-
Name
3-Chloropropionic acid
- EINECS 203-534-4
- CAS No. 107-94-8
- Article Data98
- CAS DataBase
- Density 1.29 g/cm3
- Solubility Soluble in water, ethanol, chloroform, miscible with ether
- Melting Point 37-41 °C
- Formula C3H5ClO2
- Boiling Point 199 °C at 760 mmHg
- Molecular Weight 108.525
- Flash Point 66.9 °C
- Transport Information UN 3261 8/PG 2
- Appearance White to beige crystalline low melting mass
- Safety 26-36/37/39-45
- Risk Codes 35-34-21/22
-
Molecular Structure
-
Hazard Symbols
 C
C
- Synonyms Propionicacid, 3-chloro- (7CI,8CI);Propionic acid, b-chloro- (2CI);b-Monochloropropionic acid;NSC 174;NSC 2183;b-Chloropropionic acid;
- PSA 37.30000
- LogP 0.69990
Synthetic route

| Conditions | Yield |
|---|---|
| With benzene-1,2-dicarboxylic acid for 0.75h; microwave irradiation; | 91% |
| With benzene-1,2-dicarboxylic acid at 240℃; under 3040 Torr; for 1h; | 72% |
| With hydrogenchloride | |
| With water In dimethyl sulfoxide at 26℃; for 72h; pH=7; aq. phosphate buffer; | 25 %Chromat. |
-
-
625-36-5
2-chloropropionyl chloride

-
-
100-46-9
benzylamine

-
A
-
107-94-8
chloropropionic acid

-
B
-
13304-62-6
N-benzylacrylamide

-
C
-
501-68-8
Beclamide

| Conditions | Yield |
|---|---|
| In N,N-dimethyl-formamide at 100℃; for 0.0833333h; Temperature; Concentration; | A 6% B 5% C 89% |

-
-
625-36-5
2-chloropropionyl chloride

-
-
100-46-9
benzylamine

-
A
-
107-94-8
chloropropionic acid

-
B
-
501-68-8
Beclamide

| Conditions | Yield |
|---|---|
| In N,N-dimethyl-formamide at 50℃; for 0.0166667h; Time; | A 22% B 78% |

-
-
802294-64-0
propionic acid

-
A
-
590-92-1
3-Bromopropionic acid

-
B
-
107-94-8
chloropropionic acid

-
C
-
598-72-1
2-Bromopropionic acid

-
D
-
598-78-7
(R,S)-2-chloropropionic acid

| Conditions | Yield |
|---|---|
| With hydrogenchloride; oxygen; potassium bromide; sodium nitrite In chloroform; water at 40℃; under 760.051 Torr; for 18h; Sealed tube; Irradiation; | A 65% B n/a C 23% D n/a |


| Conditions | Yield |
|---|---|
| With hydrogenchloride at 65 - 70℃; for 4h; | 32.3% |
| With hydrogenchloride | |
| With hydrogenchloride at 12℃; |
-
-
57-57-8
β-Propiolactone

-
-
6921-34-2
benzylmagnesium chloride

-
A
-
107-94-8
chloropropionic acid

-
B
-
1821-12-1
4-Phenylbutyric acid


| Conditions | Yield |
|---|---|
| With hydrogenchloride; sodium chloride at 30 - 45℃; | |
| With hydrogenchloride |
-
-
19434-65-2
3-chloropropanaldehyde

-
-
107-94-8
chloropropionic acid

| Conditions | Yield |
|---|---|
| With nitric acid | |
| With nitric acid |

| Conditions | Yield |
|---|---|
| With chlorine |

| Conditions | Yield |
|---|---|
| With sulfuric acid |

| Conditions | Yield |
|---|---|
| at 75 - 80℃; unter Belichtung; |
-
-
802294-64-0
propionic acid

-
-
107-94-8
chloropropionic acid

| Conditions | Yield |
|---|---|
| With tetrachloromethane; sulfuryl dichloride; dibenzoyl peroxide | |
| With chlorine at 400℃; in der Dampfphase; | |
| With tetrachloromethane; sulfuryl dichloride; dibenzoyl peroxide | |
| With chlorine at 120℃; in fluessiges Phase; | |
| With chlorine at 30 - 60℃; unter Belichtung; |

| Conditions | Yield |
|---|---|
| With chlorine Irradiation.im UV-Licht; | |
| With hydrogenchloride Electrolysis.an einer Platinanode; | |
| With hydrogenchloride Electrolysis.an Platinanode; |
-
-
802294-64-0
propionic acid

-
A
-
107-94-8
chloropropionic acid

-
B
-
565-64-0
2,3-dichloropropanoic acid

-
C
-
598-78-7
(R,S)-2-chloropropionic acid

| Conditions | Yield |
|---|---|
| beim Chlorieren in der fluessigen Phase unter Belichtung; |


| Conditions | Yield |
|---|---|
| With hydrogenchloride bei Siedetemperatur; |

| Conditions | Yield |
|---|---|
| With hydrogenchloride | |
| With hydrogenchloride at 100℃; | |
| With hydrogenchloride und Kochen des Reaktionsgemisches unter weiterem Durchleiten von Chlorwasserstoff; | |
| With hydrogenchloride; water und Kochen des Reaktionsgemisches unter weiterem Durchleiten von Chlorwasserstoff; |

| Conditions | Yield |
|---|---|
| With hydrogenchloride |

| Conditions | Yield |
|---|---|
| With nitric acid | |
| With alkaline KMNO4 | |
| With nitric acid | |
| With oxygen; sodium hydroxide In water at 60℃; under 8258.9 Torr; Kinetics; Autoclave; |

| Conditions | Yield |
|---|---|
| With hydrogenchloride at 120℃; |

| Conditions | Yield |
|---|---|
| With lithium chloride In dimethyl sulfoxide at 100℃; for 6h; |
-
-
40861-09-4
3-chloroperpropionic acid

-
-
107-94-8
chloropropionic acid

| Conditions | Yield |
|---|---|
| With benzonitrile; (5,10,15,20-tetraphenylporphyrinato)manganese(III) chloride at 30℃; Rate constant; |
-
-
86641-76-1
3,12-bis(3'-bromo-1'-oxopropyl)-3,12-diaza-6,9-diazoniadispiro<5.2.5.2>hexadecane dichloride

-
A
-
590-92-1
3-Bromopropionic acid

-
B
-
107-94-8
chloropropionic acid

-
C
-
121051-82-9
3,12-bis(3'-chloro-1'-oxopropyl)-3,12-diaza-6,9-diazoniadispiro<5.2.5.2>hexadecane dibromide

-
D
-
95461-34-0
3,12-Bisacrylyl-3,12-diaza-6,9-diazoniadispiro<5,2,5,2>hexadecane dichloride

-
E
-
122842-21-1
3,12-bis(3'-hydroxy-1'-oxopropyl)-3,12-diaza-6,9-diazoniadispiro<5.2.5.2>hexadecane dichloride

-
F
-
503-66-2
3-hydroxypropionic acid

| Conditions | Yield |
|---|---|
| In water at 70℃; for 5h; Product distribution; Kinetics; Rate constant; various time; other temperature; |


| Conditions | Yield |
|---|---|
| In gas at 360 - 420℃; under 49 - 209 Torr; Rate constant; Kinetics; Thermodynamic data; E(act); |
-
-
5421-53-4
4,4'-Di-tert-butyldiphenyliodonium chloride

-
A
-
107-94-8
chloropropionic acid

-
B
-
253185-03-4, 253185-04-5
tert-butylbenzene

-
C
-
35779-04-5
1-tert-butyl-4-iodobenzene

-
D
-
3972-56-3
4-(tert-butyl)chlorobenzene

| Conditions | Yield |
|---|---|
| With (+)-cis-dioxolane; 2,2'-azobis(isobutyronitrile) at 75℃; for 7h; Product distribution; Quantum yield; various reaction conditions; also photochemical reaction; |


| Conditions | Yield |
|---|---|
| Product distribution; dehalogenation; P. butanovora strain ml-15; aerobic condition; other bacterium strains; other halogeno alkanes; |

| Conditions | Yield |
|---|---|
| With water In acetonitrile Rate constant; var. pH; |


| Conditions | Yield |
|---|---|
| at 100℃; | |
| bei nachfolgenden Kochen unter weiterem Durchleiten von Chlorwasserstoff;analog verlauft die Einw. von Bromwasserstoff u Jodwasserstoff; |
-
-
107-94-8
chloropropionic acid


| Conditions | Yield |
|---|---|
| Stage #1: chloropropionic acid With (chloro-phenylthio-methylene)dimethylammonium chloride In dichloromethane at 0℃; for 0.5h; Stage #2: 2,4-dimethylcyclohexan-1-ol In dichloromethane at 20℃; for 12h; | 100% |
-
-
107-94-8
chloropropionic acid

-
-
77380-28-0
2,5-Difluorothiophenol

-
-
926232-57-7
3-((2,5-difluorophenyl)thio)propanoic acid

| Conditions | Yield |
|---|---|
| Stage #1: 2,5-Difluorothiophenol With sodium hydroxide In water at 0℃; for 0.0833333h; Stage #2: chloropropionic acid In water at 70℃; Stage #3: With hydrogenchloride In water at 0℃; | 100% |
-
-
107-94-8
chloropropionic acid

-
-
137757-24-5
4-(6-hydroxyhexyl)benzoic acid

| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid In dichloromethane at 50℃; for 18h; Dean-Stark; | 100% |

| Conditions | Yield |
|---|---|
| hydrogenchloride In water for 8h; Product distribution / selectivity; | 99% |
| With hydrogenchloride | |
| With sulfuric acid | |
| With sulfuric acid |
-
-
107-94-8
chloropropionic acid

-
-
20495-99-2
3-chloropropanoic anhydride

| Conditions | Yield |
|---|---|
| With dicyclohexyl-carbodiimide In dichloromethane at 20℃; Inert atmosphere; Glovebox; | 99% |
| With dicyclohexyl-carbodiimide for 0.0833333h; | 65 % Spectr. |
-
-
107-94-8
chloropropionic acid

-
-
887003-12-5
2-amino-4-chloro-6-methyl-10,10-dioxo-10,11-dihydro-5-oxa-10λ6-thia-dibenzo[a,d]cycloheptene-8-carboxylic acid methyl ester

-
-
887003-66-9
4-chloro-2-(3-chloro-propylamino)-6-methyl-10,10-dioxo-10,11-dihydro-5-oxa-10λ6-thia-dibenzo[a,d]cycloheptene-8-carboxylic acid methyl ester

| Conditions | Yield |
|---|---|
| Stage #1: chloropropionic acid With sodium tetrahydroborate In tetrahydrofuran; benzene at 0 - 20℃; for 1h; Stage #2: 2-amino-4-chloro-6-methyl-10,10-dioxo-10,11-dihydro-5-oxa-10λ6-thia-dibenzo[a,d]cycloheptene-8-carboxylic acid methyl ester In tetrahydrofuran; benzene for 3h; Heating / reflux; | 99% |
-
-
107-94-8
chloropropionic acid

-
-
32943-25-2
3-chloro-10,11-dihydro-5H-dibenzo[b,f]azepine

| Conditions | Yield |
|---|---|
| Stage #1: chloropropionic acid With sodium tetrahydroborate In toluene at 0 - 5℃; Stage #2: 3-chloro-10,11-dihydro-5H-dibenzo[b,f]azepine With sodium tetrahydroborate In toluene at 25 - 80℃; for 7h; Stage #3: With hydrogenchloride In water; isopropyl alcohol | 99% |

| Conditions | Yield |
|---|---|
| With bis(trichloromethyl) carbonate In toluene at 25℃; for 8h; Reagent/catalyst; Temperature; | 98.5% |
| With thionyl chloride | 63% |
| With phosphorus trichloride |
-
-
107-94-8
chloropropionic acid

-
-
95-88-5
4-Chlororesorcinol

-
-
1202889-56-2
3-chloro-1-(5-chloro-2,4-dihydroxyphenyl)propan-1-one

| Conditions | Yield |
|---|---|
| Stage #1: 4-Chlororesorcinol With trifluorormethanesulfonic acid at 4 - 10℃; Stage #2: chloropropionic acid at 10 - 55℃; Product distribution / selectivity; | 98.4% |
| Stage #1: 4-Chlororesorcinol With trifluorormethanesulfonic acid at 4 - 10℃; Stage #2: chloropropionic acid at 10 - 55℃; Product distribution / selectivity; | 98.4% |

| Conditions | Yield |
|---|---|
| at 70℃; for 50h; | 98% |
| In toluene for 5h; Reflux; Inert atmosphere; | 86% |
| In toluene Schlenk technique; | 80% |
| In ethyl acetate at 70℃; for 24h; |

| Conditions | Yield |
|---|---|
| Stage #1: chloropropionic acid With sodium tetrahydroborate In toluene at 0 - 5℃; Stage #2: 1-azaphenothiazine With sodium tetrahydroborate In toluene at 25 - 75℃; for 6h; Stage #3: With hydrogenchloride In water; isopropyl alcohol | 98% |
-
-
107-94-8
chloropropionic acid

-
-
106-45-6
para-thiocresol

-
-
13739-35-0
3-[(4-methylphenyl)thio]propionic acid

| Conditions | Yield |
|---|---|
| With sodium hydroxide In water for 0.5h; Microwave irradiation; | 97% |
| Stage #1: chloropropionic acid; para-thiocresol With potassium hydroxide; potassium carbonate In ethanol Reflux; Stage #2: With hydrogenchloride In ethanol; water Large scale reaction; | 77% |
| With sodium hydroxide |

| Conditions | Yield |
|---|---|
| Stage #1: chloropropionic acid With sodium tetrahydroborate In toluene at 0 - 5℃; Stage #2: 10H-phenothiazine With sodium tetrahydroborate In toluene at 25 - 80℃; for 5h; Stage #3: With hydrogenchloride In water; isopropyl alcohol | 97% |
-
-
107-94-8
chloropropionic acid

-
-
578-95-0
10H-acridin-9-one

-
-
226424-38-0
3-(9-oxo-9,10-dihydro-10-acridinyl)propanoic acid

| Conditions | Yield |
|---|---|
| With 1-ethyl-3-methylimidazolium hydroxide at 50℃; for 8h; Ionic liquid; | 95.9% |
-
-
107-94-8
chloropropionic acid

| Conditions | Yield |
|---|---|
| With 4-methyl-morpholine In tetrahydrofuran at 0 - 5℃; for 1.75h; | 95.7% |
-
-
107-94-8
chloropropionic acid

-
-
637-89-8
4-sulfanylphenol

-
-
30512-65-3
3-[(4-hydroxyphenyl)sulfanyl]propanoic acid

| Conditions | Yield |
|---|---|
| With sodium hydroxide In water at 100℃; for 3h; | 95% |
| With potassium hydroxide In water at 80℃; for 2.5h; pH=12; |
-
-
107-94-8
chloropropionic acid

-
-
13451-78-0
5-fluorobenzo[d]oxazole-2-thiol

| Conditions | Yield |
|---|---|
| With caesium carbonate In acetonitrile at 100℃; for 1h; Microwave irradiation; | 95% |

| Conditions | Yield |
|---|---|
| Stage #1: thiophenol With sodium hydroxide In water at 20℃; Stage #2: chloropropionic acid In water for 4h; Reflux; Stage #3: With hydrogenchloride In water at 20℃; pH=< 2; | 94% |
| With potassium carbonate; potassium hydroxide In ethanol; water at 0℃; for 5h; Reflux; | 70% |
| Stage #1: chloropropionic acid; thiophenol With potassium hydroxide; potassium carbonate In ethanol Reflux; Stage #2: With hydrogenchloride In ethanol; water | 63% |
-
-
107-94-8
chloropropionic acid

-
-
371-42-6
4-Fluorothiophenol

-
-
19543-85-2
3-(4-fluorophenylthio)propanoic acid

| Conditions | Yield |
|---|---|
| Stage #1: 4-Fluorothiophenol With potassium hydroxide In ethanol; water at 60℃; Stage #2: chloropropionic acid With potassium carbonate In ethanol; water at 20 - 96℃; for 5.5h; | 94% |
| With potassium carbonate; potassium hydroxide In ethanol; water at 0℃; for 5h; Reflux; | 92% |
| Stage #1: chloropropionic acid; 4-Fluorothiophenol With potassium hydroxide; potassium carbonate In ethanol Reflux; Stage #2: With hydrogenchloride In ethanol; water | 90% |
-
-
107-94-8
chloropropionic acid

-
-
1117-97-1
N,0-dimethylhydroxylamine

-
-
1062512-53-1
3-chloro-N-methoxy-N-methylpropanamide

| Conditions | Yield |
|---|---|
| Stage #1: chloropropionic acid; N,0-dimethylhydroxylamine In toluene at 0℃; for 0.166667h; Stage #2: With phosphorus trichloride In toluene at 20 - 60℃; for 0.5h; | 94% |
-
-
107-94-8
chloropropionic acid

-
-
603-35-0
triphenylphosphine

-
-
36626-29-6
(2-carboxyethyl)triphenylphosphonium chloride

| Conditions | Yield |
|---|---|
| In toluene at 110℃; for 18h; | 93% |
| In xylene for 18h; Heating; | 89% |
| In 5,5-dimethyl-1,3-cyclohexadiene; water for 4h; Reflux; | 84% |

| Conditions | Yield |
|---|---|
| With trifluorormethanesulfonic acid at 80℃; for 1h; | 92% |
| With trifluorormethanesulfonic acid at 80℃; for 1h; | 90% |
| With trifluorormethanesulfonic acid at 85℃; for 0.75h; | 86% |
-
-
107-94-8
chloropropionic acid

-
-
1062512-43-9
C6H18N3O3P

-
-
1062512-53-1
3-chloro-N-methoxy-N-methylpropanamide

| Conditions | Yield |
|---|---|
| In toluene at 60℃; | 92% |
-
-
107-94-8
chloropropionic acid

-
-
202582-35-2
N-[2-((2-S-(4-methoxybenzyl)sulfanyl)ethyl)amino]acetyl-S-(4-methoxybenzyl)-2-aminoethanethiol

| Conditions | Yield |
|---|---|
| With triethylamine In acetonitrile at 80℃; | 92% |
-
-
107-94-8
chloropropionic acid

| Conditions | Yield |
|---|---|
| With benzotriazol-1-ol; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride; triethylamine In N,N-dimethyl-formamide at 20℃; for 2h; | 92% |
| With 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride; triethylamine In N,N-dimethyl-formamide at 20℃; | 22% |

| Conditions | Yield |
|---|---|
| With trifluorormethanesulfonic acid; bis(4-methoxyphenyl)selenide In hexane at 20℃; for 24h; Sealed tube; | 91% |


| Conditions | Yield |
|---|---|
| at 70 - 80℃; | 90% |
-
-
107-94-8
chloropropionic acid

-
-
4559-70-0
Diphenylphosphine oxide

-
-
29874-10-0
3-(diphenyl-phosphinoyl)-propionic acid

| Conditions | Yield |
|---|---|
| With potassium hydroxide In dimethyl sulfoxide at 50℃; for 1h; | 90% |
| (i) NaOEt, EtOH, (ii) /BRN= 1098495/; Multistep reaction; |
3-Chloropropionic acid Consensus Reports
Reported in EPA TSCA Inventory.
3-Chloropropionic acid Specification
The 3-Chloropropionic acid with CAS registry number of 107-94-8 is also known as Propanoic acid,3-chloro-. The IUPAC name and product name are the same. It belongs to product categories of omega-Chlorocarboxylic Acids; omega-Functional Alkanols, Carboxylic Acids, Amines & Halides; C1 to C5; Carbonyl Compounds; Carboxylic Acids. Its EINECS registry number is 203-534-4. In addition, the formula is C3H5ClO2 and the molecular weight is 108.52. This chemical is a white to beige crystalline low melting mass that soluble in water. It may destroy living tissue on contact and should be sealed in ventilated, cool room away from fire, heat. What's more, this chemical is used as intermediate in organic synthesis and also can be used for production of anti-epilepsy drugs.
Physical properties about 3-Chloropropionic acid are: (1)ACD/LogP: 0.41; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): -1.1; (4)ACD/LogD (pH 7.4): -2.83; (5)ACD/BCF (pH 5.5): 1; (6)ACD/BCF (pH 7.4): 1; (7)ACD/KOC (pH 5.5): 1.23; (8)ACD/KOC (pH 7.4): 1; (9)#H bond acceptors: 2; (10)#H bond donors: 1; (11)#Freely Rotating Bonds: 2; (12)Index of Refraction: 1.444; (13)Molar Refractivity: 22.36 cm3; (14)Molar Volume: 84 cm3; (15)Surface Tension: 40.2 dyne/cm; (16)Density: 1.29 g/cm3; (17)Flash Point: 66.9 °C; (18)Enthalpy of Vaporization: 47.97 kJ/mol; (19)Boiling Point: 199 °C at 760 mmHg; (20)Vapour Pressure: 0.147 mmHg at 25 °C.
Preparation of 3-Chloropropionic acid: it is prepared by reaction of acrylic acid. The reaction needs reagent conc. aq. HCl at the temperature of 65 - 70 °C for 4 hours. The yield is about 32.3 %.

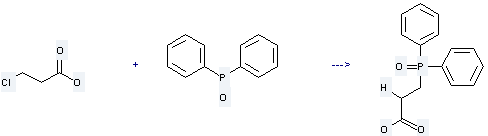
Uses of 3-Chloropropionic acid: it is used to produce 3-diphenylphosphinylpropionsaeure by reaction with diphenylphosphane oxide. The reaction occurs with reagent aq. KOH and solvent dimethylsulfoxide at the temperature of 50 °C for 1 hour. The yield is about 90 %.
When you are using this chemical, please be cautious about it. As a chemical, it is harmful in contact with skin and if swallowed. Furthermore, it causes severe burns. During using it, wear suitable protective clothing, gloves and eye/face protection. If contact with eyes accidently, rinse immediately with plenty of water and seek medical advice. In case of accident or if you feel unwell seek medical advice immediately.
You can still convert the following datas into molecular structure:
1. Canonical SMILES: C(CCl)C(=O)O
2. InChI: InChI=1S/C3H5ClO2/c4-2-1-3(5)6/h1-2H2,(H,5,6)
3. InChIKey: QEYMMOKECZBKAC-UHFFFAOYSA-N
The toxicity data is as follows:
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| mouse | LDLo | skin | 1040mg/kg (1040mg/kg) | Cancer Research. Vol. 28, Pg. 653, 1968. |
Related Products
- 3-Chloropropionic acid
- 107949-21-3
- 107-95-9
- 107-96-0
- 1079-66-9
- 107-97-1
- 107971-01-7
- 107978-77-8
- 107979-42-0
- 107-98-2
- 107983-78-8
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View