-
Name
1-Iodo-4-nitrobenzene
- EINECS 211-272-7
- CAS No. 636-98-6
- Article Data147
- CAS DataBase
- Density 2.019 g/cm3
- Solubility insoluble in water
- Melting Point 171-173 °C(lit.)
- Formula C6H4INO2
- Boiling Point 287.959 °C at 760 mmHg
- Molecular Weight 249.008
- Flash Point 127.953 °C
- Transport Information UN 2811 6.1/PG 2
- Appearance solid
- Safety 26-36/37/39
- Risk Codes 20/21/22-33-36
-
Molecular Structure
-
Hazard Symbols
 Xn,
Xn, Xi
Xi
- Synonyms 4-Iodo-1-nitrobenzene;4-Iodonitrobenzene;4-Nitro-1-iodobenzene;4-Nitroiodobenzene;4-Nitrophenyl iodide;NSC 9794;p-Iodonitrobenzene;p-Nitroiodobenzene;p-Nitrophenyl iodide;
- PSA 45.82000
- LogP 2.72260
Synthetic route

| Conditions | Yield |
|---|---|
| With potassium iodide; 10-camphorsulfonic acid; sodium nitrite In acetic acid at 20℃; for 24h; Time; | 100% |
| With Nitrogen dioxide; sodium iodide In acetonitrile at -20℃; for 10h; | 98% |
| Stage #1: 4-nitro-aniline With toluene-4-sulfonic acid In water at 20℃; Stage #2: With potassium iodide In water at 20℃; for 1.5h; | 95% |

-
-
636-98-6
p-nitrobenzene iodide

| Conditions | Yield |
|---|---|
| With potassium iodide for 24h; | 100% |

| Conditions | Yield |
|---|---|
| With iodine; dibenzyl hantzsch ester In dimethyl sulfoxide at 20℃; | 99% |
| With trimethylsilyl iodide; methyl iodide In N,N-dimethyl-formamide for 1h; Ambient temperature; | 91% |
| With iodine; potassium iodide In dimethyl sulfoxide at 15℃; |
-
-
52010-82-9
1-(4-nitrophenyl)-2-(pyrrolidin-1-yl)diazene

-
-
636-98-6
p-nitrobenzene iodide

| Conditions | Yield |
|---|---|
| With iodine In various solvent(s) at 100℃; for 12h; | 98% |
| With trifluoroacetic acid; potassium iodide In water Yield given; |

-
A
-
636-98-6
p-nitrobenzene iodide

-
B
-
100-25-4
para-dinitrobenzene

-
C
-
591-50-4
iodobenzene

-
D
-
98-95-3
nitrobenzene

| Conditions | Yield |
|---|---|
| With sodium nitrite In chloroform; water at 56℃; for 1h; | A 1% B 98% C 93 % Chromat. D 1 % Chromat. |


| Conditions | Yield |
|---|---|
| With potassium iodide In water at 20℃; for 0.0833333h; | 98% |
| With potassium iodide In water for 0.333333h; paste form; |

-
-
636-98-6
p-nitrobenzene iodide

| Conditions | Yield |
|---|---|
| With potassium iodide In water at 20℃; | 98% |
-
-
591-50-4
iodobenzene

-
-
122-04-3
4-nitro-benzoyl chloride

-
A
-
636-98-6
p-nitrobenzene iodide

-
B
-
98-88-4
benzoyl chloride

| Conditions | Yield |
|---|---|
| With (Xantphos)Pd(4-C6H4NO2)(I) In benzene at 90℃; for 20h; Sealed tube; Inert atmosphere; | A n/a B 95% |

-
-
696-62-8
para-iodoanisole

-
-
122-04-3
4-nitro-benzoyl chloride

-
A
-
636-98-6
p-nitrobenzene iodide

-
B
-
100-07-2
4-methoxy-benzoyl chloride

| Conditions | Yield |
|---|---|
| With (Xantphos)Pd(4-C6H4NO2)(I) In benzene at 90℃; for 20h; Kinetics; Solvent; Reagent/catalyst; Temperature; Sealed tube; Inert atmosphere; | A n/a B 95% |

-
-
624-38-4
para-diiodobenzene

-
-
122-04-3
4-nitro-benzoyl chloride

-
A
-
636-98-6
p-nitrobenzene iodide

-
B
-
1711-02-0
4-iodobenzoic acid chloride

| Conditions | Yield |
|---|---|
| With (Xantphos)Pd(4-C6H4NO2)(I) In benzene at 90℃; for 20h; Sealed tube; Inert atmosphere; | A n/a B 95% |

-
-
624-31-7
4-tolyl iodide

-
-
122-04-3
4-nitro-benzoyl chloride

-
A
-
636-98-6
p-nitrobenzene iodide

-
B
-
874-60-2
4-methyl-benzoyl chloride

| Conditions | Yield |
|---|---|
| With (Xantphos)Pd(4-C6H4NO2)(I) In benzene at 90℃; for 20h; Sealed tube; Inert atmosphere; | A n/a B 94% |

-
-
51934-41-9
4-iodobenzoic acid ethyl ester

-
-
122-04-3
4-nitro-benzoyl chloride

-
A
-
636-98-6
p-nitrobenzene iodide

-
B
-
27111-45-1
ethyl 4-(chlorocarbonyl)benzoate

| Conditions | Yield |
|---|---|
| With (Xantphos)Pd(4-C6H4NO2)(I) In benzene at 90℃; for 20h; Sealed tube; Inert atmosphere; | A n/a B 94% |

-
-
637-87-6
1-Chloro-4-iodobenzene

-
-
122-04-3
4-nitro-benzoyl chloride

-
A
-
636-98-6
p-nitrobenzene iodide

-
B
-
122-01-0
4-chloro-benzoyl chloride

| Conditions | Yield |
|---|---|
| With (Xantphos)Pd(4-C6H4NO2)(I) In benzene at 90℃; for 20h; Sealed tube; Inert atmosphere; | A n/a B 93% |

-
-
589-87-7
1,4-bromoiodobenzene

-
-
122-04-3
4-nitro-benzoyl chloride

-
A
-
636-98-6
p-nitrobenzene iodide

-
B
-
586-75-4
4-chlorobenzoyl chloride

| Conditions | Yield |
|---|---|
| With (Xantphos)Pd(4-C6H4NO2)(I) In benzene at 90℃; for 20h; Sealed tube; Inert atmosphere; | A n/a B 93% |

-
-
625-95-6
3-Iodotoluene

-
-
122-04-3
4-nitro-benzoyl chloride

-
A
-
636-98-6
p-nitrobenzene iodide

-
B
-
1711-06-4
3-Methylbenzoyl chloride

| Conditions | Yield |
|---|---|
| With (Xantphos)Pd(4-C6H4NO2)(I) In benzene at 90℃; for 20h; Sealed tube; Inert atmosphere; | A n/a B 93% |


-
-
636-98-6
p-nitrobenzene iodide

| Conditions | Yield |
|---|---|
| With tetra-(n-butyl)ammonium iodide In acetonitrile at 20℃; for 0.75h; Substitution; | 92% |
-
-
122-04-3
4-nitro-benzoyl chloride

-
-
5876-51-7
5-iodo-1,3-benzodioxole

-
A
-
636-98-6
p-nitrobenzene iodide

-
B
-
25054-53-9
3,4-(methylenedioxy)benzoyl chloride

| Conditions | Yield |
|---|---|
| With (Xantphos)Pd(4-C6H4NO2)(I) In benzene at 90℃; for 20h; Sealed tube; Inert atmosphere; | A n/a B 92% |


| Conditions | Yield |
|---|---|
| With tris-(dibenzylideneacetone)dipalladium(0); 4,5-bis(diphenylphos4,5-bis(diphenylphosphino)-9,9-dimethylxanthenephino)-9,9-dimethylxanthene In toluene at 100℃; for 12h; | 91% |
| With 2,2'-azobis(isobutyronitrile); 2-mercaptopyridine-1-oxide sodium salt; methyl iodide In toluene for 0.583333h; Heating; Yield given; |
-
-
352-34-1
4-fluoro-1-iodobenzene

-
-
122-04-3
4-nitro-benzoyl chloride

-
A
-
636-98-6
p-nitrobenzene iodide

-
B
-
403-43-0
4-fluorobenzoyl chloride

| Conditions | Yield |
|---|---|
| With (Xantphos)Pd(4-C6H4NO2)(I) In benzene at 90℃; for 20h; Sealed tube; Inert atmosphere; | A n/a B 91% |

-
-
22445-41-6
3,5-dimethylphenyl iodide

-
-
122-04-3
4-nitro-benzoyl chloride

-
A
-
636-98-6
p-nitrobenzene iodide

-
B
-
6613-44-1
3,5-dimethylbenzoyl chloride

| Conditions | Yield |
|---|---|
| With (Xantphos)Pd(4-C6H4NO2)(I) In benzene at 90℃; for 20h; Sealed tube; Inert atmosphere; | A n/a B 91% |

-
-
1121-86-4
1-Fluoro-3-iodobenzene

-
-
122-04-3
4-nitro-benzoyl chloride

-
A
-
636-98-6
p-nitrobenzene iodide

-
B
-
1711-07-5
3-fluorobenzoyl chloride

| Conditions | Yield |
|---|---|
| With (Xantphos)Pd(4-C6H4NO2)(I) In benzene at 90℃; for 20h; Sealed tube; Inert atmosphere; | A n/a B 91% |


| Conditions | Yield |
|---|---|
| With 1,1,1,3',3',3'-hexafluoro-propanol; N–nitrosaccharin at 60℃; for 19h; Inert atmosphere; Schlenk technique; | 91% |
-
-
122-04-3
4-nitro-benzoyl chloride

-
-
214701-35-6
2-(4-iodophenyl)-2-methyl-[1,2]-dioxolane

-
A
-
636-98-6
p-nitrobenzene iodide

| Conditions | Yield |
|---|---|
| With (Xantphos)Pd(4-C6H4NO2)(I) In benzene at 90℃; for 20h; Sealed tube; Inert atmosphere; | A n/a B 90% |

-
-
92722-14-0
tert-butyl 2-(4-nitrophenyl)diazenecarboxylate

-
-
636-98-6
p-nitrobenzene iodide

| Conditions | Yield |
|---|---|
| With iodine; trifluoroacetic acid In acetonitrile at 80℃; for 0.5h; | 89% |
| With iodine; trifluoroacetic acid In acetonitrile at 80℃; | 89% |
| With iodine; trifluoroacetic acid In acetonitrile at 80℃; for 4h; | 80% |

| Conditions | Yield |
|---|---|
| With hydrogen iodide In acetonitrile at 60℃; for 0.25h; | 88% |
-
-
82113-65-3
bis(trifluoromethanesulfonyl)amide

-
-
100-01-6
4-nitro-aniline

-
-
636-98-6
p-nitrobenzene iodide

| Conditions | Yield |
|---|---|
| Stage #1: bis(trifluoromethanesulfonyl)amide With tert.-butylnitrite; acetic acid In ethanol Cooling with ice; Stage #2: 4-nitro-aniline With tetraethylammonium iodide In water at 20℃; | 88% |

| Conditions | Yield |
|---|---|
| With tributylphosphine; sodium iodide; nickel dibromide In 1-methyl-pyrrolidin-2-one at 180℃; for 3h; Finkelstein reaction; Inert atmosphere; Molecular sieve; Sealed tube; | 87% |
| With copper(I) oxide; L-proline; potassium iodide In ethanol at 110℃; for 30h; Schlenk technique; Inert atmosphere; Sealed tube; | 85% |
| With KI-CuI-alumina In various solvent(s) at 150℃; for 6h; | 78 % Chromat. |
| With copper(l) iodide; 3-azapentane-1,5-diamine; sodium iodide In acetonitrile at 110℃; for 24h; Finkelstein Reaction; Inert atmosphere; | 60 %Chromat. |
-
-
24067-17-2
4-nitrophenylboronic acid

-
-
636-98-6
p-nitrobenzene iodide

| Conditions | Yield |
|---|---|
| With perfluoroisopropyl iodide; copper; hydroquinone In N,N-dimethyl-formamide at 20℃; for 24h; | 87% |
| With sodium nitrite In acetonitrile at 80℃; for 8h; Sealed tube; | 87% |
-
-
122-04-3
4-nitro-benzoyl chloride

-
-
174715-24-3
5-iodo-1-(4-methylphenylsulfonyl)indole

-
A
-
636-98-6
p-nitrobenzene iodide

| Conditions | Yield |
|---|---|
| With (Xantphos)Pd(4-C6H4NO2)(I) In benzene at 90℃; for 20h; Sealed tube; Inert atmosphere; | A n/a B 85% |

-
-
3058-39-7
4-iodobenzonitrile

-
-
122-04-3
4-nitro-benzoyl chloride

-
A
-
636-98-6
p-nitrobenzene iodide

-
B
-
6068-72-0
4-cyanobenzoyl chlorIde

| Conditions | Yield |
|---|---|
| With (Xantphos)Pd(4-C6H4NO2)(I) In benzene at 90℃; for 20h; Sealed tube; Inert atmosphere; | A n/a B 82% |


| Conditions | Yield |
|---|---|
| With hydrazine hydrate In methanol at 26℃; for 0.166667h; Reagent/catalyst; Solvent; | 100% |
| With cadmium sulphide; ammonium formate In water at 20℃; for 20h; Inert atmosphere; Irradiation; Sealed tube; | 100% |
| With magnesium; nickel dichloride In water at 20℃; for 12h; Sealed tube; Photolysis; Inert atmosphere; | 100% |

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 50℃; under 9000720 Torr; for 20h; | 100% |
| With copper(l) iodide; 1-(5,6,7,8-tetrahydroquinolin-8-yl)ethan-1-one; caesium carbonate In N,N-dimethyl-formamide at 25℃; for 24h; Ullmann condensation; Inert atmosphere; | 95% |
| With copper(l) iodide In N,N-dimethyl acetamide at 130℃; for 24h; | 95% |

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 50℃; under 9000720 Torr; for 20h; | 100% |
| With potassium carbonate In dimethyl sulfoxide at 50℃; | 100% |
| With sodium acetate In N,N-dimethyl-formamide for 8h; | 95% |

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 50℃; under 9000720 Torr; for 50h; | 100% |
| With [Pd{C6H4(CH2N(CH2Ph)2)}(μ-Br)]2; potassium hydroxide In dimethyl sulfoxide at 120℃; for 0.166667h; | 98% |
| With sodium t-butanolate In dimethyl sulfoxide at 110℃; for 12h; Buchwald-Hartwig Coupling; | 96% |
-
-
280-57-9
1,4-diaza-bicyclo[2.2.2]octane

-
-
636-98-6
p-nitrobenzene iodide

-
-
112473-28-6
1-(4-Nitro-phenyl)-4-aza-1-azonia-bicyclo[2.2.2]octane; iodide

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 50℃; under 4500360 Torr; for 75h; | 100% |
| In tetrahydrofuran at 50℃; under 4500360 Torr; for 20h; Yield given; |

| Conditions | Yield |
|---|---|
| With copper(l) iodide; iodophenylbis(triphenylphosphine)palladium; triethylamine In chloroform at 60℃; for 1h; | 100% |
| With bis-triphenylphosphine-palladium(II) chloride; copper(l) iodide; tributyl-amine; potassium carbonate In water at 20℃; for 4h; | 95% |
| With piperidine; 1,3-bis(5-ferrocenylisoxazoline-3-yl)benzene; tetrabutylammomium bromide; palladium diacetate In water; N,N-dimethyl-formamide at 40℃; for 3h; Sonogashira Cross-Coupling; | 94% |

| Conditions | Yield |
|---|---|
| With potassium carbonate; bis-triphenylphosphine-palladium(II) chloride In water; N,N-dimethyl-formamide at 50℃; under 760 Torr; for 3h; | 100% |
| With amphiphilic resin-supported phosphine-palladium; water; potassium carbonate at 25℃; under 760 Torr; for 12h; hydroxycarbonylation; | 100% |
| With potassium carbonate; bis-triphenylphosphine-palladium(II) chloride In water; N,N-dimethyl-formamide at 50℃; for 3h; Product distribution; effect of different catalysts; | 100% |

| Conditions | Yield |
|---|---|
| With dichloro bis(acetonitrile) palladium(II) In N,N-dimethyl-formamide at 20℃; for 3h; | 100% |
| With Palladium nanoparticle cored dendrimer In tetrahydrofuran for 4h; Stille coupling; Reflux; | 96% |
| With (4-nitrophenyl)Pd(P(C6H5)3)2I In N,N,N,N,N,N-hexamethylphosphoric triamide at 70℃; for 3.5h; Product distribution; other solvents; also in DMF in vacuum, in the air, and in O2 at various reaction times; | 94 % Chromat. |
-
-
636-98-6
p-nitrobenzene iodide

-
-
201230-82-2
carbon monoxide

-
-
1067-21-6
triethylmethoxystannane

-
-
619-50-1
4-nitrobenzoic acid methyl ester

| Conditions | Yield |
|---|---|
| With iodophenylbis(triphenylphosphine)palladium under 760 Torr; for 1h; | 100% |


| Conditions | Yield |
|---|---|
| With bis(triethyltin) sulfide; iodophenylbis(triphenylphosphine)palladium In dimethyl sulfoxide for 4h; Heating; | 100% |
| With potassium thioacyanate; cetyltrimethylammonim bromide; potassium hydroxide In water at 135℃; for 7h; Green chemistry; | 100% |
| With copper(l) iodide; sodiumsulfide nonahydrate; potassium carbonate In N,N-dimethyl-formamide at 120℃; for 18h; Inert atmosphere; | 98% |

| Conditions | Yield |
|---|---|
| With 1-Benzyl-1,4-dihydronicotinamide; RhCl(PPh3)3 In acetonitrile at 70℃; for 3h; Product distribution; other organic halides; var. times, var. temp., also in the presence of PNAH as reductant and Pd(OAc)2 as catalyst; | 100% |
| With (1,1'-bis(diphenylphosphino)ferrocene)palladium(II) dichloride; cesium fluoride In 2-pentanol at 100℃; for 6h; Inert atmosphere; | 100% |
| With 18-crown-6 ether In diethyl ether; N,N-dimethyl-formamide for 4h; Heating; | 84% |
-
-
636-98-6
p-nitrobenzene iodide

-
-
37496-13-2
2-(trimethylstannyl)thiophene

-
-
59156-21-7
2-(4-nitrophenyl)thiophene

| Conditions | Yield |
|---|---|
| With lithium chloropalladite In N,N,N,N,N,N-hexamethylphosphoric triamide at 20℃; for 0.5h; other reagents; | 100% |

| Conditions | Yield |
|---|---|
| With copper(l) iodide; triethylamine; titanium(IV) oxide at 20℃; for 24h; | 100% |
| With 1,4-diaza-bicyclo[2.2.2]octane; caesium carbonate; palladium diacetate In acetonitrile at 20℃; for 6h; Sonogashira cross-coupling reaction; | 100% |
| With 1,4-diaza-bicyclo[2.2.2]octane; air; palladium diacetate In acetonitrile at 20℃; for 18h; Sonogashira reaction; | 100% |
-
-
636-98-6
p-nitrobenzene iodide

-
-
292638-85-8
acrylic acid methyl ester

-
-
637-57-0
p-nitrocinnamic acid methyl ester

| Conditions | Yield |
|---|---|
| With triethylamine; fluorine-substituted palladium In N,N-dimethyl-formamide at 140℃; for 3h; Heck reaction; | 100% |
| With [P5(η3-allyl)(4,4'-(CF3(CF2)9CH2OCH2)-2,2'-bpy)][OTf]; triethylamine In N,N-dimethyl-formamide at 145℃; for 0.25h; Catalytic behavior; Reagent/catalyst; Heck Reaction; Microwave irradiation; | 100% |
| With palladium diacetate; triethylamine In toluene at 110℃; for 2.5h; Heck reaction; | 99% |

| Conditions | Yield |
|---|---|
| With [PdCl2(2-ethyl-2-oxazoline)2]; sodium acetate In N,N-dimethyl-formamide at 140 - 150℃; for 24h; Heck reaction; | 100% |
| With tetrabutyl ammonium fluoride; palladium dichloride at 40℃; for 12h; Heck reaction; Inert atmosphere; neat (no solvent); | 100% |
| With tributyl-amine In N,N-dimethyl-formamide at 120℃; for 24h; Heck-Mizoroki reaction; stereoselective reaction; | 99% |

| Conditions | Yield |
|---|---|
| With palladium diacetate; 4-Ph2P-C6H4-2-N-glucosamine; sodium carbonate In water at 60℃; for 1h; Product distribution; Further Variations:; Temperatures; Reagents; Suzuki cross-coupling reaction; | 100% |
| With polyhydroxylated phosphine; sodium carbonate; palladium diacetate In ethanol; water; toluene at 60℃; for 1h; | 100% |
| With 1,4-diaza-bicyclo[2.2.2]octane; potassium carbonate; palladium diacetate In acetone at 110℃; for 2h; Suzuki-Miyaura cross-coupling reaction; | 100% |
-
-
636-98-6
p-nitrobenzene iodide

-
-
153026-78-9
1-(thiophen-2-yl)prop-2-yn-1-ol

-
-
275815-89-9
(E)-3-(4-nitropheny)-1-thiophen-3-yl-propenone

| Conditions | Yield |
|---|---|
| With triethylamine; bis-triphenylphosphine-palladium(II) chloride; copper(l) iodide In tetrahydrofuran Isomerization; Sonogashira coupling; Heating; | 100% |
| With triethylamine; bis-triphenylphosphine-palladium(II) chloride; copper(l) iodide In tetrahydrofuran for 9h; Sonogashira coupling-isomerization reaction; Heating; | 95% |
| With copper(l) iodide; triethylamine; bis-triphenylphosphine-palladium(II) chloride In tetrahydrofuran for 16h; Sonogashira coupling; Isomerization; Heating; |

| Conditions | Yield |
|---|---|
| With 1,4-diaza-bicyclo[2.2.2]octane; tetrabutyl ammonium fluoride; palladium diacetate In 1,4-dioxane at 100℃; for 8h; Stille reaction; | 100% |
| With palladium diacetate; orotic acid; cesium fluoride In 1,4-dioxane at 100℃; for 18h; Concentration; Time; Stille Cross Coupling; Sealed tube; Inert atmosphere; Schlenk technique; | 100% |
| With C31H33INPPd In N,N-dimethyl-formamide at 80℃; for 24h; Stille coupling; | 99% |

| Conditions | Yield |
|---|---|
| With copper phthalocyanine; sodium hydroxide In dimethyl sulfoxide at 100℃; | 100% |
| With potassium phosphate; copper(l) iodide; (S,S)-1,2-diaminocyclohexane In 1,4-dioxane; dodecane at 110℃; for 24h; | 99% |
| With 1,1'-bis-(diphenylphosphino)ferrocene; palladium 10% on activated carbon; caesium carbonate In 1,3,5-trimethyl-benzene at 180℃; for 24h; Inert atmosphere; | 99% |
-
-
636-98-6
p-nitrobenzene iodide

-
-
141-32-2
acrylic acid n-butyl ester

-
-
86622-84-6, 131061-15-9
(E)-n-butyl 4-nitrocinnamate

| Conditions | Yield |
|---|---|
| With C29H33Cl2N5Pd; tetrabutylammomium bromide; sodium carbonate In N,N-dimethyl acetamide at 140℃; for 3h; Heck reaction; Inert atmosphere; | 100% |
| With potassium carbonate In N,N-dimethyl-formamide at 100℃; for 12h; Heck reaction; Inert atmosphere; regioselective reaction; | 99% |
| With 4,4’‐bis(trimethylammoniummethyl)‐2,2’‐bipyridine; diamminedichloropalladium(II); tributyl-amine In water at 140℃; for 12h; Mizoroki-Heck cross-coupling; Sealed tube; | 99% |

| Conditions | Yield |
|---|---|
| With potassium hydroxide; palladium on activated charcoal In water Heating; | 100% |

| Conditions | Yield |
|---|---|
| With copper phthalocyanine; sodium hydroxide In dimethyl sulfoxide at 100℃; | 100% |
| With Hippuric Acid; copper diacetate; caesium carbonate In N,N-dimethyl-formamide at 140℃; for 30h; Ullmann coupling reaction; | 99% |
| With caesium carbonate In dimethyl sulfoxide at 110℃; for 1h; Ullmann Condensation; | 98% |
-
-
636-98-6
p-nitrobenzene iodide

-
-
5720-07-0
4-methoxyphenylboronic acid

-
-
2143-90-0
4-methoxy-4'-nitrobiphenyl

| Conditions | Yield |
|---|---|
| With sodium carbonate In ethanol; water at 80℃; for 1h; Suzuki-Miyaura Coupling; Green chemistry; | 100% |
| With palladium diacetate; 4-Ph2P-C6H4-2-N-glucosamine; sodium carbonate In tetrahydrofuran; ethanol; water at 60℃; for 18h; | 99% |
| With polyhydroxylated phosphine; sodium carbonate; palladium diacetate In ethanol; water; toluene at 60℃; for 18h; | 99% |
-
-
849213-47-4
(Z)-3-methyl-4-phenyl-4-trimethylsilanylbut-3-en-2-ol

-
-
636-98-6
p-nitrobenzene iodide

| Conditions | Yield |
|---|---|
| With copper(l) iodide; lithium tert-butoxide; bis-triphenylphosphine-palladium(II) chloride In N,N-dimethyl-formamide at 20℃; for 0.666667h; | 100% |

| Conditions | Yield |
|---|---|
| With 1,4-diaza-bicyclo[2.2.2]octane; caesium carbonate; palladium diacetate In acetonitrile at 20℃; for 6h; Sonogashira cross-coupling reaction; | 100% |
| With caesium carbonate; 2-aminopyrimidine-4,6-diol; palladium diacetate In acetonitrile at 20℃; for 5h; Sonogashira reaction; | 95% |
| With tetrabutyl ammonium fluoride; copper diacetate at 130 - 135℃; for 14h; Sonogashira cross-coupling reaction; | 95% |
| With 1,4-diaza-bicyclo[2.2.2]octane; air; palladium diacetate In acetonitrile at 20℃; for 20h; Sonogashira reaction; | 90% |
| With 1,4-diaza-bicyclo[2.2.2]octane; caesium carbonate; copper(l) iodide In N,N-dimethyl-formamide at 135 - 140℃; for 4h; Sonogashira cross-coupling; |
-
-
636-98-6
p-nitrobenzene iodide

-
-
1663-39-4
tert-Butyl acrylate

-
-
370839-59-1
(E)-tert-butyl 3-(4-nitrophenyl)prop-2-enoate

| Conditions | Yield |
|---|---|
| With 1,4-diaza-bicyclo[2.2.2]octane; potassium carbonate; palladium diacetate In N,N-dimethyl-formamide at 80℃; for 4h; Heck cross-coupling reaction; | 100% |
| With tetrabutyl ammonium fluoride; palladium dichloride at 40℃; for 3h; Heck reaction; Inert atmosphere; neat (no solvent); | 100% |
| With 1,4-diaza-bicyclo[2.2.2]octane; poly(ethylene glycol)-400; potassium carbonate; palladium diacetate In water at 80℃; for 4h; Heck cross-coupling; | 98% |
-
-
636-98-6
p-nitrobenzene iodide

-
-
3757-88-8
tributylstannylethynylbenzene

-
-
1942-30-9
4-(phenylethynyl)nitrobenzene

| Conditions | Yield |
|---|---|
| With potassium fluoride; 1,4-dicyclohexyl-diazabutadiene; bis(dibenzylideneacetone)-palladium(0) In 1,4-dioxane at 100℃; for 16h; Stille cross-coupling reaction; | 100% |
| diphenylphosphine-based palladium(0) complex on MCM-41 In water; N,N-dimethyl-formamide at 60℃; for 6h; Stille coupling; | 87% |
| Stage #1: p-nitrobenzene iodide With diphenylphosphinoethane functionalized polystyrene resin-supported Pd(0) complex In N,N-dimethyl-formamide at 20℃; for 0.25h; Stille Cross Coupling; Stage #2: tributylstannylethynylbenzene In N,N-dimethyl-formamide at 65℃; for 7h; Stille Cross Coupling; | 86% |
-
-
636-98-6
p-nitrobenzene iodide

-
-
118486-94-5
2-(tributylstannyl)furan

-
-
28123-72-0
2-(4-nitrophenyl)furan

| Conditions | Yield |
|---|---|
| With potassium fluoride; 1,4-dicyclohexyl-diazabutadiene; bis(dibenzylideneacetone)-palladium(0) In 1,4-dioxane at 100℃; for 16h; Stille cross-coupling reaction; | 100% |
1-Iodo-4-nitrobenzene Specification
The 1-Iodo-4-nitrobenzene, with the CAS registry number 636-98-6, is also known as p-Nitroiodobenzene. It belongs to the product categories of Aromatic Hydrocarbons (substituted) & Derivatives; Benzene Derivates; API Intermediates; Nitro Compounds; Nitrogen Compounds; Organic Building Blocks. Its EINECS registry number is 202-676-4. This chemical's molecular formula is C6H4INO2 and molecular weight is 249.01. What's more, its IUPAC name is the same with its product name. It should be stored in a cool, dry and well-ventilated place.
Physical properties about 1-Iodo-4-nitrobenzene are: (1)ACD/LogP: 2.897; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): 2.90; (4)ACD/LogD (pH 7.4): 2.90; (5)ACD/BCF (pH 5.5): 93.69; (6)ACD/BCF (pH 7.4): 93.69; (7)ACD/KOC (pH 5.5): 897.35; (8)ACD/KOC (pH 7.4): 897.35; (9)#H bond acceptors: 3; (10)#H bond donors: 0; (11)#Freely Rotating Bonds: 1; (12)Polar Surface Area: 45.82 Å2; (13)Index of Refraction: 1.663; (14)Molar Refractivity: 45.705 cm3; (15)Molar Volume: 123.348 cm3; (16)Polarizability: 18.119×10-24cm3; (17)Surface Tension: 56.627 dyne/cm; (18)Density: 2.019 g/cm3; (19)Flash Point: 127.953 °C; (20)Enthalpy of Vaporization: 50.602 kJ/mol; (21)Boiling Point: 287.959 °C at 760 mmHg; (22)Vapour Pressure: 0.00400 mmHg at 25 °C.
Preparation of 1-Iodo-4-nitrobenzene: this chemical can be prepared by 4-nitro-aniline. This reaction needs reagents NO2, NaI and solvent acetonitrile at temperature of -20 °C. The reaction time is 10 hours. The yield is 98 %.

Uses of 1-Iodo-4-nitrobenzene: it is used to produce other chemicals. For example, it can produce 4,4'-dinitro-biphenyl. The reaction occurs with reagent Cu and other condition of heating for 15 min. The yield is 75 %.
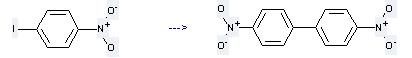
When you are dealing with this chemical, you should be very careful. This chemical is inflammation to the skin, eyes and respiratory system. It may cause damage to health by inhalation, in contacting with skin and if swallowed. Therefore, you should wear suitable protective clothing, gloves and eye/face protection. In case of contacting with eyes, you should rinse immediately with plenty of water and seek medical advice.
You can still convert the following datas into molecular structure:
(1) SMILES: O=[N+]([O-])c1ccc(I)cc1
(2) InChI: InChI=1S/C6H4INO2/c7-5-1-3-6(4-2-5)8(9)10/h1-4H
(3) InChIKey: SCCCFNJTCDSLCY-UHFFFAOYSA-N
Related Products
- 1-Iodo-4-nitrobenzene
- 6369-96-6
- 636-99-7
- 63699-78-5
- 63700-19-6
- 6370-08-7
- 6370-10-1
- 63701-37-1
- 637-01-4
- 63701-55-3
- 637020-57-6
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View