-
Name
Methyl acetoacetate
- EINECS 203-299-8
- CAS No. 105-45-3
- Article Data104
- CAS DataBase
- Density 1.039 g/cm3
- Solubility 460 g/L in water at 20 °C
- Melting Point -80 °C
- Formula C5H8O3
- Boiling Point 169.4 °C at 760 mmHg
- Molecular Weight 116.117
- Flash Point 61.6 °C
- Transport Information
- Appearance Colorless transparent liquid
- Safety 26
- Risk Codes 36
-
Molecular Structure
-
Hazard Symbols
 Xi
Xi
- Synonyms Acetoaceticacid, methyl ester (6CI,8CI);3-Oxobutanoic acid methyl ester;3-Oxobutyricacid methyl ester;Acetoacetate methyl ester;Methyl 3-oxobutanoate;Methyl3-oxobutyrate;Methyl acetylacetate;
- PSA 43.37000
- LogP 0.13850
Synthetic route
-
-
67-56-1
methanol

-
-
72324-39-1
5-acetyl-2,2-dimethyl-1,3-dioxane-4,6-dione

-
-
105-45-3
acetoacetic acid methyl ester

| Conditions | Yield |
|---|---|
| for 0.0666667h; microwave irradiation; | 98% |
| for 5h; Reflux; |

-
A
-
105-45-3
acetoacetic acid methyl ester

| Conditions | Yield |
|---|---|
| With triethylamine In tetrahydrofuran | A 49% B 96% |
-
-
67-56-1
methanol

-
-
201230-82-2
carbon monoxide

-
-
78-95-5
chloroacetone

-
-
105-45-3
acetoacetic acid methyl ester

| Conditions | Yield |
|---|---|
| With tributyl-amine; triphenylphosphine; bis(dibenzylideneacetone)-palladium(0) at 110℃; under 11250.9 Torr; for 1h; Product distribution; Further Variations:; Reagents; Temperatures; time; | 95% |
| With tributyl-amine; bis-triphenylphosphine-palladium(II) chloride at 110℃; under 7500.6 Torr; for 2h; | 80% |

-
-
67-56-1
methanol

-
-
5394-63-8
2,2,6-trimethyl-4H-1,3-dioxin-4-one

-
-
105-45-3
acetoacetic acid methyl ester

| Conditions | Yield |
|---|---|
| Esterification; | 92% |
| In 5,5-dimethyl-1,3-cyclohexadiene Reflux; | 77% |
| at 110 - 120℃; | 72% |
| In 5,5-dimethyl-1,3-cyclohexadiene at 150℃; |

| Conditions | Yield |
|---|---|
| With di(n-butyl)tin oxide In methanol for 12h; Heating; | 90% |
| In xylene Heating; |
-
-
109514-04-7
-essigsaeure-methylester

-
A
-
79-20-9
acetic acid methyl ester

-
B
-
13253-76-4
acetic acid-(1-methoxy-vinyl ester)

-
C
-
2279-76-7
tripropyltin chloride

-
D
-
105-45-3
acetoacetic acid methyl ester

| Conditions | Yield |
|---|---|
| With acetyl chloride | A 18% B 46% C 88% D 9% |

-
-
67-56-1
methanol

-
-
77-92-9
citric acid

-
A
-
1830-54-2
3-oxopentanedioic acid dimethyl ester

-
B
-
100009-70-9
dimethyl 2-methoxypropene-1,3-dicarboxylate

-
C
-
20820-77-3
trimethyl aconitate

-
D
-
1587-20-8
trimethyl citrate

-
E
-
105-45-3
acetoacetic acid methyl ester

| Conditions | Yield |
|---|---|
| Stage #1: citric acid With chlorosulfonic acid In dichloromethane at 10 - 15℃; for 5 - 6h; Stage #2: methanol In dichloromethane at 3 - 35℃; for 2h; Conversion of starting material; | A 87.3% B n/a C n/a D n/a E n/a |

-
-
70-11-1
α-bromoacetophenone

-
-
2911-22-0
6-methyl-4-oxo-2-thioxo-3,4-dihydro-2H-1,3-oxazine

-
A
-
105-45-3
acetoacetic acid methyl ester

-
B
-
5399-30-4
2-oxo-2-phenylethylthiocyanate

| Conditions | Yield |
|---|---|
| With sodium methylate In methanol for 12h; Ambient temperature; | A 29% B 87% |


| Conditions | Yield |
|---|---|
| With oxygen; nickel dichloride In methanol; water at 100℃; under 7500.75 Torr; for 12h; Reagent/catalyst; Solvent; Temperature; Autoclave; | 85.3% |

| Conditions | Yield |
|---|---|
| With lithium hexamethyldisilazane In tetrahydrofuran; hexane at -45℃; 1.) 30 min, 2.) 90 min; | 84% |

| Conditions | Yield |
|---|---|
| With sodium hydride In tetrahydrofuran for 20h; Heating; | 83% |
| With sodium methylate at 60℃; | |
| With sodium anschliessend mit Essigsaeure oder Kohlendioxyd; |
-
-
67-56-1
methanol

-
-
2033-24-1
cycl-isopropylidene malonate

-
-
75-36-5
acetyl chloride

-
-
105-45-3
acetoacetic acid methyl ester

| Conditions | Yield |
|---|---|
| Stage #1: cycl-isopropylidene malonate; acetyl chloride With pyridine In dichloromethane at 20℃; for 2h; Stage #2: methanol In dichloromethane for 2h; Further stages.; | 75% |

-
-
623-43-8, 4358-59-2, 18707-60-3, 130981-57-6
methyl crotonate

-
-
105-45-3
acetoacetic acid methyl ester

| Conditions | Yield |
|---|---|
| With methanol; water; oxygen; toluene-4-sulfonic acid; palladium dichloride In N,N-dimethyl acetamide at 80℃; under 4560.31 Torr; for 3h; Autoclave; regioselective reaction; | 73% |
| With tert.-butylhydroperoxide; sodium tetrachloropalladate(II) In acetic acid; tert-butyl alcohol at 50℃; for 3h; |

| Conditions | Yield |
|---|---|
| for 4.5h; Heating; | 72.5% |
| With dmap | |
| 1,4-diaza-bicyclo[2.2.2]octane at 20℃; for 0.343333h; |
-
-
67-56-1
methanol

-
-
2911-21-9
2,4-dioxo-6-methyl-3,4-dihydro-2H-1,3-oxazine

-
A
-
105-45-3
acetoacetic acid methyl ester

-
B
-
598-55-0
methyl carbamate

| Conditions | Yield |
|---|---|
| With triethylamine for 24h; Heating; | A 70% B 15% |

-
-
186581-53-3, 908094-01-9
diazomethane

-
-
1694-31-1
tert-butyl acetoacetate

-
A
-
926-05-6
tert-butyl nitrate

-
B
-
105-45-3
acetoacetic acid methyl ester

| Conditions | Yield |
|---|---|
| With nitric acid In dichloromethane at 0℃; for 2h; | A 60% B 20% |


| Conditions | Yield |
|---|---|
| With sodium methylate In methanol Ambient temperature; | 57% |

-
A
-
22322-43-6
2-methylsulfanyl-ethanethiol

-
B
-
105-45-3
acetoacetic acid methyl ester

| Conditions | Yield |
|---|---|
| With water at 40℃; for 0.0833333h; | A 56% B 13% |

| Conditions | Yield |
|---|---|
| With manganese(IV) oxide; palladium dichloride In water; acetonitrile at 60℃; for 26h; | 52% |
-
-
10199-63-0
1-(3,5-dimethyl-1H-pyrazol-1-yl)ethanone

-
-
96-32-2
bromoacetic acid methyl ester

-
-
105-45-3
acetoacetic acid methyl ester

| Conditions | Yield |
|---|---|
| With zinc In tetrahydrofuran for 1h; Heating; | 50% |
-
-
70569-71-0
β-(N,N-dimethoxyamino)-isovaleric acid methyl ester

-
-
105-45-3
acetoacetic acid methyl ester

| Conditions | Yield |
|---|---|
| With boron trifluoride diethyl etherate In tetrachloromethane at 20℃; | 43.8% |
-
-
74590-73-1
1,3-bis(trimethylsiloxy)-1-methoxybuta-1,3-diene

-
-
75-36-5
acetyl chloride

-
A
-
29736-80-9
3,5-dioxohexanoic acid methyl ester

-
B
-
105-45-3
acetoacetic acid methyl ester

| Conditions | Yield |
|---|---|
| In diethyl ether at -78℃; for 4h; Yields of byproduct given; | A 30% B n/a |

-
-
79-20-9
acetic acid methyl ester

-
A
-
1427722-55-1
methyl 5-hydroxy-2-methoxycarbonyl-5-methyl-3-oxocyclohexen-1-acetate

-
B
-
105-45-3
acetoacetic acid methyl ester

| Conditions | Yield |
|---|---|
| Stage #1: acetic acid methyl ester With lithium hexamethyldisilazane In tetrahydrofuran at -78℃; Stage #2: methyl (2SR,3RS)-1-benzyl-2-(indol-3-yl)piperidine-3-carboxylate In tetrahydrofuran at 0℃; for 15h; | A 27% B 24% |

-
-
771-03-9
3-acetyl-4-hydroxy-6-methyl-2H-pyran-2-one

-
A
-
675-10-5
4-hydroxy-6-methyl-2-pyron

-
B
-
29736-80-9
3,5-dioxohexanoic acid methyl ester

-
C
-
105-45-3
acetoacetic acid methyl ester

| Conditions | Yield |
|---|---|
| With sodium methylate In methanol; benzene for 18h; Heating; | A 7% B 18% C 16% |


| Conditions | Yield |
|---|---|
| sulfuric acid at 50℃; for 90h; | 13% |
-
-
67-56-1
methanol

-
-
76352-65-3
ethyl (E)-2-acetyl-3-oxo-5-phenylpent-4-enoate

-
-
124-41-4
sodium methylate

-
A
-
79-20-9
acetic acid methyl ester

-
B
-
42996-88-3
methyl 3-oxo-5-phenyl-4-pentenoate

-
C
-
103-26-4
Methyl cinnamate

-
D
-
105-45-3
acetoacetic acid methyl ester

-
-
1487-49-6
3-hydroxybutyric acid methyl ester

-
A
-
105-45-3
acetoacetic acid methyl ester

-
B
-
67-64-1
acetone

| Conditions | Yield |
|---|---|
| With copper at 330℃; |

| Conditions | Yield |
|---|---|
| at 57 - 80℃; Selbstkondensation; |
-
-
15506-53-3
cyclobutane-1,3-dione

-
-
105-45-3
acetoacetic acid methyl ester

| Conditions | Yield |
|---|---|
| With methanol; sodium methylate |

| Conditions | Yield |
|---|---|
| With sulfuryl dichloride; 2,2'-azobis(isobutyronitrile) In benzene at 20℃; for 2h; Inert atmosphere; | 100% |
| With sulfuryl dichloride In dichloromethane for 1h; Ambient temperature; | 98% |
| With ammonium cerium(IV) nitrate at 25 - 28℃; for 3h; | 85% |
| With sulfuryl dichloride | |
| With sulfuryl dichloride at 0℃; |
-
-
100-52-7
benzaldehyde

-
-
105-45-3
acetoacetic acid methyl ester

-
-
53063-63-1
5-hydroxy-3-oxo-5-phenyl-valeric acid methyl ester

| Conditions | Yield |
|---|---|
| Stage #1: acetoacetic acid methyl ester With sodium hydride In tetrahydrofuran at 0℃; for 0.166667h; Stage #2: With n-butyllithium In tetrahydrofuran; hexane at 20℃; for 0.333333h; Stage #3: benzaldehyde In tetrahydrofuran; hexane at -78 - 20℃; for 1.08333h; | 100% |
| Stage #1: acetoacetic acid methyl ester With sodium hydride In tetrahydrofuran at 20℃; for 0.5h; Inert atmosphere; Stage #2: With n-butyllithium In tetrahydrofuran; hexane at -15 - -10℃; for 0.5h; Inert atmosphere; Stage #3: benzaldehyde Further stages; | 92% |
| Stage #1: acetoacetic acid methyl ester With sodium hydride In tetrahydrofuran at 20℃; for 0.5h; Inert atmosphere; Stage #2: With n-butyllithium In tetrahydrofuran; hexane at -15 - -10℃; for 0.5h; Stage #3: benzaldehyde Further stages; | 92% |
-
-
105-45-3
acetoacetic acid methyl ester

-
-
870-63-3
prenyl bromide

-
-
53067-23-5
methyl 7-methyl-3-oxooct-6-enoate

| Conditions | Yield |
|---|---|
| Stage #1: acetoacetic acid methyl ester With sodium hydride In tetrahydrofuran; mineral oil at 0℃; for 0.166667h; Stage #2: With n-butyllithium In tetrahydrofuran; mineral oil for 0.166667h; Stage #3: prenyl bromide In tetrahydrofuran; mineral oil for 0.0833333h; | 100% |
| With n-butyllithium; sodium hydride In tetrahydrofuran at 0 - 20℃; | 100% |
| With n-butyllithium; sodium hydride In tetrahydrofuran at 0℃; | 79% |

| Conditions | Yield |
|---|---|
| In methanol at 25℃; for 3h; | 100% |

| Conditions | Yield |
|---|---|
| In methanol at 25℃; for 10h; | 100% |
-
-
100-52-7
benzaldehyde

-
-
105-45-3
acetoacetic acid methyl ester

-
-
70677-78-0
dimethyl 4-phenyl-1,4-dihydro-2,6-dimethylpyridine-3,5-dicarboxylate

| Conditions | Yield |
|---|---|
| With C23H3BF16N2O; ammonium acetate In toluene at 100℃; for 10h; Reagent/catalyst; Hantzsch Dihydropyridine Synthesis; | 100% |
| With ammonium acetate In ethanol at 60℃; for 0.5h; Catalytic behavior; Hantzsch Dihydropyridine Synthesis; | 98% |
| With ammonium acetate at 90℃; for 0.916667h; Hantzsch synthesis; Neat (no solvent); | 97% |
-
-
67-56-1
methanol

-
-
105-45-3
acetoacetic acid methyl ester

-
-
29267-46-7
3,3-bismethoxybutyric acid methyl ester

| Conditions | Yield |
|---|---|
| With RuCl2-(S)-(2,2'-bis(diphenylphosphino)-1,1'-binaphthyl)(benzonitrile)2; hydrogen at 50℃; under 76000.1 Torr; for 48h; | 100% |

| Conditions | Yield |
|---|---|
| In methanol at 25℃; for 3h; | 100% |
-
-
105-45-3
acetoacetic acid methyl ester

-
-
78443-74-0
(E)-3-<2,4-dichloro-6-(phenylmethoxy)phenyl>-2-propenal

-
-
78443-77-3
methyl (E)-7-<2,4-dichloro-6-(phenylmethoxy)phenyl>-5-hydroxy-3-oxo-6-heptenoate

| Conditions | Yield |
|---|---|
| With hydrogenchloride; n-butyllithium In tetrahydrofuran; hexane | 100% |
| With hydrogenchloride; n-butyllithium In tetrahydrofuran; hexane | 100% |
| With n-butyllithium; sodium hydride Yield given. Multistep reaction; |
-
-
105-45-3
acetoacetic acid methyl ester

-
-
53562-86-0
(S)-3-hydroxybutyric acid methyl ester

| Conditions | Yield |
|---|---|
| With cat; hydrogen In methanol; dichloromethane at 25℃; under 77572.2 Torr; for 48h; | 100% |
| With (S)-BiphempRuBr2; hydrogen In methanol at 80℃; under 7600 Torr; for 1h; | 100% |
| With hydrogen; [RuCl2(Sax-4,4'-bis(3-diphenylphosphineestra...)(DMF)]n In methanol at 100℃; under 76000 Torr; for 1h; Catalytic hydrogenation; | 100% |
-
-
105-45-3
acetoacetic acid methyl ester

-
A
-
53562-86-0
(S)-3-hydroxybutyric acid methyl ester

-
B
-
3976-69-0
Methyl (R)-3-hydroxybutyrate

| Conditions | Yield |
|---|---|
| With Ru2Cl4-(S)-(2,2'-bis(diphenylphosphino)-1,1'-binaphthyl)2*NEt3; hydrogen In methanol at 35℃; under 76000.1 Torr; for 48h; Title compound not separated from byproducts; | A 100% B n/a |
| With <((R)-(1,1'-binaphthyl-2,2'-diyl)bis(diphenylphosphine))RuCl2>2NEt3; hydrogen In methanol at 35℃; under 76000.1 Torr; for 48h; Title compound not separated from byproducts; | A n/a B 100% |
| With hydrogen; Ru(BICP)Br2 In methanol at 40 - 65℃; under 750.075 - 3750.38 Torr; | A n/a B 95% |

| Conditions | Yield |
|---|---|
| With cat; hydrogen In methanol; dichloromethane at 25℃; under 77572.2 Torr; for 48h; | 100% |
| With (R)-MeOBiphepRuBr2; hydrogen In methanol at 50℃; under 15200 Torr; for 48h; | 100% |
| With (S)-4,12-bis(diphenylphosphino)-<2.2>paracyclophane-Ru(II)bis(trifluoroacetate); hydrogen; tetra-(n-butyl)ammonium iodide In methanol; water at -5℃; under 2585.7 Torr; for 18h; | 100% |
-
-
105-45-3
acetoacetic acid methyl ester

-
-
156832-27-8
(1S,3R,5R,7R)-3-Chloro-10,10-dimethyl-3-phenyl-4-oxa-3λ4-selena-tricyclo[5.2.1.01,5]decane

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane for 1h; Ambient temperature; | 100% |
-
-
123-11-5
4-methoxy-benzaldehyde

-
-
105-45-3
acetoacetic acid methyl ester

-
-
57-13-6
urea

-
-
205999-87-7
5-methoxycarbonyl-4-(4-methoxyphenyl)-6-methyl-3,4-dihydropyrimidin-2(1H)-one

| Conditions | Yield |
|---|---|
| With sulfuric acid; copper(l) chloride In methanol Biginelli Pyrimidone Synthesis; Reflux; Inert atmosphere; | 100% |
| With ytterbium(III) triflate at 100℃; for 0.333333h; Biginelli reaction; | 99% |
| With calcium chloride In ethanol for 2h; Biginelli reaction; Heating; | 98% |

-
-
105-45-3
acetoacetic acid methyl ester

-
-
60-23-1
Cysteamine

-
-
503616-48-6
(Z)-3-(2-Mercapto-ethylamino)-but-2-enoic acid methyl ester

| Conditions | Yield |
|---|---|
| for 24h; Ambient temperature; | 100% |
-
-
105-45-3
acetoacetic acid methyl ester

-
-
618-36-0
rac-methylbenzylamine

-
-
374559-41-8
(Z)-3-(1-Phenyl-ethylamino)-but-2-enoic acid methyl ester

| Conditions | Yield |
|---|---|
| In methanol for 12h; Heating; | 100% |
-
-
105-45-3
acetoacetic acid methyl ester

-
-
100-46-9
benzylamine

-
-
72002-24-5
3-benzylaminobut-2-enoic acid methyl ester

| Conditions | Yield |
|---|---|
| In methanol for 12h; Heating; | 100% |
| In methanol at 20℃; for 0.5h; | 95% |
| With calcium bistrifluoroacetate at 20℃; for 0.5h; chemoselective reaction; | 95% |
-
-
105-45-3
acetoacetic acid methyl ester

-
-
20989-17-7, 56613-80-0, 71006-16-1, 7568-92-5, 188586-38-1
Phenylglycinol

-
-
374559-28-1
(Z)-3-(2-Hydroxy-1-phenyl-ethylamino)-but-2-enoic acid methyl ester

| Conditions | Yield |
|---|---|
| In methanol for 12h; Heating; | 100% |
-
-
20989-17-7
(2S)-2-phenylglycinol

-
-
105-45-3
acetoacetic acid methyl ester

-
-
476187-10-7
(Z)-3-((S)-2-Hydroxy-1-phenyl-ethylamino)-but-2-enoic acid methyl ester

| Conditions | Yield |
|---|---|
| In methanol for 24h; Heating; | 100% |
-
-
111-71-7
heptanal

-
-
105-45-3
acetoacetic acid methyl ester

-
-
869211-56-3
5-hydroxy-3-oxo-undecanoic acid methyl ester

| Conditions | Yield |
|---|---|
| Stage #1: acetoacetic acid methyl ester With sodium hydride In tetrahydrofuran at 0℃; for 0.166667h; Stage #2: With n-butyllithium In tetrahydrofuran; hexane at 20℃; for 0.333333h; Stage #3: heptanal In tetrahydrofuran; hexane at -78 - 20℃; for 1.08333h; | 100% |
| Stage #1: acetoacetic acid methyl ester With sodium hydride In tetrahydrofuran at 0℃; Stage #2: With n-butyllithium In tetrahydrofuran at -78℃; Stage #3: heptanal In tetrahydrofuran Further stages.; |
-
-
105-45-3
acetoacetic acid methyl ester

-
-
78-84-2
isobutyraldehyde

-
-
55848-84-5
5-hydroxy-6-methyl-3-oxo-heptanoic acid methyl ester

| Conditions | Yield |
|---|---|
| Stage #1: acetoacetic acid methyl ester With sodium hydride In tetrahydrofuran at 0℃; for 0.166667h; Stage #2: With n-butyllithium In tetrahydrofuran; hexane at 20℃; for 0.333333h; Stage #3: isobutyraldehyde In tetrahydrofuran; hexane at -78 - 20℃; for 1.08333h; | 100% |
| Stage #1: acetoacetic acid methyl ester With lithium diisopropyl amide In tetrahydrofuran; hexane at -60 - 20℃; for 0.416667h; Weiler method; Inert atmosphere; Stage #2: isobutyraldehyde In tetrahydrofuran; hexane at -60 - 20℃; for 4h; Weiler method; Inert atmosphere; Stage #3: With water; ammonium chloride In tetrahydrofuran; hexane Inert atmosphere; | 95% |
| Stage #1: acetoacetic acid methyl ester With sodium hydride In tetrahydrofuran at 0℃; Stage #2: With n-butyllithium In tetrahydrofuran at -78℃; Stage #3: isobutyraldehyde In tetrahydrofuran Further stages.; |
-
-
120417-13-2
(S)-2-(tert-butyldimethylsilyloxy)-1-phenylethylamine

-
-
105-45-3
acetoacetic acid methyl ester

-
-
725250-70-4
(Z)-3-[(S)-2-(tert-Butyl-dimethyl-silanyloxy)-1-phenyl-ethylamino]-but-2-enoic acid methyl ester

| Conditions | Yield |
|---|---|
| In methanol for 24h; Heating; | 100% |
| In methanol |

| Conditions | Yield |
|---|---|
| With hydrogen; palladium on activated charcoal In ethyl acetate at 20℃; | 100% |

| Conditions | Yield |
|---|---|
| With silica-supported ferric hydrogensulfate/[Fe(HSO4)3*SiO2] In neat (no solvent) at 20℃; for 0.2h; regioselective reaction; | 100% |
| zirconium(IV) chloride at 20℃; for 0.25h; | 95% |
| With sulfated zirconia at 20℃; for 0.416667h; | 92% |

| Conditions | Yield |
|---|---|
| With N-Bromosuccinimide; 2,2'-azobis(isobutyronitrile) In benzene at 20℃; for 2h; Inert atmosphere; | 100% |
| With N-Bromosuccinimide; toluene-4-sulfonic acid In dichloromethane at 20℃; for 0.416667h; | 94% |
| With ammonium cerium(IV) nitrate at 25 - 28℃; for 2.5h; | 87% |

| Conditions | Yield |
|---|---|
| Stage #1: acetoacetic acid methyl ester With sodium hydride In tetrahydrofuran at 0℃; for 0.166667h; Stage #2: With n-butyllithium In tetrahydrofuran; hexane at 20℃; for 0.333333h; Stage #3: nonan-1-al In tetrahydrofuran; hexane at -78 - 20℃; for 1.08333h; | 100% |
| Stage #1: acetoacetic acid methyl ester With sodium hydride In tetrahydrofuran; mineral oil at 0 - 5℃; for 0.166667h; Stage #2: With n-butyllithium In tetrahydrofuran; mineral oil at 5℃; for 0.166667h; Stage #3: nonan-1-al In tetrahydrofuran; mineral oil at 5 - 10℃; for 0.5h; |
-
-
14221-01-3
tetrakis(triphenylphosphine) palladium(0)

-
-
341-02-6
trityl tetrafluoroborate

-
-
105-45-3
acetoacetic acid methyl ester

-
-
80583-72-8
bis(triphenylphosphine)(methyl acetylacetato)palladium(II) tetrafluoroborate

| Conditions | Yield |
|---|---|
| In acetone; benzene (N2); a soln. of CPh3BF4 and methyl acetoacetate in acetone was added to a soln. of the Pd complex in benzene, the soln. was agitated for 48 h; recrystn. from acetone-ether; elem. anal.; | 100% |


| Conditions | Yield |
|---|---|
| Stage #1: acetoacetic acid methyl ester With sodium hydride In tetrahydrofuran; mineral oil at 0 - 25℃; for 1h; Inert atmosphere; Stage #2: benzyl halide In tetrahydrofuran; mineral oil at 0℃; | 100% |
| Stage #1: acetoacetic acid methyl ester With sodium hydride In tetrahydrofuran; mineral oil at 20℃; Inert atmosphere; Stage #2: benzyl halide In tetrahydrofuran; mineral oil at 20 - 40℃; Inert atmosphere; | |
| Stage #1: acetoacetic acid methyl ester With sodium hydride In tetrahydrofuran; mineral oil at 0 - 20℃; Inert atmosphere; Stage #2: benzyl halide In tetrahydrofuran; mineral oil for 12h; Inert atmosphere; Reflux; |
-
-
504-29-0
2-aminopyridine

-
-
105-45-3
acetoacetic acid methyl ester

-
-
1693-94-3
2-methyl-4H-pyrido[1,2-a]pyrimidin-4-one

| Conditions | Yield |
|---|---|
| With bismuth(III) chloride at 100℃; for 3h; Catalytic behavior; Solvent; Temperature; Reagent/catalyst; Green chemistry; | 100% |
| With trichlorophosphate |
-
-
57260-73-8
N-BOC-1,2-diaminoethane

-
-
105-45-3
acetoacetic acid methyl ester

-
-
1240195-07-6
(Z)-methyl 3-((2-((tert.-butoxycarbonyl)amino)ethyl)amino)but-2-enoate

| Conditions | Yield |
|---|---|
| With silica gel at 20℃; for 16h; | 100% |
| With silica gel In dichloromethane at 10 - 20℃; for 1h; | 127.9 g |
| With silica gel In dichloromethane at 10 - 20℃; for 1h; | 127.9 g |
Methyl acetoacetate Consensus Reports
Methyl acetoacetate Specification
The Butanoic acid, 3-oxo-,methyl ester, with CAS registry number of 105-45-3, is also known as 3-Oxobutanoic acid methyl ester. Its EINECS registry number is 203-299-8. In addition, the formula is C5H8O3 and the molecular weight is 116.12. What's more, its systematic name is Methyl 3-oxobutanoate. Its classification code is Skin / Eye Irritant. This chemical is a colorless transparent liquid and should be stored in a cool, ventilated place away from fire and heat. It is used as a mixed solvent of cellulose acetate. It is also used as an intermediate for pesticides, pharmaceuticals and organic synthesis.
Physical properties about Butanoic acid, 3-oxo-,methyl ester are: (1)ACD/LogP: 0.19; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): 0.18; (4)ACD/LogD (pH 7.4): 0.18; (5)ACD/BCF (pH 5.5): 1; (6)ACD/BCF (pH 7.4): 1; (7)ACD/KOC (pH 5.5): 30.03; (8)ACD/KOC (pH 7.4): 30.01; (9)#H bond acceptors: 3; (10)#H bond donors: 0; (11)#Freely Rotating Bonds: 3; (12)Polar Surface Area: 43.37 Å2; (13)Index of Refraction: 1.399; (14)Molar Refractivity: 27.01 cm3; (15)Molar Volume: 111.6 cm3; (16)Polarizability: 10.71×10-24cm3; (17)Surface Tension: 29.7 dyne/cm; (18)Density: 1.039 g/cm3; (19)Flash Point: 61.6 °C; (20)Enthalpy of Vaporization: 40.58 kJ/mol; (21)Boiling Point: 169.4 °C at 760 mmHg; (22)Vapour Pressure: 1.54 mmHg at 25°C.
Preparation of Methyl acetylacetate: it is prepared by reaction of diketene with methanol. The reaction mixture is esterificated with catalyst of concentrated sulfuric acid. Then product is obtained by crude distillation and distillation. Besides, diketene is derived from cleavage of acetic acid.
C4H4O2 + CH3OH → C5H8O3
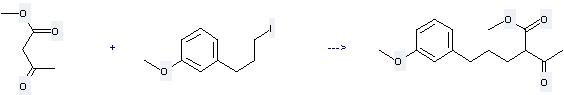
Uses of Butanoic acid, 3-oxo-,methyl ester: it can be used to produce 2-acetyl-5-(3-methoxy-phenyl)-pentanoic acid methyl ester by heating. It will need reagent K2CO3 and solvent acetone. The yield is about 67%.
When you are using this chemical, please be cautious about it as the following:
It is irritating to eyes. In case of contact with eyes, you should rinse immediately with plenty of water and seek medical advice.
You can still convert the following datas into molecular structure:
(1)Canonical SMILES: CC(=O)CC(=O)OC
(2)InChI: InChI=1S/C5H8O3/c1-4(6)3-5(7)8-2/h3H2,1-2H3
(3)InChIKey: WRQNANDWMGAFTP-UHFFFAOYSA-N
The toxicity data is as follows:
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| rat | LD50 | oral | 3228mg/kg (3228mg/kg) | Journal of Industrial Hygiene and Toxicology. Vol. 30, Pg. 63, 1948. |
Related Products
- Methyl 1-Benzyl-5-oxopyrrolidine-3-carboxylate
- Methyl (+)-(3R)-7-[4-(4-fluorophenyl)-6-isopropyl-2-(N-methyl-N-methanesulfonylamino)pyrimidin-5-yl]-3-hydroxy-5-oxo-(6E)-heptenoate
- Methyl (2-amino-5-methyl-1,3-thiazol-4-yl)acetate
- Methyl (2-chloromethyl)oxazole-4-carboxylate
- Methyl (2E)-3-(4-methylphenyl)propenoate
- Methyl (2E)-3-cyclohexylprop-2-enoate
- Methyl (2R)-2-[(tert-butoxycarbonyl)amino]-3-iodopropanoate
- Methyl (2R)-2-[4-(2,4-dichlorophenoxy)phenoxy]propanoate
- Methyl (2R)-2-amino-2-cyclohexylethanoate hydrochloride
- Methyl (2R)-2-bromo-2-(2-chlorophenyl)acetate
- 10545-58-1
- 10545-99-0
- 105462-24-6
- 105-46-4
- 10546-65-3
- 105469-21-4
- 10548-10-4
- 10548-46-6
- 105-48-6
- 105486-72-4
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View