuticals Monthly. Vol. 22, Pg. 883, 1980. |
Related Products
- 10,10'-Bis([1,1'-biphenyl]-4-yl)-9,9'-bianthracene
- 10,10'-Dibromo-9,9'-bianthryl
- 10,10-Dimethylanthrone
- 10,10-Oxybisphenoxarsine
- 10-[1,1'-Biphenyl]-4-yl-2-(1-methylethyl)-9-oxo-9H-thioxanthenium hexafluorophosphate
- 10,11-Dehydroimipramine
- 10,11-Dihydro-11-oxodibenzo[b,f][1,4]thiazepine
- 10,11-Dihydro-2-(4-methyl-1-piperazinyl)-11-(2-athiazolyl)-pyridazino(3,4-b)(1,4)benzoxazepine
- 10,11-DIHYDRO-2-(4-METHYL-1-PIPERAZINYL)-11-(3,4-XYLYL)PYRIDAZINO(3,4b)(1,4)-BENZOXAZEPINE MALEATE
- 10,11-Dihydro-5-(3-dimethylamino-2-methylpropyl)-5h-dibenz (b,f)azepine
- 95480-29-8
- 95480-80-1
- 954-81-4
- 95481-62-2
- 95-48-7
- 95-49-8
- 95-50-1
- 955016-25-8
- 955028-67-8
- 955028-82-7
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View