-
Name
1,3-Dibromobenzene
- EINECS 203-574-2
- CAS No. 108-36-1
- Article Data68
- CAS DataBase
- Density 1.937 g/cm3
- Solubility insoluble in water
- Melting Point -7 °C
- Formula C6H4Br2
- Boiling Point 217.537 °C at 760 mmHg
- Molecular Weight 235.906
- Flash Point 93.889 °C
- Transport Information UN 2711
- Appearance clear colourless to light yellow liquid
- Safety 26-36-24/25
- Risk Codes 36/37/38
-
Molecular Structure
-
Hazard Symbols
 Xi,
Xi, C
C
- Synonyms Benzene,m-dibromo- (8CI);m-Dibromobenzene;Benzene, 1,3-dibromo-;
- PSA 0.00000
- LogP 3.21160
Synthetic route

-
-
108-36-1
1,3-dibromobenzene

| Conditions | Yield |
|---|---|
| With dihydrogen peroxide In tetrahydrofuran for 0.333333h; Heating; | 92% |
| Multi-step reaction with 2 steps 1: 95 percent / aq. NaOH / 0.5 h / 0 - 5 °C 2: 11 percent / aq. HI; HBF4 / acetonitrile / 24 h / 60 °C View Scheme | |
| Multi-step reaction with 2 steps 1: 95 percent / aq. NaOH / 0.5 h / 0 - 5 °C 2: 12 percent / aq. HCl; Cu / acetonitrile / 0.25 h / 20 - 25 °C View Scheme |

| Conditions | Yield |
|---|---|
| With bromine; dibromoisocyanuric acid In dichloromethane at 20℃; for 24h; UV-irradiation; | 92% |
-
-
109-72-8, 29786-93-4
n-butyllithium

-
-
17197-37-4
(3,5-dibromophenyl)trimethyltin

-
B
-
108-36-1
1,3-dibromobenzene

-
C
-
1527-99-7
n-butyltrimethyltin

-
D
-
17113-76-7
(3-bromophenyl)trimethylstannane

| Conditions | Yield |
|---|---|
| In diethyl ether; hexane byproducts: C4H9Br; n-BuLi in hexane adding to an ether soln. of the stannane at -78°C, stirring for 30 min, hydrolyzing with 2 N HCl, org. layer sepg., drying over MgSO4; identified by gas chromy. and chromato-mass spectrometry; | A 1% B 82% C n/a D 13% |


| Conditions | Yield |
|---|---|
| Stage #1: 2,6-dibromoaniline With sulfuric acid; sodium nitrite In water at 0 - 5℃; for 3h; Stage #2: With hypophosphorous acid In water at 70 - 80℃; for 3h; Reagent/catalyst; Temperature; | 81% |
| Diazotization.Behandlung der Diazoniumsalz-Loesungen mit H3PO2 bei 5grad; | |
| With tetrahydrofuran; n-Amyl nitrite |

-
-
108-36-1
1,3-dibromobenzene

| Conditions | Yield |
|---|---|
| With tetrabutylammomium bromide; copper In acetonitrile at 20℃; for 0.75h; Substitution; | 80% |
| With tetrabutylammomium bromide In acetonitrile at 60℃; for 0.75h; Substitution; | 78% |

| Conditions | Yield |
|---|---|
| With tetrafluoroboric acid; hydrogen iodide In acetonitrile at 60℃; for 24h; | A 11% B 79% |

| Conditions | Yield |
|---|---|
| With hydrogenchloride; copper In acetonitrile at 20 - 25℃; for 0.25h; | A 12% B 79% |
-
-
66107-31-1
3-bromophenyl trifluoromethanesulfonate

-
-
108-36-1
1,3-dibromobenzene

| Conditions | Yield |
|---|---|
| With tris-(dibenzylideneacetone)dipalladium(0); t-BuBrettPhos; triisobutylaluminum; butanone; potassium bromide In toluene at 100℃; for 24h; Inert atmosphere; | 78% |
-
-
89598-96-9
(3-bromophenyl)boronic acid

-
-
108-36-1
1,3-dibromobenzene

| Conditions | Yield |
|---|---|
| With N-Bromosuccinimide In acetonitrile at 81℃; for 8h; | 65% |

| Conditions | Yield |
|---|---|
| at 450℃; under 0.0001 Torr; for 4h; Title compound not separated from byproducts; | A n/a B 60% C n/a |


| Conditions | Yield |
|---|---|
| With Bromotrichloromethane; bromine; dibromoisocyanuric acid at 120℃; for 60h; Time; UV-irradiation; | 17% |
-
-
56-23-5
tetrachloromethane

-
-
128-08-5
N-Bromosuccinimide

-
-
110-83-8
cyclohexene

-
-
94-36-0
dibenzoyl peroxide

-
A
-
108-36-1
1,3-dibromobenzene

-
B
-
106-37-6
1.4-dibromobenzene


| Conditions | Yield |
|---|---|
| With aluminium trichloride | |
| With aluminium trichloride |

| Conditions | Yield |
|---|---|
| With sulfuric acid; sodium nitrite |

| Conditions | Yield |
|---|---|
| With ethyl nitrite | |
| Diazotization.Behandlung der Diazoniumsalz-Loesungen mit H3PO2 bei 5grad; | |
| ueberfuehrung in das Diazoniumnitrat und erhitzen desselben mit Wasser; |
-
-
67713-23-9
2,6-dibromobenzaldehyde

-
-
108-36-1
1,3-dibromobenzene

| Conditions | Yield |
|---|---|
| With potassium hydroxide |

| Conditions | Yield |
|---|---|
| With aluminium trichloride beim Bromieren; |
-
-
108-86-1
bromobenzene

-
A
-
108-36-1
1,3-dibromobenzene

-
B
-
106-37-6
1.4-dibromobenzene

-
C
-
583-53-9
1,2-dibromobenzene

| Conditions | Yield |
|---|---|
| With hypobromous acid In 1,4-dioxane; water at 25℃; Rate constant; | A 1 % Chromat. B 64.3 % Chromat. C 35.7 % Chromat. |


| Conditions | Yield |
|---|---|
| With bromobenzene; Cu-HZSM-5 zeolite at 399.9℃; Mechanism; Product distribution; | A 11.6 % Chromat. B 3.2 % Chromat. |
-
-
608-21-9
1,2,3-tribromobenzene

-
-
71-43-2
benzene

-
A
-
108-36-1
1,3-dibromobenzene

-
B
-
59080-32-9
2,6-dibromo-1,1'-biphenyl

| Conditions | Yield |
|---|---|
| at 25℃; for 24h; Irradiation; | A 9.5 % Chromat. B 14.0 % Chromat. C 12.0 % Chromat. D 3.3 % Chromat. |

-
-
615-54-3
1,2,4-tribromobenzene

-
A
-
108-86-1
bromobenzene

-
B
-
108-36-1
1,3-dibromobenzene

-
C
-
106-37-6
1.4-dibromobenzene

-
D
-
71-43-2
benzene

-
E
-
583-53-9
1,2-dibromobenzene

| Conditions | Yield |
|---|---|
| With potassium hydroxide; hydrogen; palladium on activated charcoal In 2,2,4-trimethylpentane at 50℃; for 0.666667h; Product distribution; var. cat.: Raney-Ni, add. of Aliquat 336; | A 18 % Chromat. B 3 % Chromat. C 1 % Chromat. D 42 % Chromat. E 30 % Chromat. |


| Conditions | Yield |
|---|---|
| With n-butyllithium 1.) Et2O, -78 deg C, 2.) Et2O, -78 deg C; Multistep reaction; |


| Conditions | Yield |
|---|---|
| With n-butyllithium 1.) Et2O, -78 deg C, 2.) Et2O, -78 deg C; Multistep reaction; |


| Conditions | Yield |
|---|---|
| With n-butyllithium 1.) Et2O, -78 deg C, 2.) Et2O, -78 deg C; Multistep reaction; | |
| With hydrocarbon substituted imidazolidine at 20℃; | |
| With 1,3-bis(1,1-dimethylethyl)imidazolidin-2-ylidene In 1,4-dioxane at 120℃; for 4h; | |
| With 1,2,3,4-tetrakis(carbazol-9-yl)-5,6-dicyanobenzene; triethylamine In 1-methyl-pyrrolidin-2-one at 20℃; for 1.33333h; Inert atmosphere; Irradiation; | 98 %Chromat. |
-
-
626-39-1
1,3,5-trisbromobenzene

-
-
141-78-6
ethyl acetate

-
A
-
108-36-1
1,3-dibromobenzene

-
B
-
14401-73-1
3,5-dibromoacetophenone

| Conditions | Yield |
|---|---|
| With n-butyllithium 1.) Et2O, -78 deg C, 2.) Et2O, -78 deg C; Yield given. Multistep reaction; | |
| With n-butyllithium 1.) Et2O, -78 deg C, 2.) Et2O, -78 deg C; Multistep reaction; |


| Conditions | Yield |
|---|---|
| With n-butyllithium 1.) Et2O, -78 deg C, 2.) Et2O, -78 deg C; Multistep reaction; |


| Conditions | Yield |
|---|---|
| With 1-sulfanyl-2-(dimethylaminomethyl)-3-Me3Si-benzene-Cu(I); potassium carbonate In 1-methyl-pyrrolidin-2-one at 160℃; for 16h; | 100% |
| With copper(l) iodide; potassium carbonate In dimethyl sulfoxide at 150℃; for 48h; | 99% |
| With copper(l) iodide; dimethylaminoacetic acid; potassium carbonate In dimethyl sulfoxide at 110℃; for 48h; Inert atmosphere; | 93% |
-
-
108-36-1
1,3-dibromobenzene

-
-
73183-34-3
bis(pinacol)diborane

-
-
196212-27-8
4,4,5,5-tetramethyl-2-[3-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)phenyl]-1,3,2-dioxaborolane

| Conditions | Yield |
|---|---|
| With dichloro(1,1'-bis(diphenylphosphanyl)ferrocene)palladium(II)*CH2Cl2; potassium acetate In N,N-dimethyl-formamide at 90℃; for 48h; Inert atmosphere; Schlenk technique; | 100% |
| With (1,1'-bis(diphenylphosphino)ferrocene)palladium(II) dichloride; potassium acetate In N,N-dimethyl-formamide at 80℃; for 16h; Inert atmosphere; Schlenk technique; | 96% |
| With (1,1'-bis(diphenylphosphino)ferrocene)palladium(II) dichloride; potassium acetate In N,N-dimethyl-formamide at 80℃; for 24h; Suzuki-Miyaura Coupling; | 93% |
-
-
108-36-1
1,3-dibromobenzene

-
-
536-90-3
m-Anisidine

-
-
202127-21-7
N,N'-bis(3-methoxyphenyl)-1,3-benzenediamine

| Conditions | Yield |
|---|---|
| With sodium t-butanolate; palladium diacetate; bis(diphenylphosphinyl)ferrocene In toluene at 100 - 110℃; for 12h; | 100% |
| With [(π-cinnamyl)Pd(tBuXPhos)]OTf In water at 45℃; for 36h; | 91% |
-
-
108-36-1
1,3-dibromobenzene

-
-
196960-96-0
3-(t-butyl)-4-methoxyphenyl boronic acid

-
-
1202392-73-1
1,3-bis(3-tert-butyl-4-methoxyphenyl)benzene

| Conditions | Yield |
|---|---|
| With tetrakis(triphenylphosphine) palladium(0); barium hydroxide monohydrate In ISOPROPYLAMIDE; water at 80℃; for 48h; Suzuki-Miyaura double cross-coupling; Inert atmosphere; | 100% |

-
-
108-36-1
1,3-dibromobenzene

-
-
74-88-4
methyl iodide

-
-
778582-06-2
3-benzyl-8-(3-bromo-phenyl)-8-methoxy-3-aza-bicyclo[3.2.1]octane

| Conditions | Yield |
|---|---|
| Stage #1: 1,3-dibromobenzene With n-butyllithium In hexanes; diethyl ether at -78℃; for 1h; Stage #2: (1R,5S)-3-benzyl-3-azabicyclo[3.2.1]octan-8-one In hexanes; diethyl ether at -78℃; for 0.75h; Stage #3: methyl iodide In tetrahydrofuran at 20℃; for 60h; | 100% |


-
-
108-36-1
1,3-dibromobenzene

-
-
778581-93-4
3-benzyl-8-(3-bromo-phenyl)-8-hydroxy-3-aza-bicyclo[3.2.1]octane

| Conditions | Yield |
|---|---|
| Stage #1: 1,3-dibromobenzene With n-butyllithium In hexanes; diethyl ether at -78℃; for 1.33333h; Stage #2: (1R,5S)-3-benzyl-3-azabicyclo[3.2.1]octan-8-one In hexanes; diethyl ether at -78 - 20℃; Stage #3: With ammonium chloride In water at 20℃; for 18h; | 100% |
-
-
108-36-1
1,3-dibromobenzene

-
-
1160604-94-3
5-{4,4,5,5-tetramethyl-1,3,2-(2-dioxaborolanyl)}-2-{(E)-2-phenyl-2-diphenylborylethenyl}pyridine

-
-
1160604-69-2
1,3-bis[5-{(E)-2-(2-phenyl-2-diphenyl-borylethenyl)pyridyl}]benzene

| Conditions | Yield |
|---|---|
| With sodium hydroxide; bis(tri-t-butylphosphine)palladium(0) In tetrahydrofuran; water at 60℃; for 12h; Inert atmosphere; | 100% |

| Conditions | Yield |
|---|---|
| With dicyclohexyl-(2',6'-dimethoxybiphenyl-2-yl)-phosphane; potassium phosphate; palladium diacetate In tetrahydrofuran; water at 65℃; for 12h; Suzuki-Miyaura coupling; | 100% |
-
-
108-36-1
1,3-dibromobenzene

-
-
10365-98-7
3-methoxyphenylboronic acid

-
-
171820-21-6
3,3''-dimethoxy-1,1':3',1''-terphenyl

| Conditions | Yield |
|---|---|
| With potassium carbonate In ethanol at 80℃; for 36h; Suzuki-Miyaura Coupling; Inert atmosphere; | 100% |

| Conditions | Yield |
|---|---|
| With C24H12Cu2F9N4O7; tetrabutylammomium bromide; ammonia; caesium carbonate In water at 110 - 140℃; for 16h; | 99% |
| With {(o-C6H4(NCHC6H4O)2)cobalt(II)}2; ammonia at 150℃; under 1500.15 Torr; for 10h; Autoclave; Inert atmosphere; Large scale; | 73% |
| With ammonia; water; copper(II) sulfate at 175 - 180℃; |
-
-
108-36-1
1,3-dibromobenzene

-
-
59020-06-3
4-trimethylstannyl-pyridine

-
-
170165-79-4
1,3-bis(4-pyridyl)benzene

| Conditions | Yield |
|---|---|
| With bis-triphenylphosphine-palladium(II) chloride; lithium chloride In toluene at 110℃; | 99% |
-
-
108-36-1
1,3-dibromobenzene

-
-
68-12-2, 33513-42-7
N,N-dimethyl-formamide

-
-
3132-99-8
m-bromobenzoic aldehyde

| Conditions | Yield |
|---|---|
| Stage #1: 1,3-dibromobenzene With tri-n-butyllithium magnesate complex In toluene at 0℃; for 1.5h; Stage #2: N,N-dimethyl-formamide In toluene at 0℃; for 0.5h; Stage #3: With citric acid at 20℃; | 99% |
| Stage #1: 1,3-dibromobenzene With n-butyllithium; isopropylmagnesium chloride In tetrahydrofuran; hexane at 0 - 5℃; for 1h; Stage #2: N,N-dimethyl-formamide In tetrahydrofuran at 0℃; for 1h; Further stages.; | 90% |
| Stage #1: 1,3-dibromobenzene With n-butyllithium In tetrahydrofuran at -78℃; for 0.583333h; Stage #2: N,N-dimethyl-formamide In tetrahydrofuran at -78 - 20℃; for 1.5h; | 63.21% |

| Conditions | Yield |
|---|---|
| With tris-(dibenzylideneacetone)dipalladium(0); 2,2'-bis-(diphenylphosphino)-1,1'-binaphthyl; sodium t-butanolate In toluene at 80℃; for 4.5h; Sealed tube; | 99% |
| With tris-(dibenzylideneacetone)dipalladium(0); 2,2'-bis-(diphenylphosphino)-1,1'-binaphthyl; sodium t-butanolate In toluene at 80℃; for 4.5h; Sealed tube; | 99% |
| With 2,2'-bis-(diphenylphosphino)-1,1'-binaphthyl; sodium t-butanolate; tris-(dibenzylideneacetone)dipalladium(0) In toluene at 80℃; for 4.5h; | 78% |
-
-
109-72-8, 29786-93-4
n-butyllithium

-
-
75-77-4
chloro-trimethyl-silane

-
-
108-36-1
1,3-dibromobenzene

-
-
17878-47-6
(3-bromophenyl)trimethylsilane

| Conditions | Yield |
|---|---|
| In water | 99% |

-
-
108-36-1
1,3-dibromobenzene

-
-
100-61-8
N-methylaniline

-
-
308123-55-9
N1,N3-dimethyl-N1,N3-diphenylbenzene-1,3-diamine

| Conditions | Yield |
|---|---|
| With (6-Dipp)PdCl2-SPhos; sodium t-butanolate In neat (no solvent) at 110℃; for 24h; Buchwald-Hartwig Coupling; | 99% |
| With potassium hexamethylsilazane In 1,4-dioxane at 100℃; for 1h; metal-free Buchwald-Hartwig amination; Inert atmosphere; | 96 %Chromat. |
-
-
108-36-1
1,3-dibromobenzene

-
-
53078-71-0
4-methoxy-3-methylbenzonitrile

-
-
1260433-04-2
(3-bromophenyl)(4-methoxy-3-methylphenyl)methanimine

| Conditions | Yield |
|---|---|
| Stage #1: 1,3-dibromobenzene With n-butyllithium In diethyl ether at -78℃; for 0.5h; Stage #2: 4-methoxy-3-methylbenzonitrile In diethyl ether at -78 - 20℃; for 2h; Stage #3: With ammonium acetate | 99% |
-
-
108-36-1
1,3-dibromobenzene

-
-
243990-53-6
(4-(benzyloxy)-3-methoxyphenyl)boronic acid

| Conditions | Yield |
|---|---|
| With (1,1'-bis(diphenylphosphino)ferrocene)palladium(II) dichloride; potassium carbonate In N,N-dimethyl-formamide at 60℃; Suzuki Coupling; Inert atmosphere; | 99% |
-
-
108-36-1
1,3-dibromobenzene

-
-
96843-23-1
1,5-dibromo-2,4-diiodobenzene

| Conditions | Yield |
|---|---|
| With sulfuric acid; iodine at 125 - 135℃; for 48h; Inert atmosphere; | 98.6% |
| With sulfuric acid; iodine at 125 - 135℃; for 6h; | 70% |
| With sulfuric acid; iodine at 130℃; for 6h; Inert atmosphere; Schlenk technique; | 60% |

| Conditions | Yield |
|---|---|
| With sodium t-butanolate; tris(dibenzylideneacetone)dipalladium (0); 2,2'-bis-(diphenylphosphino)-1,1'-binaphthyl In toluene at 100℃; for 24h; | 98% |
| With C30H25BrN3PPdS; potassium carbonate In iso-butanol at 100℃; for 24h; Buchwald-Hartwig Coupling; Inert atmosphere; | 77.3% |
| With potassium hexamethylsilazane In 1,4-dioxane at 100℃; for 0.5h; metal-free Buchwald-Hartwig amination; Inert atmosphere; | 86 %Chromat. |
-
-
108-36-1
1,3-dibromobenzene

-
-
5720-07-0
4-methoxyphenylboronic acid

-
-
1568-74-7
4,4’’-dimethoxy-1,1′:3′,1’’-terphenyl

| Conditions | Yield |
|---|---|
| With potassium carbonate In ethanol; water at 20℃; for 4h; Suzuki-Miyaura Coupling; | 98% |
| With potassium phosphate; C17H15N4O3S(1-)*Na(1+); palladium dichloride In water at 100℃; for 6h; Suzuki-Miyaura Coupling; Inert atmosphere; Green chemistry; | 92% |
| With sodium carbonate; palladium diacetate In water; N,N-dimethyl-formamide at 60℃; for 12h; Suzuki; | 90% |
-
-
108-36-1
1,3-dibromobenzene

-
-
195730-31-5
4-bicyclo[4.2.0]octa-1,3,5-trienylboronic acid

| Conditions | Yield |
|---|---|
| With borax; tetrabutylammomium bromide; palladium dichloride In ethanol at 20℃; for 3h; Suzuki coupling; | 98% |
-
-
108-36-1
1,3-dibromobenzene

-
-
73183-34-3
bis(pinacol)diborane

-
-
408492-26-2
2-(3,5-dibromophenyl)-4,4,5,5-tetramethyl-1,3,2-dioxaborolane

| Conditions | Yield |
|---|---|
| With (1,5-cyclooctadiene)(methoxy)iridium(I) dimer; cyclopentyl methyl ether; 1,1'-di(pyridin-2-yl)-1,1',3,3'-tetrahydro-2,2'-bibenzo[d][1,3,2]diazaborole at 100℃; for 16h; Schlenk technique; Inert atmosphere; | 98% |
| Stage #1: bis(pinacol)diborane With C24H28ClIrN2O In n-heptane; isopropyl alcohol at 75℃; for 1h; Sealed tube; Inert atmosphere; Stage #2: 1,3-dibromobenzene In n-heptane; isopropyl alcohol Sealed tube; Inert atmosphere; | 98% |
| With 4,4'-di-tert-butyl-2,2'-bipyridine; [Ir(COD)(OMe)]2 In tetrahydrofuran at 80℃; for 16h; |
-
-
108-36-1
1,3-dibromobenzene

-
-
24424-99-5
di-tert-butyl dicarbonate

-
-
69038-74-0
tert-butyl 3-bromobenzoate

| Conditions | Yield |
|---|---|
| Stage #1: 1,3-dibromobenzene With n-butyllithium; butyl magnesium bromide In tetrahydrofuran; hexane; toluene at -5℃; Stage #2: di-tert-butyl dicarbonate In tetrahydrofuran; hexane; toluene at -5℃; Stage #3: With citric acid In tetrahydrofuran; hexane; toluene Further stages.; | 98% |
| Stage #1: 1,3-dibromobenzene With n-butyllithium; butyl magnesium bromide In tetrahydrofuran; hexane; toluene at -5℃; for 1h; Inert atmosphere; Stage #2: di-tert-butyl dicarbonate In tetrahydrofuran; hexane; toluene at -5℃; for 2h; Inert atmosphere; | 98% |
-
-
108-36-1
1,3-dibromobenzene

-
-
1260433-19-9
4-(difluoromethoxy)-3,5-dimethylbenzonitrile

-
-
1260433-20-2
(3-bromophenyl)(4-(difluoromethoxy)-3,5-dimethylphenyl)methanimine

| Conditions | Yield |
|---|---|
| Stage #1: 1,3-dibromobenzene With n-butyllithium In diethyl ether at -78℃; for 0.5h; Inert atmosphere; Stage #2: 4-(difluoromethoxy)-3,5-dimethylbenzonitrile In tetrahydrofuran; diethyl ether at 0 - 20℃; for 2h; Stage #3: With ammonium acetate In tetrahydrofuran; methanol; diethyl ether | 98% |
-
-
108-36-1
1,3-dibromobenzene

-
-
5720-06-9
2-Methoxyphenylboronic acid

-
-
337535-26-9
3'-bromo-2-methoxybiphenyl

| Conditions | Yield |
|---|---|
| With tetrakis(triphenylphosphine) palladium(0); potassium carbonate In methanol; toluene for 24h; Suzuki reaction; Reflux; Inert atmosphere; | 98% |
| Stage #1: 1,3-dibromobenzene; 2-Methoxyphenylboronic acid With sodium carbonate In ethanol; toluene for 0.5h; Sealed tube; Inert atmosphere; Stage #2: With tetrakis(triphenylphosphine) palladium(0) In ethanol; toluene at 80℃; Inert atmosphere; | 57.76% |
| Stage #1: 1,3-dibromobenzene; 2-Methoxyphenylboronic acid With sodium carbonate In ethanol; toluene for 0.5h; Inert atmosphere; Stage #2: With tetrakis(triphenylphosphine) palladium(0) In ethanol; toluene at 80℃; Sealed tube; Inert atmosphere; | 57.76% |
-
-
108-36-1
1,3-dibromobenzene

-
-
1666-13-3
diphenyl diselenide

-
-
1393715-73-5
3,5-dibromophenyl phenyl selenide

| Conditions | Yield |
|---|---|
| Stage #1: 1,3-dibromobenzene With (1,5-cyclooctadiene)(methoxy)iridium(I) dimer; bis(pinacol)diborane; 4,4'-di-tert-butyl-2,2'-bipyridine In tetrahydrofuran at 80℃; for 24h; Inert atmosphere; Stage #2: diphenyl diselenide With [2,2]bipyridinyl; copper(l) chloride In water; dimethyl sulfoxide at 80℃; for 24h; Inert atmosphere; regioselective reaction; | 98% |
-
-
108-36-1
1,3-dibromobenzene

| Conditions | Yield |
|---|---|
| With 1,1'-bis-(diphenylphosphino)ferrocene; tris-(dibenzylideneacetone)dipalladium(0); sodium t-butanolate In toluene at 130℃; for 24h; Inert atmosphere; Sealed tube; | 98% |

| Conditions | Yield |
|---|---|
| With copper(l) iodide; 1,10-Phenanthroline; potassium carbonate In N,N-dimethyl-formamide; toluene at 135 - 145℃; Ullmann reaction; | 97% |
-
-
108-36-1
1,3-dibromobenzene

-
-
51686-78-3
2,4-dibromonitrobenzene

| Conditions | Yield |
|---|---|
| With sulfuric acid; nitric acid at 0 - 20℃; for 1.5h; | 97% |
| With sulfuric acid; nitric acid In dichloromethane at 0 - 20℃; for 0.5h; | 95% |
| With sulfuric acid; nitric acid at 20℃; for 5h; | 90% |
-
-
108-36-1
1,3-dibromobenzene

-
-
30389-25-4
3,3,4,4,5,5,6,6,7,7,8,8,9,9,10,10,11,11,12,12,12-henicosafluorododec-1-ene

| Conditions | Yield |
|---|---|
| With tetrabutylammomium bromide; sodium acetate; palladium diacetate In tetrahydrofuran; N,N-dimethyl-formamide at 120℃; for 84h; | 97% |
1,3-Dibromobenzene Consensus Reports
1,3-Dibromobenzene Specification
1,3-Dibromobenzene is an organic compound with the formula C6H4Br2, and its systematic name is the same with the product name. With the CAS registry number 108-36-1, it is also named as m-Dibromobenzene. It belongs to the product categories of Benzene derivates; Bromine Compounds; Aryl; C6; Halogenated Hydrocarbons. Its EINECS number is 203-574-2. In addition, the molecular weight is 235.90. This chemical should be sealed and stored in a cool and ventilated place. Moreover, it should be protected from oxides, heat and fire. It is used as solvent and organic synthesis intermediate.
Physical properties of 1,3-Dibromobenzene are: (1)ACD/LogP: 3.715; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): 3.72; (4)ACD/LogD (pH 7.4): 3.72; (5)ACD/BCF (pH 5.5): 392.16; (6)ACD/BCF (pH 7.4): 392.16; (7)ACD/KOC (pH 5.5): 2500.35; (8)ACD/KOC (pH 7.4): 2500.35; (9)#H bond acceptors: 0; (10)#H bond donors: 0; (11)#Freely Rotating Bonds: 0; (12)Index of Refraction: 1.599; (13)Molar Refractivity: 41.633 cm3; (14)Molar Volume: 121.803 cm3; (15)Polarizability: 16.505×10-24cm3; (16)Surface Tension: 41.03 dyne/cm; (17)Density: 1.937 g/cm3; (18)Flash Point: 93.889 °C; (19)Enthalpy of Vaporization: 43.542 kJ/mol; (20)Boiling Point: 217.537 °C at 760 mmHg; (21)Vapour Pressure: 0.19 mmHg at 25°C.
Preparation: this chemical can be prepared by (3-bromo-phenyl)-dihydroxy-borane at the temperature of 81 °C. This reaction will need reagent NBS and solvent acetonitrile with the reaction time of 8 hours. The yield is about 65%.

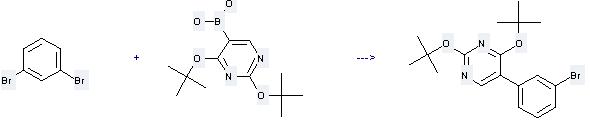
Uses of 1,3-Dibromobenzene: it can be used to produce 5-(3-bromo-phenyl)-2,4-di-tert-butoxy-pyrimidine by heating. It will need reagents 1M aq. NaHCO3, Pd(0)(PPh3)4 and solvent 1,2-dimethoxy-ethane with the reaction time of 1 hour. The yield is about 89%.
When you are using this chemical, please be cautious about it as the following:
This chemcial is irritating to eyes, respiratory system and skin. In case of contact with eyes, you should rinse immediately with plenty of water and seek medical advice. When using it, you must avoid contact with skin and eyes and you need to wear suitable protective clothing.
You can still convert the following datas into molecular structure:
(1)SMILES: Brc1cccc(Br)c1
(2)Std. InChI: InChI=1S/C6H4Br2/c7-5-2-1-3-6(8)4-5/h1-4H
(3)Std. InChIKey: JSRLURSZEMLAFO-UHFFFAOYSA-N
The toxicity data is as follows:
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| mouse | LD50 | intraperitoneal | 900mg/kg (900mg/kg) | Gigiena i Sanitariya. For English translation, see HYSAAV. Vol. 44(12), Pg. 19, 1979. | |
| mouse | LD50 | oral | 2250mg/kg (2250mg/kg) | Gigiena i Sanitariya. For English translation, see HYSAAV. Vol. 44(12), Pg. 19, 1979. |
Related Products
- 10,10'-Bis([1,1'-biphenyl]-4-yl)-9,9'-bianthracene
- 10,10'-Dibromo-9,9'-bianthryl
- 10,10-Dimethylanthrone
- 10,10-Oxybisphenoxarsine
- 10-[1,1'-Biphenyl]-4-yl-2-(1-methylethyl)-9-oxo-9H-thioxanthenium hexafluorophosphate
- 10,11-Dehydroimipramine
- 10,11-Dihydro-11-oxodibenzo[b,f][1,4]thiazepine
- 10,11-Dihydro-2-(4-methyl-1-piperazinyl)-11-(2-athiazolyl)-pyridazino(3,4-b)(1,4)benzoxazepine
- 10,11-DIHYDRO-2-(4-METHYL-1-PIPERAZINYL)-11-(3,4-XYLYL)PYRIDAZINO(3,4b)(1,4)-BENZOXAZEPINE MALEATE
- 10,11-Dihydro-5-(3-dimethylamino-2-methylpropyl)-5h-dibenz (b,f)azepine
- 108365-31-7
- 108-37-2
- 108372-48-1
- 108373-05-3
- 108381-28-8
- 108-38-3
- 108-39-4
- 108-40-7
- 108412-04-0
- 108-41-8
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View